Introduction
Sclerotinia blight of peanut (Arachis hypogaea L.), caused by the soilborne fungus Sclerotinia minor Jagger (Kohn, 1979), is a serious disease in Texas, Oklahoma, Virginia, and North Carolina. The first report of the disease in the United States was in Virginia in 1971 (Porter and Beute, 1974). By 1972, it was found in Oklahoma (Wadsworth, 1979), where it has become widespread and the most costly disease by a soilborne pathogen of peanut.
Sclerotinia minor is a facultative saprophyte that produces black sclerotia (0.5 to 2.0 mm), which serves as overwintering propagules in soil. Infective hyphae emerge during sclerotia germination and initiate primary infections. During humid conditions, cottony, mycelium is visible on infected stems. Once the stem is girdled, the branch wilts and dies. Ultimately, stems and infected pegs decay resulting in a significant number of pods remaining after harvest (Porter and Melouk, 1997).
Management of Sclerotinia blight in infested fields depend on fungicides (Akem et al., 1992; Brenneman et al., 1987; Damicone and Jackson, 1996; Smith et al., 1991a) and resistant genotypes to reduce disease incidence (DI) and yield loss (Coffelt and Porter, 1982; Smith et al., 1991a). Efforts by peanut breeders and plant pathologists in the last 20 years were successful in developing varieties with resistance to Sclerotinia blight (Coffelt et al., 1982; Kirby et al., 1998; Simpson et al., 2000; Smith et al., 1991b).
Breeding for disease resistance is a long-term program, requiring several years of field testing before a cultivar can be released. Limiting the evaluation of germplasm and breeding lines to field plots prolongs the time required to develop resistant cultivars. Greenhouse methods that allow for speedy and reliable evaluation of genotype reaction to Sclerotinia blight would enhance recognition and selection of resistant germplasm. A detached stem technique that uses a mycelial agar plug as inoculum was developed for screening of spanish genotypes and breeding lines (Akem et al., 1992). Intact stem tissue of runner genotypes have also been beneficial to screen germplasm (Goldman et al., 1995). In these studies, mycelial agar plugs overcame low but yet useful levels of resistance. A uniform source of inoculum without an exogenous energy source may provide greater sensitivity in recognition of susceptible and moderately resistant germplasm. This methodology could accelerate the development of resistant cultivars, and improve recognition of useful levels of disease resistance.
Material and Methods
Fungal Isolate
A single isolate of S. minor isolated from peanut cv. Florunner in Stillwater, OK and maintained at 25 ± 2 C on potato dextrose agar (Difco Laboratories, Detroit, MI) containing 100 µg/ml of streptomycin sulfate (Sigma Chemical Co., St. Louis MO) (SPDA) was used throughout this study.
Production of Inocula
Sclerotial inoculum was produced from stem sections (4 to 8-cm) of the susceptible peanut cultivar Okrun. Excised stems were placed in petri plates lined with a moistened Whatman #1 filter paper (Maidstone, England). A 5-mm dia. agar plug with mycelium was taken from a 2-day-old culture grown on SPDA and placed on each stem. Stems were incubated at 25 ±2 C for 14 d when sclerotia began to form; plates were uncovered to allow gradual drying of infected stems. Sclerotia were collected 20 d after inoculation and stored for 3 to 5 wk at 22 C in a desiccator containing anhydrous calcium sulfate. Dry sclerotia were surface disinfected for 3 min in an aqueous solution of 0.25% sodium hypochlorite, soaked in 100% acetone for 4 min, then transferred to a petri dish lined with Whatman #1 filter paper saturated with sterile water. Petri dishes containing sclerotia were incubated for 4 to 5 d in darkness at 25 ±2 C, and sclerotia showing mycelogenic germination were used as inoculum.
The second type of inoculum consisted of dry mycelia, (Melouk and Bowen, 1990; Melouk et al., 1999). The fungus was cultured on 100 ml of potato dextrose broth (Difco Laboratories, Detroit, MI) in 200 ml Erlenmeyer flasks, on a rotary shaker at 100 rpm for 3 d. Mycelial masses were collected by filtration on Whatman #1 filter paper (Maidstone, England), agar plugs were removed, and the mycelial mass was suspended in a 15% aqueous solution of polyethylene glycol (Sigma Chemical Co., St Louis, MO) for 5 min, and collected again by filtration. Mycelial mats were stored at 22 C in a desiccator containing anhydrous calcium sulfate. Small fragments (20 to 25 µg) were used as inoculum and all fragments were viable on SPDA.
The third type of inoculum consisted of perlite granules coated with fresh mycelial fragments of S. minor. The mycelium was cultured in potato dextrose broth as described above. Approximately 80 mg (fresh weight) of mycelium was fragmented in 20 ml distilled water for 1 min using a Mark II Tissuemizer (Telemar, Cincinnati, OH) at 13,500 rpm. Three grams of perlite granules (2 to 3 mm dia.) were stirred with the fragmented mycelial mixture for 1 min. The perlite inoculum was covered and stored at 5 ±2 C for 24 hr. One perlite granule was used per inoculation. Sclerotinia minor was recovered from 100% of the perlite granules on SPDA.
The fourth type of inoculum consisted of 5-mm dia. agar plugs from the periphery of a 2-d-old culture of S. minor grown on SPDA as described above.
Plant Materials
Selection of susceptible cultivar Okrun and moderately resistant genotypes, cv. Southwest runner, TX 901338-2, TX 961738, and TX 961678, were based on previous Sclerotinia blight experiments (Banks et al., 1989; Kirby et al., 1998).
Intact peanut main stems were selected from whole plants grown in 10-cm dia. pots, and detached main stem tissue from five plants grown in a 17.5-cm dia. pot. Plants were grown in a mixture of sand, soil and shredded peat moss (2:1:1; v/v/v) for 56 d and were fertilized bimonthly with 75 ml of a 0.45% ammonium nitrate solution.
Inoculation of Intact Stem Tissue
Eight-week-old whole plants were prepared for inoculation by removing all but 2 to 3 apical leaves along the main stem. Leaves on secondary shoots were kept intact. Inoculum was placed between the intact main stem and stipules at a central node. Inoculated plants were placed in humidity chambers (150 × 60 × 60-cm) framed by polyvinyl chloride pipes (2.5-cm dia.) and covered in clear polyethylene plastic. Temperature averaged of 19± 2 C at night and 26 ±2 C during the day and relative humidity was maintained at 95 to 100%. The chamber allowed adequate light (13.5 µE/s/m2) to sustain healthy plants throughout experiments. Experimental design consisted of a randomized complete block design (RCBD) with two blocks per chamber. Treatments were replicated 8 times/experiment and the experiment was replicated four times.
Inoculation of Detached Stem Tissue
Detached stem tissue was prepared prior to inoculation from 8-wk-old plants as described by Melouk et al. (1992). Shoots from main stem tissue was excised and placed in test tubes and arranged in racks. Detached stems were inoculated and incubated as described above. Experimental design was a RCBD with 3 blocks/chamber. Treatments were replicated 12 times/experiment and the experiment was replicated four times.
Data Collection and Statistical Analysis
Lesion lengths were recorded daily from 3 to 6 days after inoculation (DAI) on intact stem and detached stem. Disease incidence was the percentage of inoculated main stems exhibiting a lesion at the site of infection on the sixth DAI. Chi square analysis was used to separate observed DI compare to expected (100%) DI. All main effects of lesion development were subjected to analysis of variance (ANOVA) and mean separations was made using Least Significant Difference procedure (PROC MIXED, SAS Institute Inc., 1999 version 6.11).
Results
Disease Incidence
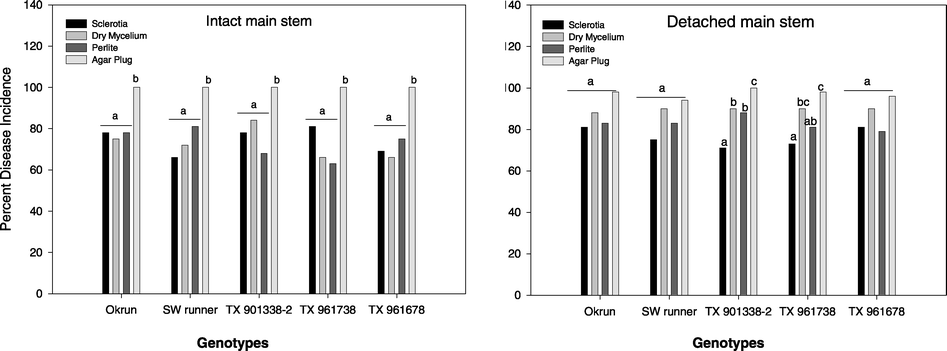
Disease incidence of inocula on intact stem tissue was consistent among genotypes, but there was no observation of a genotype by inoculum interaction. Germinating sclerotia, dry mycelium, and perlite inoculum had similar DI whereas mycelial agar plug had a greater (P < 0.05) DI (Fig. 1). No genotype by inoculum interaction was observed on detached stem tissue. Disease incidence of all inoculum was similar for Okrun, Southwest runner, and TX 961678. However, the mycelial agar plug DI was greater (P < 0.05) on only TX 901338-2 compared to germinating sclerotia, dry mycelium and perlite inoculum (Fig. 1).
Effect of Inoculum on Lesion Development
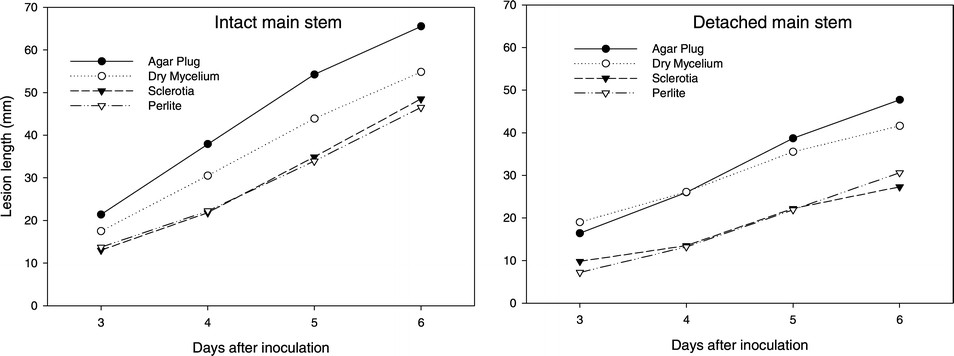
Only those cases that resulted in infection were used to assess lesion development. No inoculum by genotype interaction was observed for intact stem or detached stem tissue. Thus, results are presented across all genotypes (Fig. 2). Lesion development initiated by agar plugs with mycelium on intact stem tissue resulted in a longer (P < 0.05) lesion for each DAI compared to germinating sclerotia, dry mycelium, and perlite inoculum. Further, dry mycelium inoculum produced longer (P < 0.05) lesions for each DAI compared to sclerotia or perlite inoculum. On detached stem tissue, lesion development by mycelial agar plug and dry mycelium resulted in longer lesions for each DAI than sclerotia or perlite inoculum (Fig. 2). All inocula resulted in a faster rate of lesion development on intact stem than detached stem tissue.
Evaluation of Genotype Resistance
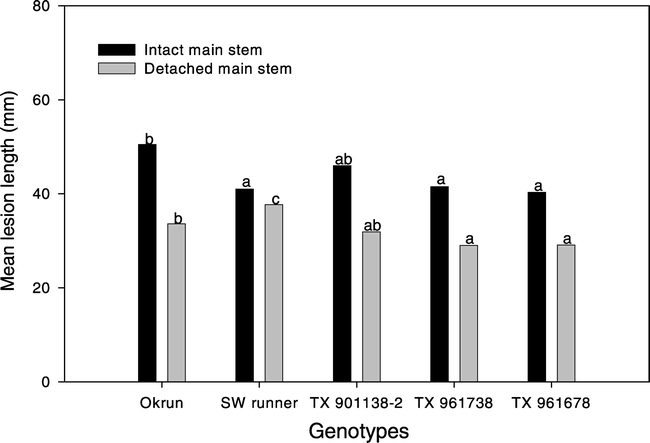
No single inoculum was superior in distinguishing susceptible and moderately resistant peanut genotypes on intact stem or detached stem tissue. All inocula produced similar mean lesion length 6 DAI on intact stem tissue. Thus, results are presented across inocula, which differentiate (P < 0.05) Okrun (susceptible genotype) from Southwest Runner, TX 961738, and TX 961678 (moderately resistant runner genotypes). However, moderately resistant genotype TX 901338-2 was similar in lesion length on intact stem tissue to Okrun (Fig. 3). On detached stem tissue a longer (P < 0.05) lesion length developed on Okrun compared to TX 961738 and TX 961678, but not TX 901338-2. Further, lesion length on detached stem tissue for the moderately resistant control (SW runner) was longer (P < 0.05) susceptible control (Okrun).
Lesion length on intact and detached peanut main stem tissue inoculated with Sclerotinia minor. Only observations in which lesions developed are represented and presented across all genotypes for both intact and detached peanut main stem tissue. Bars with same color tone with different letters are significantly different at α = 0.05 according to Least Significance Difference procedure.
Discussion
In this study, infection was lower for all three inocula without an exogenous nutrient source compared to the mycelial agar plug. Dry mycelium inoculum became active once hydrated resulting in a small flush of hyphae that infected plant tissue. Disease incidence of dry mycelium may be increased by using a larger piece of inoculum. Perlite coated with fresh mycelial fragment inoculum was vulnerable to dehydration, which delayed or inhibited infection. Germinating sclerotia inoculum had variable DI because infectious hyphae would collapse when sclerotia were inoculated on plant tissue. Sclerotia are the primary inoculum and would be an asset as an inoculum in greenhouse screening if 100% germination could be consistently predicted. Thus, sclerotia could be applied to plant prior to germination. Using the germination method described in this study approximantly 30% of sclerotia erupted myceliogenically. Additional hyphae produced from the exogenous nutrient (agar plug) source probably contributed to a higher DI due to multiple infection sites. Agar plug removal after initial infection could limit multiple infection sites, while providing a high DI. Disease incidence using a mycelial agar plug was 100 and 97% on intact stem and detached stem tissue, respectively. The mean DI for germinating sclerotia, dry mycelium, and perlite inoculum across all genotypes was 73 to 83% on intact and detached main stem tissue, respectively. Although, mycelial agar plug can overcome low levels of resistance (Akem et al., 1992; Goldman et al., 1995), in this study it was the most consistent inoculum in developing infection regardless of peanut resistance.
Lesion development from 3 to 6 DAI was greater on intact and detached main stem tissue inoculated with a mycelial agar plug than germinating sclerotia, dry mycelium or perlite inoculum. However, lesions expanded at a similar rate after infection, possibly because they were all from the same isolate of S. minor. Lesion length was significantly different (P < 0.05) 6 DAI among genotypes on intact stem tissue, which identified 75% of the moderately resistant genotypes tested. Intact stem tissue was more reliable in this study and supports the utility of whole plant screening in detecting Sclerotinia blight resistance of runner genotypes (Goldman et al., 1995). Greenhouse screening is difficult to relate to the field since morphological features influence disease development; therefore, morphological features as well as other plant mechanisms (Brenneman et al., 1988; Chappell et al., 1995; Coffelt and Porter, 1982; Goldman et al., 1995) are as important when identifying resistance to S. minor.
Literature Cited
Akem C. N. , Melouk H. A. , and Smith O. D. 1992 Field evaluation of peanut genotypes for resistance to Sclerotinia blight. Crop Prot 11 : 345 – 348 .
Banks B. J. , Kirby J. S. , and Sholar J. R. 1989 Registration of Okrun peanut. Crop Sci 29 : 1574.
Brenneman T. B. , Phipps P. M. , and Stipes R. J. 1987 Control of Sclerotinia blight of peanut: sensitivity and resistance of Sclerotinia minor to vinclozolin, iprodione, dicloran, and PCNB. Plant Dis 71 : 87 – 90 .
Brenneman T. B. , Phipps P. M. , and S. R. J. 1988 A rapid method for evaluating genotype resistance, fungicide activity, and isolate pathogenicity of Sclerotinia minor in peanut. Peanut Sci 15 : 104 – 107 .
Chappell G. F. , Shew B. B. , Ferguson J. M. , and Beute M. K. 1995 Mechanisms of resistance to Sclerotinia minor in selected peanut genotypes. Crop Sci 35 : 692 – 696 .
Coffelt T. A. and Porter D. M. 1982 Screening peanuts for resistance to Sclerotinia blight. Plant Dis 65 : 385 – 387 .
Coffelt T. A. , Porter D. M. , and Mozingo R. W. 1982 Registration of Virginia 81 Bunch peanut. Crop Sci 22 : 1085 – 1086 .
Damicone J. P. and Jackson K. E. 1996 Disease and yield responses to fungicides among peanut cultivars differing in reaction to Sclerotinia blight. Peanut Sci 23 : 81 – 85 .
Goldman J. J. , Smith O. D. , Simpsom C. E. , and Melouk H. A. 1995 Progress in breeding Sclerotinia blight-resistant runner-type peanut. Peanut Sci 22 : 109 – 113 .
Kirby J. S. , Melouk H. A. , Stevens T. E. , Banks D. J. , Sholar J. R. , Damicone J. P. , and Jackson K. E. 1998 Registration of ‘Southwest Runner’ peanut. Crop Sci 38 : 545 – 546 .
Kohn L. M. 1979 A monographic revision of genus Sclerotinia. Mycotaxon 9 : 365 – 444 .
Melouk H. A. and Bowen C. 1990 Preparation of dry, viable mycelia of Sclerotinia minor. Phytopathology 80 : 1015 .
Melouk H. A. , Bowen C. , and Aboshosha S. S. 1999 A procedure for isolation and determination of incidence of Sclerotinia minor in peanut seed. Alexandria J. of Ag. Res 44 : 261 – 270 .
Porter D. M. and Beute M. K. 1974 Sclerotinia blight of peanuts. Phytopathology 64 : 263 – 264 .
Porter D. M. and Melouk H. A. 1997 Sclerotinia blight. 38 – 40 In Kokalis-Burelle N. et al eds. Compendium of peanut diseases American Pathological Society St. Paul, Mn.
Simpson C. E. , Smith O. D. , and Melouk H. A. 2000 Registration of ‘Tamrun 98’ peanut. Crop Sci 40 : 859 .
Smith F. D. , Phipps P. M. , and Stipes R. J. 1991a Agar plate, soil plate, and field evaluation of fluazinam and other fungicides for control of Sclerotinia minor on peanut. Plant Dis 75 : 1138 – 1143 .
Smith O. D. , Simpson C. E. , Grichar W. J. , and Melouk H. A. 1991b Registration of ‘Tamspan 90’ peanut. Crop Sci 31 : 1711 .