Introduction
Tomato spotted wilt, caused by Tomato spotted wilt tospovirus (TSWV) is a serious problem in peanut (Arachis hypogaea L.) producing areas of the southeastern U.S. The use of field resistant cultivars is a critical component of an integrated program for management of spotted wilt in that region. Since 1998, the moderately resistant cultivar Georgia Green has been the predominant cultivar grown in peanut producing areas of Georgia, Alabama, and Florida. The level of field resistance in Georgia Green is not sufficient alone to prevent damage from spotted wilt during severe epidemics, but Georgia Green typically responds very well to cultural practices such as planting date, twin row culture, and higher seeding rates and to phorate insecticide in an integrated system for managing this very damaging disease 3,4.
Developing cultivars with greater levels of field resistance to TSWV is a major objective in peanut breeding programs in the southeastern U.S. Several cultivars have been released and many breeding lines with field resistance similar to or greater than that of Georgia Green have been identified 3,5,6. Breeding line C 11-2-39 from the USDA-ARS breeding program in Tifton, GA has been reported to have much lower final intensity of spotted wilt than moderately resistant cultivars Georgia Green and C-99R and to be among the most resistant lines evaluated in field tests 5. C 11-2-39 has also been reported to be more resistant to TSWV than Georgia Green and C-99R based on mechanical inoculation in growth chamber tests 10. C 11-2-39 is being considered for release as a cultivar. However this line has red testa, which currently is not commercially acceptable to most shellers and manufacturers. C 11-186 is a line selected from a cross of the same parents of C 11-2-39 and is similar in maturity and growth habit to C 11-2-39. C 11-186 has a tan testa. Preliminary evaluations indicate that C 11-186 has field resistance to TSWV similar to that of C 11-2-39. One objective of this study was to compare the spotted wilt intensity and pod yield in C 11-186 to that of Georgia Green and C 11-2-39.
The presumed source of resistance for most of the cultivars and breeding lines developed to date with field resistance to TSWV, including C 11-2-39 and C 11-186, is PI 203396 4,8. There has been a range of responses to TSWV among lines derived from crosses that included PI 203396, as evidenced by differences among Georgia Green, C-99R, and C-11-2-39 in intensity of spotted wilt in the field 5,6 13 and reaction to mechanical inoculation with TSWV 10. Identification of additional sources of resistance would be highly desirable, especially if different mechanisms or different genes are responsible for the resistance.
There has been some screening of the USDA National Peanut Germplasm Collection for resistance to TSWV, and resistance has been found in several accessions of A. hypogaea 1 and other species in the genus Arachis 9. Accessions from both hypogaea and fastigiata subspecies of A. hypogaea have been evaluated 1. However, within subspecies hypogaea, only evaluations of lines from the botanical variety hypogaea have been reported.
In the U.S., work with A. hypogaea subsp. hypogaea var. hirsuta Köhler has been limited. Before 1992, the U.S. National Peanut Germplasm Collection contained only three accessions identified as hirsuta type lines 2. In 1993, Sanchez-Dominguez and Williams reported collecting 12 hirsuta lines from highland areas of Mexico 12, and eighteen hirsuta accessions were added to the National Peanut Germplasm Collection during 1992-1993 2. Initially, interest in those lines was primarily related to flavor, oil content and oil composition 2,11. A few hirsuta lines have been used in crosses with virginia-type genotypes, and preliminary evaluations indicated some of the resultant lines had good field resistance to TSWV.
The breeding line NC 94022 was developed from a cross between N91026E, an early maturing virginia-type line 2, and a tan-seeded component selected from PI 576638. PI 576638 is a hirsuta type line introduced by D. E. Williams and S. Sanchez-Dominguez from the highlands of Mexico 2. N91026E is moderately susceptible to TSWV (T. G. Isleib, unpublished data). F NC94022-1-2-1-1-b3-B resulted from additional selection within an F2-derived family in the F3 and subsequent generations in Florida by D.W. Gorbet. Selections were made from spaced field plantings under heavy pressure from spotted wilt. The objectives of this study also included comparing spotted wilt intensity and pod yield in F NC94022-1-2-1-1-b3-B to those of moderately resistant cultivar Georgia Green and the highly resistant breeding line C 11-2-39.
Materials and Methods
Field tests were conducted at the University of Georgia Coastal Plain Experiment Station, Tifton, GA, and at the University of Florida North Florida Research and Education Center, Marianna, FL in 2003 and 2004. The soil type was a Tifton sandy loam (pH 5.8) for fields at Tifton and a Chipola loamy sand (pH 6.0) for fields at Marianna. Randomized complete block designs with six replications were used in all tests. Tobacco thrips (Frankliniella fusca Hinds) and western flower thrips (F. occidentalis Pergande), known vectors of TSWV that occur in the southeastern U.S., and TSWV occurred naturally at each location. Planting dates were 5 May 2003 and 6 May 2004 at Tifton, and 21 April 2003 and 8 April 2004 at Marianna. Seeding rates were 12.3 seed/m of row for all entries. Plant populations established in both years were lower than the 13 plants/m recommended for commercial peanut production 3. Low plant densities promote higher incidence and severity of spotted wilt 3. Plots consisted of two rows, 6.1 m long and 0.9 m apart except at Marianna in 2004, where plots were 4.6 m long. Each plot was bordered on one side by susceptible SunOleic 97R and was adjacent on one side to another randomly assigned genotype. No insecticides were applied for thrips control.
Genotypes evaluated included cultivars SunOleic 97R and Georgia Green, and three breeding lines, C 11-186, C 11-2-39, and F NC94022-1-2-1-1-b3-B. Based on previous investigations and use in the southeastern region 3,6, Georgia Green and SunOleic 97R represented moderately resistant and susceptible standards, respectively. Spotted wilt intensity was evaluated in each plot using a disease intensity rating that represented a combination of incidence and severity, as described by Culbreath et al. 7. The number of 0.31-m portions of row containing severely stunted, chlorotic, wilted, or dead plants was counted for each plot 91 and 121 days after planting (DAP) in 2003 and 71 and 131 DAP in 2004 at Marianna and 109 and 133 DAP in 2003 and 76 and 109 DAP in 2004 at Tifton. Only the final evaluations are presented in this paper. The number of row portions severely affected was converted to a percentage of total row length for comparison of genotypes. All tests were maintained as recommended for commercial production. Chlorothalonil (Bravo WeatherStik or Bravo Ultrex), tebuconazole (Folicur 3.6 F), azoxystrobin (Abound 2.08F), or pyraclostrobin (Headline 250 EC) were applied as foliar sprays at 7-14 d intervals for control of foliar and/or soilborne fungal diseases.
Plants were dug and inverted at 151 DAP in 2003 and 137 DAP for Georgia Green and SunOleic 97R and 155 DAP for the other entries in 2004 at Marianna, and 144 DAP in 2003 and 148 DAP in 2004 at Tifton. Inverted plants were dried in windrows for 3-7 d. Pods were harvested mechanically, and pod yields were determined for each plot.
All data were subjected to analysis of variance. Data were analyzed across locations and years. Fisher's protected least significant difference (LSD) values were calculated for comparison of genotypes. Differences described in the text are significant at P ≤ 0.05 unless otherwise indicated.
Results
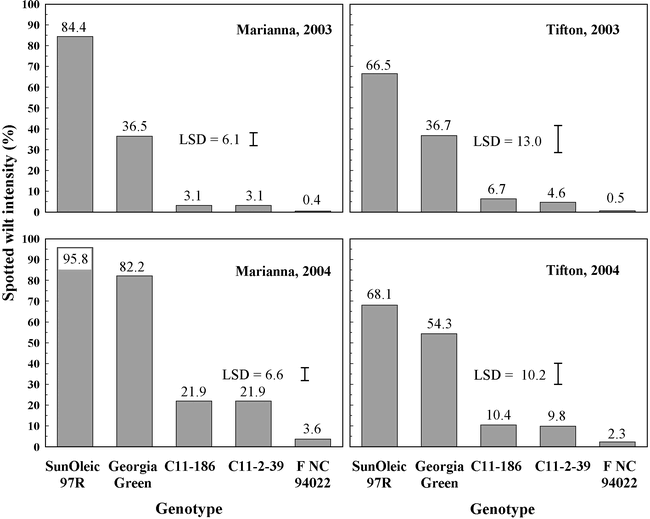
Spotted wilt epidemics were severe in both years and locations and were more severe in 2004 than in 2003 at both locations. There were significant location by genotype interactions for final spotted wilt intensity ratings and yield in both years. Therefore, analyses were conducted within each year and location. In spite of significant interactions, there were similar trends across years and locations. In all tests, final intensity of spotted wilt was highest for SunOleic 97R (Fig. 1). Final intensity of spotted wilt was lower for C 11-2-39, C 11-186, and F NC94022-1-2-1-1-b3-B than for Georgia Green in all tests. Final spotted wilt intensity ratings did not differ among C 11-2-39, C 11-186 and F NC94022-1-2-1-1-b3-B at either location in 2003 or at Tifton in 2004 (Fig. 1). In 2004 at Marianna, final spotted wilt intensity ratings were lower for F NC94022-1-2-1-1-b3-B than for any other genotype.
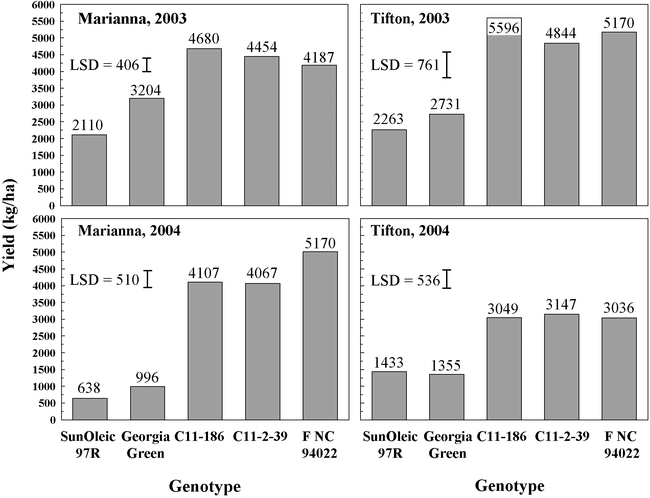
Yields were higher for C 11-2-39, C 11-186 and F NC94022-1-2-1-1-b3-B than for Georgia Green in both years and locations (Fig. 2). At Marianna, yields were higher for C-11-186 than for F NC94022-1-2-1-1-b3-B in 2003, whereas in 2004, yields were higher for F NC94022-1-2-1-1-b3-B than for C-11-186 or C-11-2-39. Yields for these three genotypes were similar in both years at Tifton (Fig. 2).
Discussion
This study corroborates a previous report of lower intensity of spotted wilt in breeding line C-11-2-39 than in Georgia Green 5, and indicates that breeding lines C 11-186 and F NC94022-1-2-1-1-b3-B also have much greater levels of field resistance than Georgia Green. Under extremely heavy epidemic of spotted wilt encountered at Marianna in 2004, intensity ratings in F NC94022-1-2-1-1-b3-B indicate that it is even more resistant than C 11-2-39 or C 11-186, and represents the highest level of field resistance to TSWV among breeding lines of A. hypogaea reported. Based on symptoms observed in the field, F NC94022-1-2-1-1-b3-B is not immune to TSWV. However, F NC94022-1-2-1-1-b3-B had few plants with symptoms of spotted wilt and very few plants with severe symptoms. Experiments are in progress to compare reactions of these lines to mechanical inoculations with TSWV.
This study also indicates that F NC94022-1-2-1-1-b3-B, C 11-186 and C 11-2-39 have excellent yield potential, even when planted at lower than recommended seeding rates in fields in which spotted wilt was severe in Georgia Green and SunOleic 97R standards. Breeding lines C 11-2-39 and C 11-186 are being considered for release as cultivars. They have similar growth habit and pod and seed characteristics except for differences in testa color described earlier.
Previous interest in hirsuta lines in the U.S. has been related to factors other than pathogen resistance, primarily flavor and oil characteristics 2,11. Sanchez-Dominguez and Williams 12 indicated that hirsuta accessions from Mexico may provide breeders with new sources of insect resistance and drought tolerance, as well as improved flavor. Results from this study indicate that the hirsuta line PI 566738, one parent of F NC94022-1-2-1-1-b3-B, may also be a source of resistance to TSWV. These results should prompt the characterization of the reaction of PI 566738 and other hirsuta lines to TSWV.
F NC94022-1-2-1-1-b3-B came from a cross of N91026E with PI 576623 and has several characteristics reminiscent of the hirsuta type. These include tall viny plants, purple stem pigmentation, dark green leaflets, pubescence on stems, and deeply reticulated pods. Plants are spreading to semi-bunch in growth habit, often measuring over 60 cm in height, and much taller than plants of Georgia Green or SunOleic 97R cultivars grown in the same tests. Pod and kernel type in this line would not be desirable in a runner market-type cultivar, and pod and kernel size that would not be large enough for a virginia-type cultivar. Crossing programs using F NC94022-1-2-1-1-b3-B as a parent have been initiated, including crossing with runner-type cultivars and breeding lines with good levels of field resistance to TSWV.
The mechanisms responsible for and genetic control of the field resistance in F NC94022-1-2-1-1-b3-B are not known. However, the presumed source of the resistance is an accession that is taxonomically diverse from all parental lines of current TSWV resistant U.S. cultivars and that was collected from an area geographically isolated from the original source of the parental lines of current cultivars 2,8. Therefore, a different mechanism of resistance, or control of the resistance by different genes, compared to currently available resistant cultivars might be likely.
Regardless of the mechanism, F NC94022-1-2-1-1-b3-B, C-11-2-39, and C-11-186 all have field resistance to TSWV that is much greater than that of the standard moderately resistant cultivar Georgia Green. F NC94022-1-2-1-1-b3-B showed an even greater level of field resistance to TSWV than C 11-2-39 and C 11-186 when spotted wilt epidemics were severe enough that intensity ratings of those two lines exceeded 20%. Additional studies are needed to determine the mechanism and heritability of the factors responsible for the resistance in all of those lines and to characterize the level of field resistance in PI 576638. F NC94022-1-2-1-1-b3-B is one of several lines developed from crosses made using hirsuta type lines. These results should also prompt additional screening for resistance to TSWV among other hirsuta lines.
Acknowledgements
This research was supported, in part, by Georgia and Florida peanut growers through grants from the Georgia Agricultural Commodity Commission for Peanuts, the Florida Peanut Producers' Association, and the National Peanut Board. The essential efforts of Stephanie Carter, Russ Griffin, Mike Heath, Walker Jones and Simmy McKeown are gratefully acknowledged.
Literature Cited
Anderson W. F. , Holbrook C. C. , and Culbreath A. K. 1996 Screening the peanut core collection for resistance to Tomato spotted wilt virus. Peanut Sci 23 : 57 – 61 .
Barrientos-Priego L. , Isleib T. G. , and Pattee H. E. 2002 Variation in oil content among Mexican and Peruvian hirsuta peanut landraces and virginia-type hypogaea lines. Peanut Sci 29 : 72 – 77 .
Brown S. , Todd J. , Culbreath A. , Baldwin J. , Beasley J. , Kemerait B. , Prostko E. , and Smith N. 2004 Minimizing spotted wilt of peanut. Univ. Georgia Coop. Ext. Serv. Bull No. 1165.
Culbreath A. K. , Todd J. W. , and Brown S. L. 2003 Epidemiology and management of tomato spotted wilt in peanut. Ann. Rev. Phytopathol 41 : 53 – 75 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Brown S. L. , Baldwin J. A. , Pappu H. R. , Holbrook C. C. , and Shokes F. M. 1999 Response of early, medium, and late maturing peanut breeding lines to field epidemics of tomato spotted wilt. Peanut Sci 26 : 100 – 106 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Brown S. L. , Baldwin J. A. , Pappu H. R. , and Shokes F. M. 2000 Reaction of peanut cultivars to spotted wilt. Peanut Sci 27 : 35 – 39 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Shokes F. M. , and Pappu H. R. 1997 Field response of new peanut cultivar UF 91108 to Tomato spotted wilt virus. Plant Dis 81 : 1410 – 1415 .
Isleib T. G. , Holbrook C. C. , and Gorbet D. W. 2001 Use of plant introductions in peanut cultivar development. Peanut Sci 28 : 96 – 113 .
Lyerly J. H. , Stalker H. T. , Moyer J. W. , and Hoffman K. 2002 Evaluation of Arachis species for resistance to Tomato spotted wilt virus. Peanut Sci 29 : 79 – 84 .
Mandal B. , Pappu H. R. , Culbreath A. K. , Holbrook C. C. , Gorbet D. W. , and Todd J. W. 2002 Differential response of selected peanut (Arachis hypogaea) genotypes to mechanical inoculation by Tomato spotted wilt virus. Plant Dis 86 : 939 – 944 .
Pattee H. E. , Giesbrecht F. G. , Sanchez-Dominguez S. , and Williams D. E. 1995 Evaluation of six landrace accessions of Arachis hypogaea ssp. hypogaea var. hirsuta Köhler. I. Descriptive and sensory. Peanut Sci 22 : 18 – 22 .
Sanchez-Dominguez S. and Williams D. E. 1993 Results of a recent plant exploration in Mexico to collect the hirsuta peanut. Proc. Amer. Peanut Res. Ed. Soc 25 : 35 (abstr.) .
Wells M. L. , Culbreath A. K. , Todd J. W. , Brown S. L. , and Gorbet D. W. 2002 A regression approach for comparing field resistance of peanut cultivars to Tomato spotted wilt tospovirus. Crop Prot 21 : 467 – 474 .
Notes
- 1Dept. of Plant Pathology, Univ. of Georgia, Coastal Plain Exp. Sta. Tifton, GA 31793-0748 [^]
- 2University of Florida, N. Florida Res. and Educ. Cntr., Marianna, FL 32446-7906 [^]
- 3USDA-ARS, Crop Genetics and Breeding, Coastal Plain Exp. Sta. Tifton, GA 31793-0748 [^]
- 4Dept. of Entomology, Univ. of Georgia, Coastal Plain Exp. Sta. Tifton, GA 31793-0748 [^]
- 5Dept. of Crop Science, North Carolina State Univ., Raleigh, NC 27695-7629 [^] *Corresponding author (email: spotwilt@tifton.uga.edu).