Introduction
Botrytis blight of peanut, caused by Botrytis cinerea Pers., was first described from Maryland and Virginia in 1963 (Orellana and Bailey, 1964) and has subsequently been reported from most peanut producing regions of the world (Porter, 1997). However, this disease has been observed only infrequently and has rarely been considered to be of economic importance. In 2004 there was a substantial outbreak of Botrytis blight in the peanut production area of the high plains of west Texas and in Georgia (Woodward et al., 2005), and again in Texas in 2005. The occurrence in Texas was attributed to an unusual period of cool temperatures with increased rainfall. Average daily maximum temperatures recorded at Lubbock in July and August 2004 were 1 to 3°C lower, with rainfall 58 mm greater during this period than the 30-yr average (www.srh.noaa/lub/climate/). The reported optimal temperature range for development of Botrytis blight is 15 to 20°C (Orellana and Bailey, 1964). The increased occurrence in Georgia was attributed to increased planting of cultivars with resistance to Tomato Spotted Wilt Virus that tend to be of longer maturity than susceptible cultivars (Woodward et al., 2005). The longer time to maturity results in a greater percentage of the crop remaining in the field later into the fall when cool temperatures favor the development of Botrytis blight.
Because of the infrequent occurrence of Botrytis blight in peanut fields in Texas and other production areas in the United States, few data are available on such important considerations as variation among common cultivars in susceptibility to the pathogen and variation in the pathogen population in sensitivity to fungicides currently registered for use on peanut. There are numerous reports of resistance in different populations of B. cinerea to benizimidazole, dicarboximide, and pyrimethanil fungicides on several crops (http://www.pesticides.gov.uk/rags_print.asp?id = 1693).
The objectives of this study were to determine the relative susceptibility of several currently grown peanut cultivars to B. cinerea and to determine variation in sensitivity to fungicides among isolates of B. cinerea collected from peanut in west Texas. A preliminary report of these data has been presented (Starr et al., 2006).
Materials and Methods
Fifty-three isolates of B. cinerea were collected as sclerotia from symptomatic plants in an arbitrary fashion from 11 peanut (Arachis hypogaea) fields in three west Texas counties (Gaines, Terry, and Yoakum). The identity of the isolates was confirmed based on morphometrics of conidia and conidiophores. Isolates were routinely cultured on potato dextrose agar and were stored as dried cultures at 4°C.
Pathogenicity of each isolate was determined by inoculating a 3-mm-diam. agar plug taken from the advancing edge of an actively growing colony onto a fully expanded peanut leaflet in a moisture chamber (Hollowell et al., 2003). Lesion diameters were measure at 6 to 10 d after inoculation (DAI). Initial tests confirmed that 20°C was the optimal temperature for lesion development and all subsequent pathogenicity tests were conducted at that temperature. Initial tests of pathogenicity were conducted using the cultivars Florunner, NemaTam, and Tamrun 96. Variation in susceptibility among eight additional peanut cultivars ab (AT VC2, Flavorunner 458, Jupiter, OLin, NC 7, Tamrun OL02, Tamspan 90, Valencia C) and two breeding lines (Tx607 and Tx630) was tested in a similar manner with nine replications of each treatment in a completely randomized design.
Pathogen sensitivity to the fungicides fluazinam (Omega 500F, Syngenta Crop Science, Greensboro, NC), dicloran (Botran 75W, Gowan Company, Yuma, AZ), and iprodione (Rovral, Bayer Crop Science, Research Triangle Park, NC) was determined based on hyphal growth on PDA amended separately with a commerical formulation each fungicide. In initial tests, the dose response of three isolates of B. cinerea to each fungicide was determined by amending PDA with different concentrations of each fungicide. A 6-mm-diam. agar plug taken from the advancing margin of an actively growing colony was placed in the center of a 10-cm-diam. petri dish containing the desired concentration of the fungicide. Cultures were incubated at 25°C and colony diameters were measure after 3 d. A completely randomized experimental design was used with three replications of each treatment. Data from the dose response experiment were used to select a discriminating concentration that would inhibit hyphal growth by approximately 70%. The growth of additional isolates on the selected discriminating concentration of each fungicide was then tested in a similar manner.
Data were subjected to analysis of variance using the SAS ver. 9.1 (SAS, Inc, Cary, NC) general linear model procedure. Mean separations, when appropriate, were by least significant differences. Correlation analyses were by the SAS correlation procedure. The fungicide dose response curves were fitted using the regression analysis function of SigmaPlot ver. 8.0 (SPSS, Inc., Chicago, IL).
Results
In preliminary pathogenicity tests, 34 isolates of B. cinerea caused rapidly expanding, necrotic lesions on detached leaflets from peanut cultivars Florunner, NemaTam, and Tamrun 96. These lesions encompassed the entire leaflet by 6 to 7 DAI with conidia production usually observed at 10 to 14 DAI. Infected leaf tissue was typically completely destroyed by 14 DAI. Three other isolates caused smaller lesions that were only 1 to 2 cm diameter at 6 DAI. Sixteen isolates were nonpathogenic and did not induce any symptoms of disease.
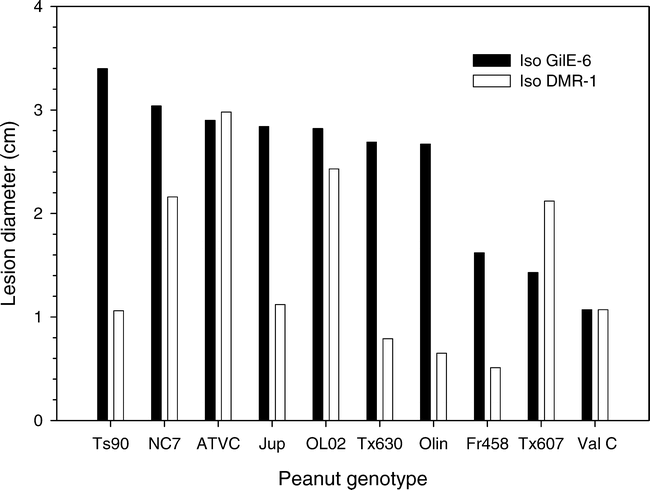
Variation in susceptibility of eight additional peanut cultivars and breeding lines was tested using one arbitrarily selected aggressive isolate (GilE-6) and one weakly aggressive isolate (DM1-R). In those tests Flavorunner 458, Valencia C, and TX 607 had smaller lesions (P = 0.001) that were 1.07 to 1.62-cm-diam at 6 DAI whereas the lesion diameter for the other peanut genotypes were >2.67-cm-diam. (Fig. 1). Across all treatments the mean lesion diameter for genotypes inoculated with the aggressive isolate GilE-6 was larger (2.45-cm-diam., P = 0.001) at 6 DAI than when the genotypes were inoculated with the less aggressive isolate DM1-R (1.49-cm-diam.). In all cases, however, the lesions eventually encompassed the entire leaflet. Despite this overall difference in aggressiveness between the two pathogen isolates, a significant isolate by genotype interaction was observed (P = 0.001). Lesion sizes on ATVC and Jupiter were similar for the two pathogen isolates, and the lesions induced by isolate DM1-R on the breeding line TX 607 were larger than those induced by isolate GilE-6 (Fig. 1).
Variation in lesion diameter among six peanut cultivars and two breeding lines when detacted leaflets were inoculated with either an aggressive (GilE-6) or weakly aggressive (DM1-R) isolate of Botrytis cinerea.
The LSD0.05 across both isolates was 0.40, the LSD0.05 values for GilE-6 and DM1-R individually were 0.36 and 0.75, respectively.
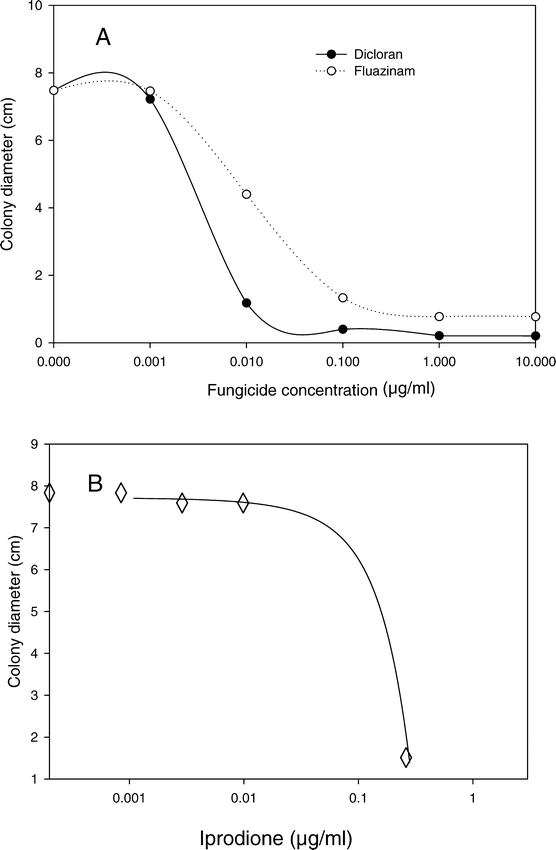
In the determination of the dose response of B. cinerea to different fungicides, response to fluazinam and dicloran was measured in one experiment. Dose response to iprodione was measured in a separate experiment. In each experiment one of the three arbitrarily selected pathogen isolates (GilE-7) had a growth rate that was slightly less (71% to 88%, P = 0.0010) than growth of the other two isolates (DM12 and SE6), but no isolate by fungicide interaction was observed and the data from the different isolates were pooled to determine the overall dose response. The growth responses of B. cinerea to different concentrations of dicloran and fluazinam were best described by negative exponential functions (Fig. 2), with coefficients of determination greater than 0.94. Variation in growth of the same three isolates on different concentrations of iprodione was best described as a negative quadratic model with a coefficient of determination of 0.99 (Fig. 2). Based on these data, fungicide concentrations of 3.0 µg dicloran/ml, 0.1 µg fluazinam/ml, and 0.28 µg iprodione/ml were used to test several isolates of B. cinerea for variation in sensitivity to these fungicides.
Growth response of Botrytis cinerea potato dextrose agar amended separately with different concentrations of three fungicides.
Data are pooled from three isolates, with three replicates of each isolate for each fungicide treatment. A. Exponential decay models for dicloran (Y = 7.5 e−25.7X, r2 = 0.97) and fluazinam (Y = 7.6e−51.7X, r2 = 0.94). B. Quadratic model for iprodione (Y = 17.7 − 10.2 X − 43.8 X2, r2 = 0.99).
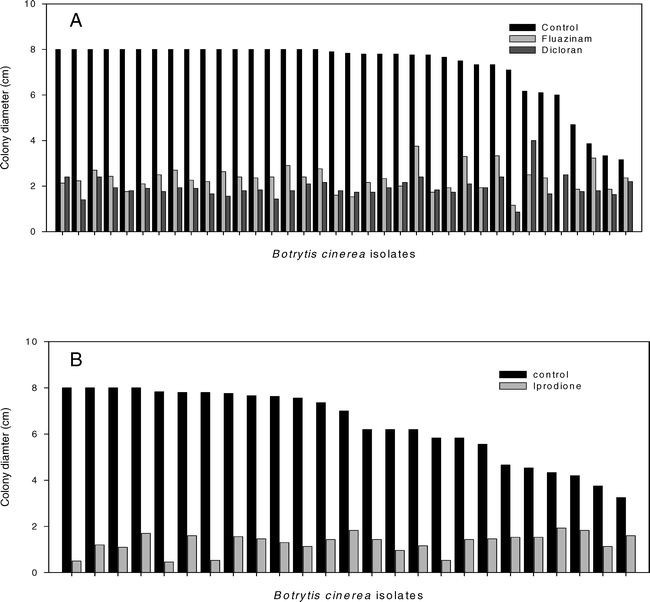
In the tests for variation in sensitivity to dicloran and fuazinam, the colony diameters of the 47 isolates tested varied from 1.0 to 8.0-cm at 3 DAI (P = 0.01), 10 isolates for which growth was <3.0 cm were excluded from further analysis based on abnormally slow growth. Growth of the remaining isolates on PDA amended with dicloran or fluazinam varied, with colony diameters ranging from 0.9 to 4.0 cm and 0.5 to 4.8 cm (P = 0.001), respectively (Fig. 3). For each of these fungicides the ratio of growth on the fungicide amended medium to growth on the unamended PDA varied from 0.12 to 0.69 for dicloran (P = 0.001) and from 0.08 to 0.83 for fluazinam (P = 0.001). A negative correlation between the ratio of growth on fungicide-amended medium to growth on unamended PDA and colony diameters on unamended PDA was observed for both dicloran (r = −0.78, P = 0.001) and fluazinam (r = −0.62, P = 0.001). This test was repeated with a subset of 10 isolates with similar results (data not shown).
Of the 25 isolates of B. cinerea tested for variation in sensitivity to iprodione, colony diameters ranged from 3.25 to 8.0 cm at 3 DAI on unamended PDA (P = 0.01). Colony diameters on PDA amended with iprodione ranged from 0.46 to 1.93 cm (P = 0.01) (Fig. 3B). The ratio of growth on the fungicide-amended medium to growth on the unamended PDA varied from 0.06 to 0.49 (P = 0.01). It was again noted that the ratio of growth on the amended medium to that on unamended PDA was negatively correlated with colony diameters on the unamended PDA (r = −0.76, P = 0.0001).
No correlation in sensitivity to the different fungicides was detected among the collection of isolates tested.
Discussion
Botrytis cinerea is a common pathogen of many plant species, generally causing foliage and fruit rots and blights, however it is a relatively rare pathogen of peanut. Recent epidemics in peanut are attributed to periods of cooler, wet weather in Texas and to recent use of later maturing cultivars in Georgia (Woodward et al., 2005). In the present study, we documented the general susceptibility of commonly grown peanut cultivars to botrytis blight, but we did observe sufficient variation in the in vitro assays to suggest that low levels of resistance may exist in some peanut genotypes. Additional evaluations of differences among peanut genotypes in field tests are needed to confirm these initial observations.
Although resistance to several classes of fungicides is common in different populations of B. cinerea (Beevers and Brien, 1983; LaMondia and Douglas, 1997; Yourman and Jeffers, 1999), the isolates collected from peanut in west Texas and tested for variation in sensitivity to dicloran, fluazinam, and iprodione were generally sensitive to these fungicides. The significant variation in sensitivity that was observed was of a relatively low order of magnitude, with the lowest levels of sensitivity (based on a higher ratio of growth on fungicide-amended media to the unamended medium) observed in isolates with relatively low growth rates. The absence of any high levels of fungicide insensitivity in the isolates of B. cinerea collected from peanut in west Texas may be due to the relatively limited use of fungicides in the area. Peanut is a relatively recent crop in west Texas, with a nearly 20-fold increase in hecterage planted over the past 20 years. The other major crops in the region are cotton, sorghum, and wheat. Of these crops only wheat is likely to receive a foliar application of fungicides and then only infrequently. There is limited production of grape (ca 400 ha) in the region, along with limited production of vegetables (<1,000 ha) and fungicides are applied several times each season to this crop. This combination of predominant crops that are rarely treated with foliar fungicides coupled with a typically arid climate means that the extant population of B. cinerea has probably not been heavily exposed to fungicides, thus there has been little selection for fungicide insensitivity.
The variation in fungicide sensitivity observed, however, may be sufficient to result in less than desired levels of disease suppression from fungicide application in some fields, but is not likely to cause a major failure of the fungicide to suppress disease. However, given the frequency with which high levels of insensitivity to several classes of fungicides has been observed among populations of B. cinerea, it will be important to monitor the pathogen population for the occurrence of isolates with lower levels of sensitivity.
Literature Cited
Beever R. E. and Brien H. M. R. 1983 A survey of resistance to the dicarboximide fungicides in Botrytis cinerea. N. Z. J. Agric. Res 26 : 391 – 400 .
Hollowell J. E. , Shew B. B. , and Isleib T. G. 2003 Evaluating isolate aggressiveness and host resistance from peanut inoculation with Sclerotinia minor. Plant Dis 87 : 402 – 406 .
LaMondia J. A. and Douglas S. M. 1997 Sensitivity of Botrytis cinerea from Connecticut greenhouses to benzimidazole and dicarboximide fungicides. Plant Dis 81 : 729 – 732 .
Orellana R. G. and Bailey W. K. 1964 Blight of peanut in Maryland and Virginia in 1963. Plant Dis. Rep 48 : 519 – 521 .
Porter D. M. 1997 Botrytis blight. 10 – 11 In Kokalis-Burelle N. , Porter D. M. , Rodríguez-Kábana R. , Smith D. H. , and Subrahmanyan P. eds. Compendium of Peanut Diseases. 2nd ed APS Press St. Paul, MN .
Starr J. L. , Raghvan T. , Henry M. A. , Kenerley C. M. , and Wheeler T. A. 2006 Botrytis blight of peanut — Pathogen fungicide tolerances and peanut cultivar susceptibility. Proc. Amer. Peanut Res. and Edu. Soc. (IN PRESS)
Woodward J. E. , Brenneman T. B. , Kemerait R. C. , and Culbreath A. K. 2005 First report of botrytis blight of peanut caused by Botrytis cinerea in Georgia. Plant Dis 89 : 910 .
Yourman L. F. and Jeffers S. N. 1999 Resistance to benzimidazole and dicarboximide fungicides in greenhouse isolates of Botrytis cinerea. Plant Dis 83 : 569 – 575 .
Notes
- This research was supported in part by grants from the National Peanut Board. [^]
- Department of Plant Pathology and Microbiology, Texas A&M University, College Station, TX 77843-2132. [^]
- Texas Agricultural Experiment Station, Lubbock, TX 79403. [^] *Corresponding author (email: j-starr@tamu.edu).
Author Affiliations