Introduction
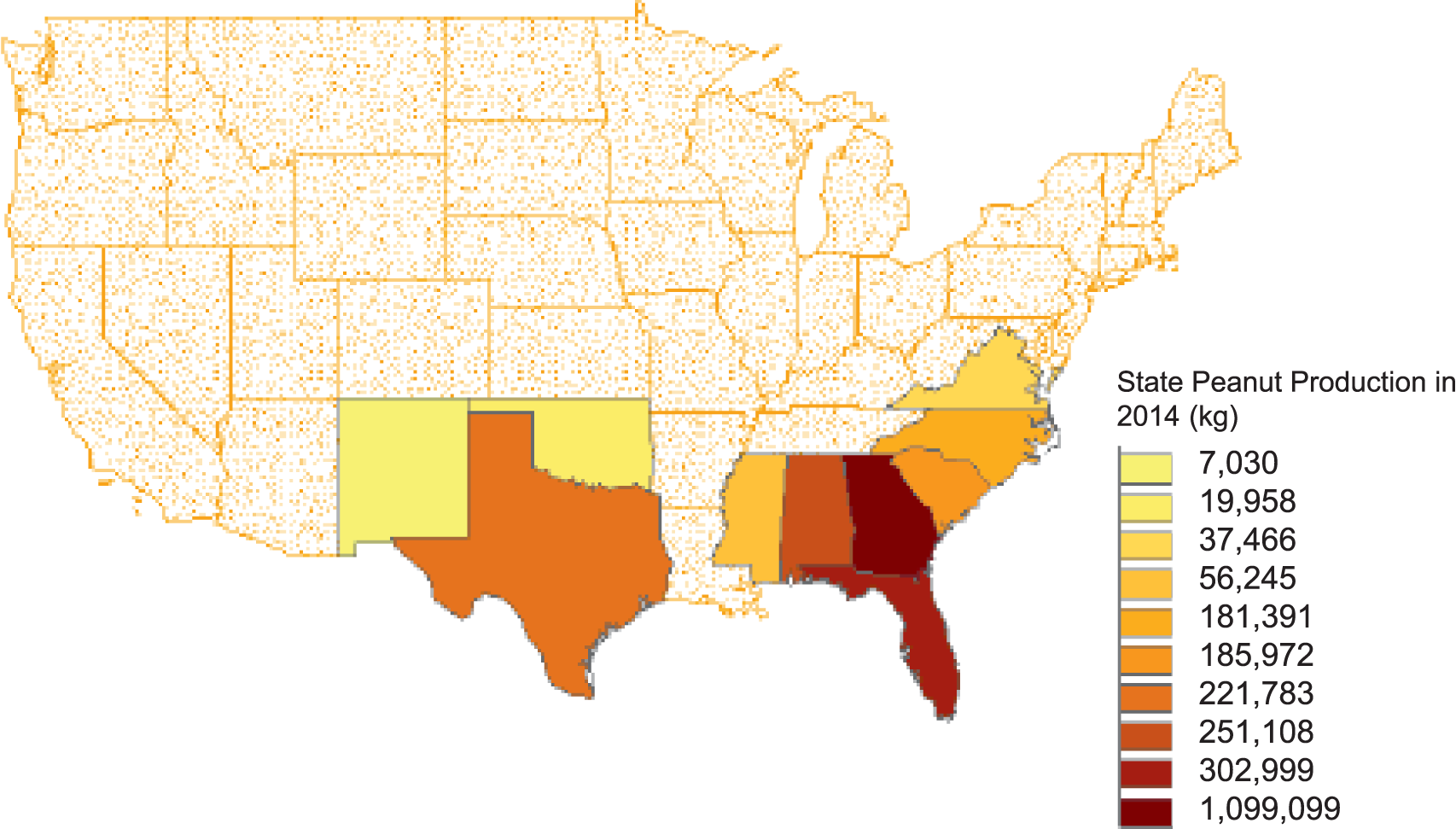
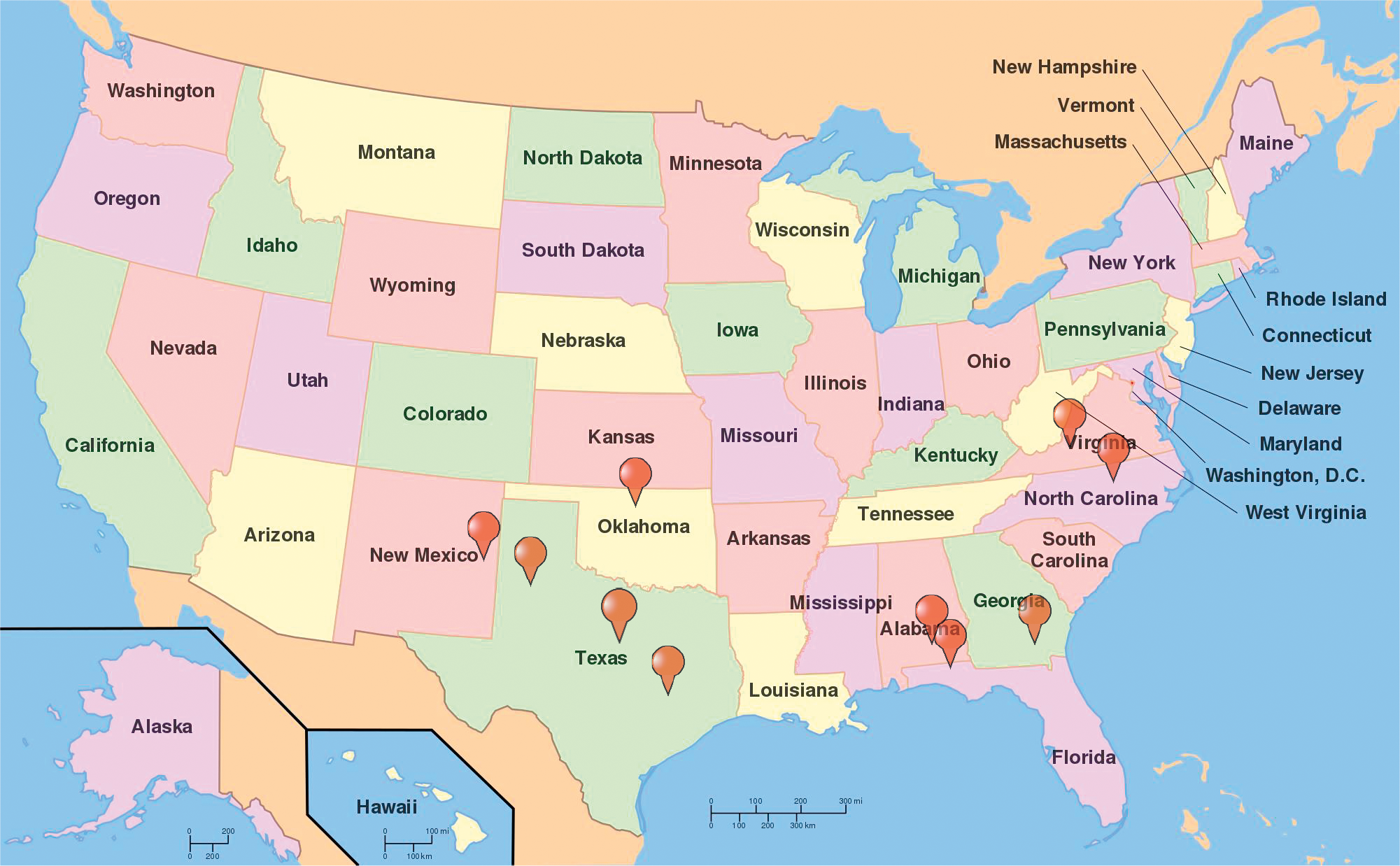
The U.S. currently ranks 3rd in the world in peanut production behind China and India and produces 10% of the world's crop. Production in the US has risen overall in the last 50 years to a high of 3,200 kg and valued at $1.6 billion reported in 2017 (NASS, 2017). Most of peanut production in the United States has traditionally been located 3 geographic regions: Southeast (Alabama, Florida, Georgia), Southwest (New Mexico, Oklahoma, Texas), and the Virginia-Carolinas (North Carolina, Virginia). Within the last decade, production has also been reported in Arkansas, Mississippi, and South Carolina with the top 10 peanut producing states shown in Figure 1. Because the three peanut production regions vastly differ in aspects biotic and abiotic stressors, peanuts developed in a specific region generally do not perform well in other regions. Therefore, public peanut breeding programs are located strategically within each growing region (Figure 2). Most likely areas of peanut production in the US will remain geographically stable unless shifted by a catastrophic weather event or significant change in the agricultural economic arena.
Public peanut breeding programs in the United States. (Auburn University, Auburn, AL; University of Georgia, Tifton, GA; University of Florida, Marianna, FL; New Mexico State University, Clovis, NM; North Carolina State University, Raleigh, NC; Texas AgriLife Research: Lubbock, TX, College Station, TX, Stephenville, TX; USDA-ARS locations: Stillwater, Oklahoma, Tifton, Georgia; Virginia Tech: Suffolk, VA.
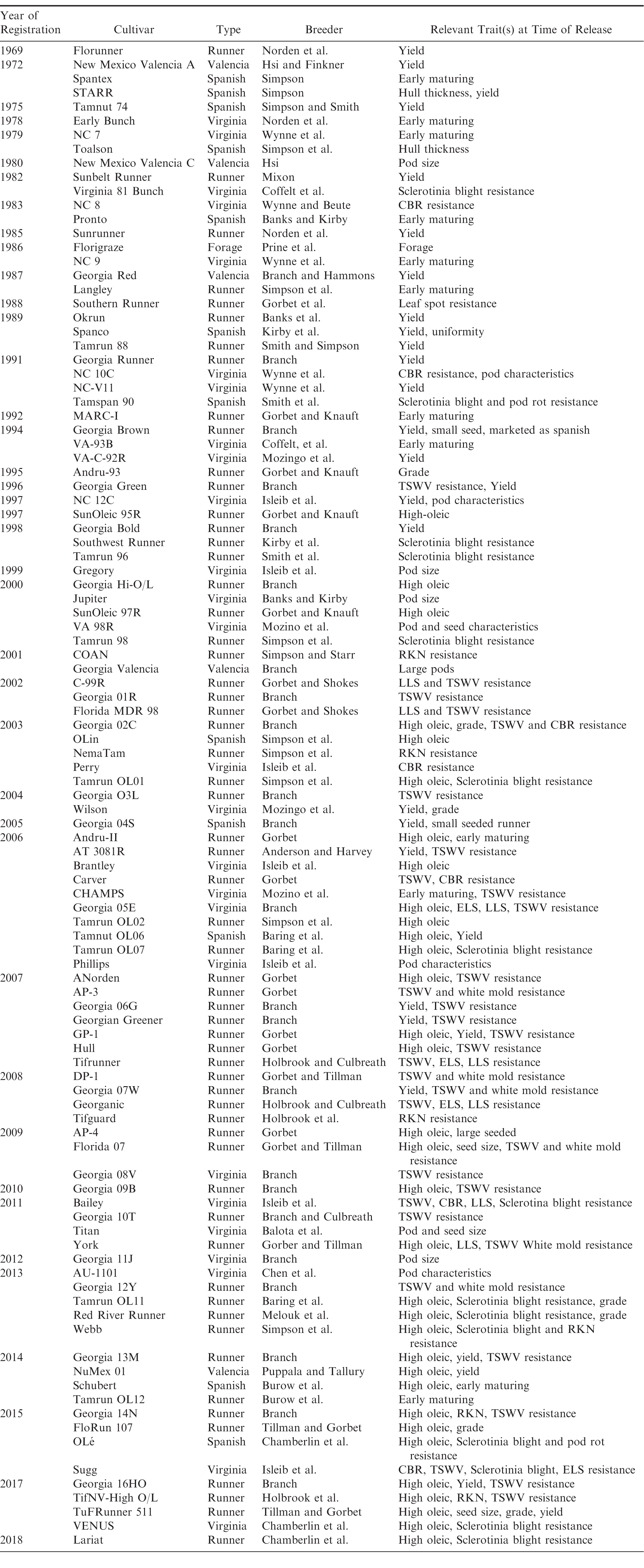
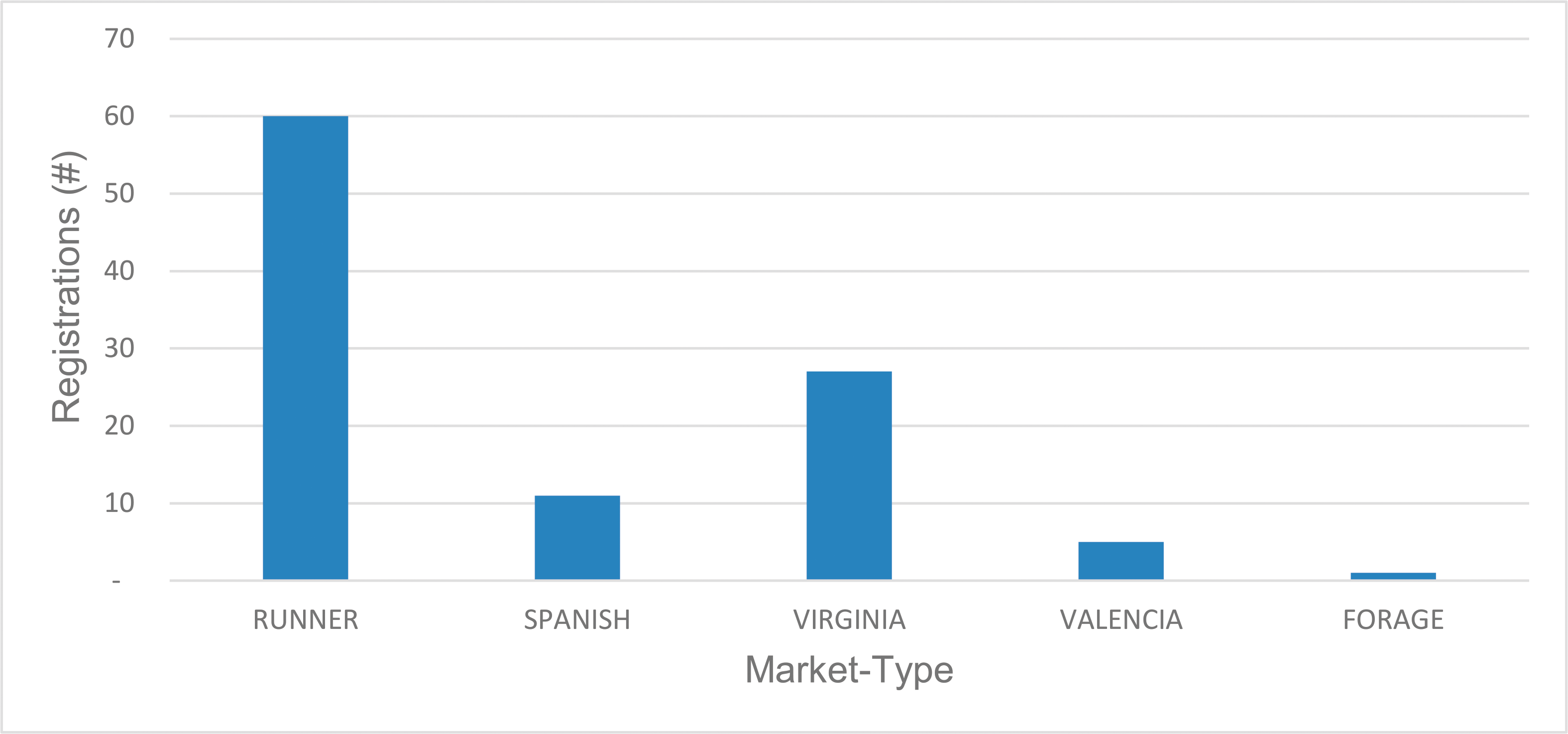
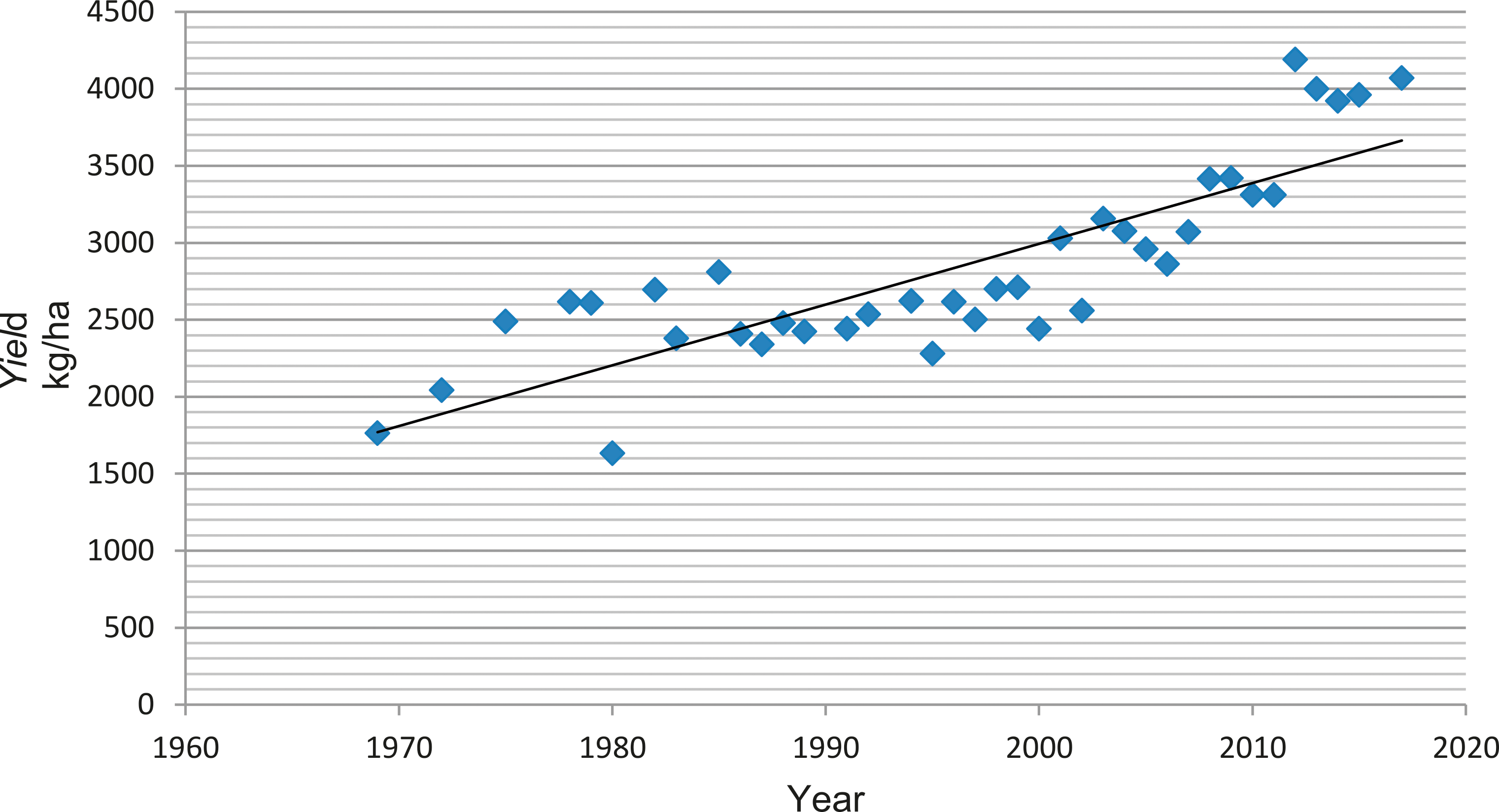
Public peanut breeding programs have been extremely successful in cultivar development, registering over 100 variety releases since 1969 (Table 1). Since the release of Florunner (Norden et al, 1969) there have been 59 runner-type, 27 virginia-type, 11 spanish-type, 5 valencia-type, and 1 forage-type cultivar releases registered in the U.S. The number of variety releases by peanut market-type is reflected by U.S. peanut production in proportion (Figure 3), underscoring the intimate connection between breeders and producers. Over the last 50 years, peanut yields have more than doubled, increasing from 1,904 kgha-1 in 1969 to over 4,480 kgha-1 in 2017 (Figure 4). Factors contributing to this increase include precision farming equipment, improved chemicals and advisories for weed and pest control, improved field inoculants and crop rotation practices. Improved peanut cultivars available for commercial production have also contributed to increased yield, disease resistance, oil quality, drought resistance and maturity. In several cases, the release of a disease resistant cultivar has prevented the collapse of the peanut industry in a growing region. For example, the release of Georgia Green in the mid-1990s (Branch, 1996) was largely responsible for saving production in the Southeastern U.S. due to its resistance to the Tospovirus described as Tomato Spotted Wilt (Culbreath et al., 1992), a pathogen that still threatens the region today. Timely released cultivars have also shielded Southeastern producers from yield losses due to early and late leaf spot (Cercospora arachidicola and Cercosporidium personatum, respectively) as well as root-knot nematodes (Meloidogyne spp.) and Cylindrocladium black rot (Cylindrocladium parasiticum). Sclerotinia blight on peanut (Sclerotinia minor Jagger) nearly devastated peanut production in the Southwestern U.S., but the release of Tamspan 90 (Smith et al., 1991) and Tamrun 96 (Smith et al., 1998) allowed producers to overcome up to 50% yield losses caused by that disease. The development of disease resistant or otherwise improved peanut cultivars is a never-ending quest because of constantly changing biotic and abiotic stressors. Therefore, peanut breeders face the endless task of continually developing new varieties. The search continues for new and better sources of disease resistance and other value-added traits by phenotyping vast germplasm collections in lengthy and labor-intensive field trials. Incorporation of new beneficial traits into cultivated peanut using traditional breeding methods takes 10-12 years after discovery.
According to a report by the United Nations Department of Economic and Social Affairs, the world population has been predicted to reach 10 billion by 2050, and at current production rates, the world food supply is barely keeping up with demand. What does this mean to future generations of humanity? Although the amount of land available to agriculture in the U.S. has remained constant in the last 50 years, the percentage of the American workforce in agriculture has drastically declined. Fewer generations are choosing to remain on their family's farm, and instead chose to pursue other employment options. The consequence of these actions is that fewer farmers must produce more products. To produce the amount of food needed to sustain the world's growing population, much of the additional food will need to come from increases in crop yield rather than increased cropping intensity or land cultivation.
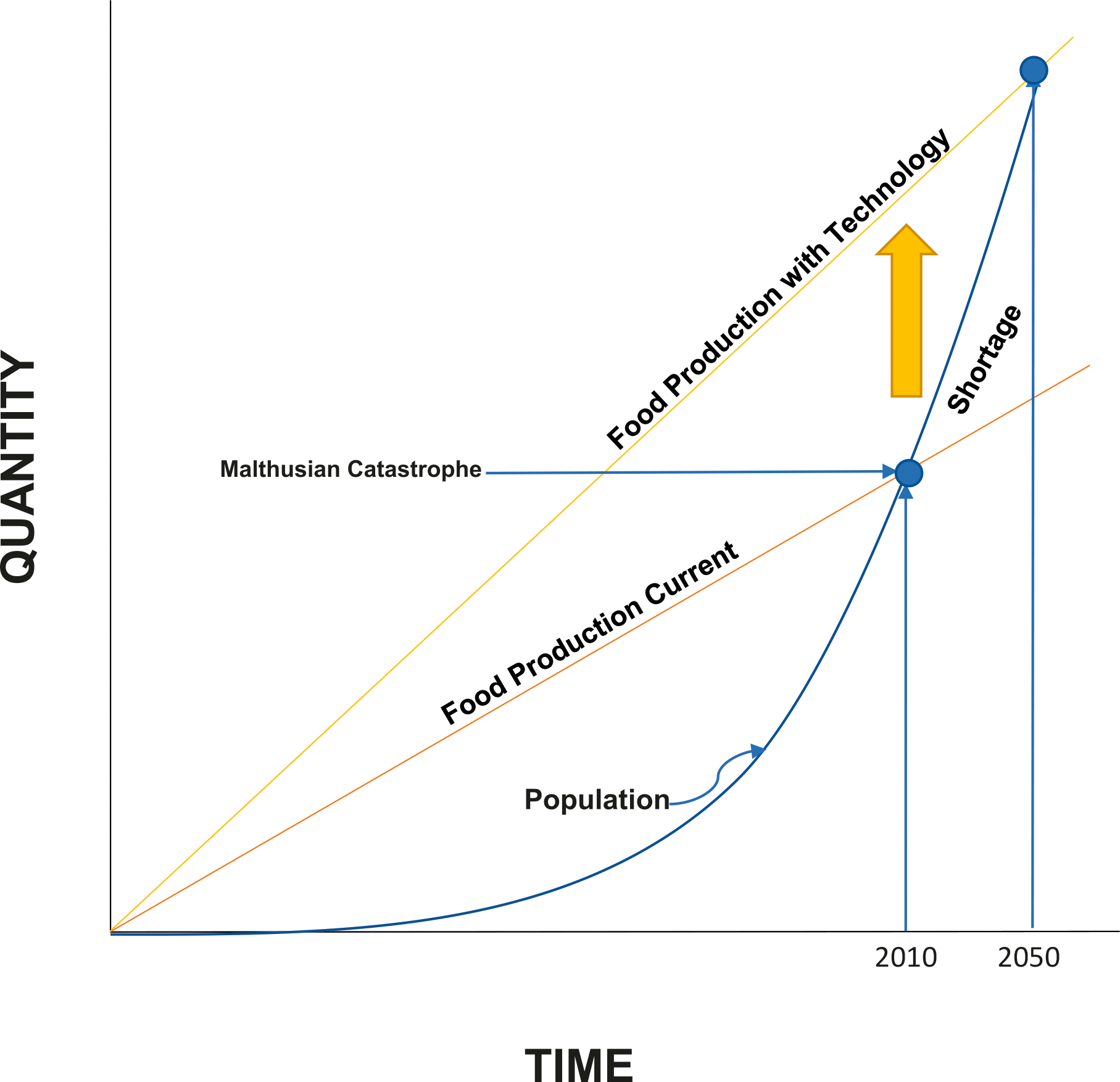
America's peanut farmers help feed the world. Peanuts and peanut products are an important source of protein in countries across the globe, especially those which are underdeveloped, and could contribute greatly to the prevention of human starvation in the future. However, peanut production (along with other food crops) will need to double over the next 30 years to provide ample food for the growing population. Is it possible to double peanut yields in the next 30 years? Researchers predict that a Malthusian Catastrophe (Figure 5) can be avoided in the future by using precision agriculture technology along with molecular breeding to boost farmer's production.
Current food production vs. future food production in relation to growing world population over time. Orange line predicts boost in production if technology such as precision agriculture and molecular breeding are used (Urbano, 2011).
Molecular breeding is described as the application of molecular tools in traditional breeding programs. One example of molecular breeding is marker-assisted selection (MAS) where molecular markers closely associated with a trait of interest are used to select for breeding material and/or advanced breeding lines during early stages of development. This is different from genetic engineering where molecular tools are used to artificially insert beneficial genes into a target genome. MAS, when used in conjunction with traditional breeding methods, increases the efficiency of the development of new cultivars by enabling the breeder to select breeding material containing the trait of interest without having to spend years phenotyping for the trait in the field (Xu, 2010).
Implementation of molecular tools such as MAS in a plant breeding program can greatly increase the efficiency of cultivar development. Early generation selection using MAS allows the breeder to discard many plants with unwanted gene combinations, especially those that lack the essential trait(s) of interest, without years of phenotyping in the field. MAS allows breeders to move fewer breeding lines forward for testing and increases the probability that advanced breeding lines will contain desired traits. These tools also allow foreground and background selection during breeding and backcross population development as well as rapid pyramiding of desired traits within the same cultivar. When used, molecular tools greatly enhance the efficiency and accuracy of breeding program.
MAS has been used successfully in breeding for many other field crops including, but not limited to, Oryza sativa (rice) (Collard et al., 2008), Glycine max (soybean) (Gavioli, 2011), and Triticum aestivum (wheat) (Arruda et al., 2016). Peanut breeders have been unable to employ MAS in their programs due to a lack of genetic resources required. Pre-requisites to the application of molecular breeding and MAS include a reliable genome sequence, numerous molecular markers on a high-density genetic map, and reliable trait-associated markers. No sequence information for the peanut genome was available until 2016 when the sequences of the diploid ancestors of cultivated peanut were reported by Bertioli et al.et al. The sequence of cultivated peanut has recently been determined and is forthcoming. Genetic maps with markers have been generated for peanut and are available for use on PeanutBase (Sudhansu et al., 2016), but few reliable trait-associated markers for peanut have been identified. Agronomic traits for which markers have been reported in peanut include high oleic acid content (Chu et al., 2009; Barkley et al., 2010, 2011). Few molecular markers have been identified in peanut for disease resistance, although markers have been reported for resistance to nematodes (Garcia et al., 1996; Chu et al., 2007,2016), tomato spotted wilt virus (Liu et al., 2015), leaf spot (Varma et al., 2005; Mace et al., 2006; Mondal and Badigannavar, 2010; Shoba et al., 2012; Shirasawa et al., 2013; Liu et al., 2015), rust (Varma et al., 2005; Mace et al., 2006; Mondal and Badigannavar, 2010; Shoba et al., 2012; Shirasawa et al., 2013).
Determination of the peanut genomic sequence was only made possible by significant financial support of the peanut industry for the International Peanut Genome Initiative (PGI), a group of scientists from the U.S., China, Brazil, India and Israel whose objectives are to delineate peanut genome sequences, characterize the genetic and phenotypic variation in cultivated and wild peanuts and develop genomic tools for peanut breeding. These investments in research made by the peanut industry moved peanut breeding closer to molecular application. An overwhelming amount of genetic information has now been generated and thousands of molecular markers within the genome have been identified and mapped. A bottleneck now exists between available information and application, and the focus of the PGI must now shift to the identification and implementation of trait-associated molecular markers. Phenotyping of the recombinant inbred line (RIL) populations already developed by the PGI and correlation of that data with genetic data already gathered will provide the information needed to define reliable trait-associated markers and implement the use of those markers in peanut breeding programs throughout the U.S. growing regions.
Once these markers are deployed for use, the breeder must be able to implement their use. Concepts in genomic selection and or marker assisted breeding may not be understood by today's traditional plant breeder, but these techniques are commonly part of the curriculum required for current students of plant breeding. Highly specialized equipment for high-throughput analysis is not available to most plant breeders today, therefore steps must be taken to either provide services and/or equipment for MAS. This action may fall on the shoulders of the peanut industry since breeding programs are normally not well funded by grants, making it difficult to fund MAS. Molecular testing is costly making high-throughput screening difficult for the average peanut breeding program. However, steps are currently underway to make it more affordable for the peanut breeding community.
While the implementation of molecular techniques in breeding programs will increase efficiency and accuracy, they will not replace the breeder's expert eye. Communication with producers and the traditional skills of crossing and selection in the field will remain vital to the successful peanut breeder. However, molecular tools used in concert with traditional techniques will expand the future peanut breeder's toolkit. Peanut breeding-by-design may be the key to achieving increased the cultivar quality and yield necessary to keep pace with expanding world demand.
Acknowledgements
The author would like to thank the American Peanut Research and Education Society for the invitation to write this review article. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Literature Cited
M.P., Arruda, A.E., Lipka, P.J., Brown, A.M., Krill, C., Thurber, G., Brown-Guedira, B.J., Foresman, and F.L Kolb, (2016). Comparing genomic selection and marker-assisted selection for Fusarium head blight resistance in wheat (Triticum aestivum L.). Mol. Breeding 36: 84.
N.A., Barkley, K.D.C., Chamberlin, M.L., Wang, and R.N Pittman, (2010). Development of real-time PCR genotyping assay to identify high oleic acid (18:1) peanuts (Arachis hypogaea L.). Mol. Breed 25: 541- 548.
N.A., Barkley, K.D.C., Chamberlin, M.S., Wang, and R.N Pittman, (2011). Genotyping and fatty acid composition analysis in segregating peanut (Arachis hypogaea L.) populations. Peanut Sci 38: 11- 19.
Bertioli, D.J., Cannon, S.B., Froenicke, L., Huang, G., Farmer, A.D., Cannon, E.K., Liu, X., Gao, D., Clevenger, J., Dash, S., Ren, L., Moretzsohn, M.C., Shirasawa, K., Huang, W., Vidigal, B., Abernathy, B., Chu, Y., Niederhuth, C.E., Umale, P., Araújo, A.C., Kozik, A., DoKim, K., Burow, M.D., Varshney, R.K., Wang, X., Zhang, X., Barkley, N., Guimarães, P.M., Isobe, S., Guo, B., Liao, B., Stalker, H.T., Schmitz, R.J., Scheffler, B.E., Leal-Bertioli, S.C., Xun, X., Jackson, S.A., Michelmore, R., Ozias-Akins, P. 2016 The genome sequences of Arachis duranensis and Arachis ipaensis, the diploid ancestors of cultivated peanut Nature Genetics 48: 438- 446 doi: 10.1038/ng.3517. PubMed PMID: 26901068.
W.D Branch, (1996). Registration of 'Georgia Green' peanut. Crop Sci 36: 806.
Y., Chu, R., Gill, J., Clevenger, P., Timper, C.C., Holbrook, and P Ozais-Akins, (2016). Identification of rare recombinants leads to tightly linked markers for nematode resistance in peanut. Peanut Sci 43: 88- 93.
Y., Chu, C.C., Holbrook, and P Ozais-Akins, (2009). Two alleles of ahFAD2Bcontrol the high oleic acid trait in cultivated peanut. Crop Sci 49: 2029- 2036.
Y., Chu, C.C., Holbrook, P., Timper, and P Ozais-Akins, (2007). Development of a PCR-based molecular marker to select for nematode resistance in peanut. Crop Sci 47: 841- 847.
Collard, B.C.Y, Vera Cruz, C.M., McNally, K.L., Virk, P.S., and Mackill, D.J 2008 Rice molecular breeding laboratories in the genomics era: Current status and future considerations Int. J. Plant Genomics DOI: 10.1155/2008/524847.
A.K., Culbreath, J.W Todd, and J.W Demski (1992). Productivity of Florunner peanut infected with tomato spotted wilt virus. Peanut Sci 19: 11- 14.
Gavioli, E.A 2011 Molecular Markers: Assisted Selection in Soybeans, Soybean - Genetics and Novel Techniques for Yield Enhancement, Prof Dora Krezhova (Ed.), ISBN: 978-953-07721-5, InTech, Available from: http://www.intechopen.com/books/soybean-genetics-and novel-techniques-for-yieldenhancement/molecular-markers-assisted-selection-in-soybeans
G.M., Garcia, H.T., Stalker, E., Shroeder, and G Kochert, (1996). Identification of RAPD, SCAR, and RFLP markers tightly linked to nematode resistance genes introgressed from Arachis cardenasiiinto Arachis hypogaea. Genome 39: 836- 845.
L., Liu, P.M., Dang, and Chen. C.Y. (2015). Development and utilization of InDel markers to identify peanut (Arachis hypogaea) disease resistance. Front. Plant Sci 6:988.Mace, E.S.,. D.T., Phong, H.D., Upadhyaya, S., Chandra, and J.H Crouch, SSR analysis of cultivated groundnut (Arachis hypogaea L.) germplasm resistant to rust and late leaf spot diseases. Euphytica 152: 317- 330.
S., Mondal, and A.M Badigannavar, (2010). Molecular diversity and association of SSR markers to rust and late leaf spot resistance in cultivated groundnut (Arachis hypogaea L.). Plant Breed 129: 68- 71.
A.J., Norden, R.W Lipscomb, and W.A Carver, (1969). Registration of 'Florunner' peanuts. Crop Sci 9: 850.
K., Shirasawa, D.J., Bertioli, R.K., Varshney, M.C., Moretzsohn, S.C.M., Leal-Bertioli, M., Thudi, M.K., Pandey, J.F., Rami, D., Foncéka, M.V.C., Gowda, H., Qin, B., Guo, Y, Hong, X., Liang, H., Hirakawa, S., Tabata, and S Isobe, (2013). Integrated consensus map of cultivated peanut and wild relatives reveals structures of the A and B genomes of Arachis and divergence of the legume genomes. DNA Res 20: 173- 184.
D., Shoba, N., Manivannan, P., Vindhiyavarman, and S.N Nigam, (2012). SSR markers associated for late leaf spot disease resistance by bulked segregant analysis in groundnut (Arachis hypogaea L.). Euphytica 188: 265- 272.
O.D., Smith, C.E., Simpson, W.J., Grichar, and H.A Melouk, (1991). Registration of 'Tamspan 90' peanut. Crop Sci 31: 1711.
O.D., Smith, C.E., Simpson, M.C Black, and B.A Besler, (1998). Registration of 'Tamrun 96' peanut. Crop Sci 38: 1403.
D., Sudhansu, E.K.S., Cannon, S.R., Kalberer, A.D., Farmer, and S.B Cannon, (2016). PeanutBase and Other Bioinformatic Resources for Peanut. In. Peanuts Genetics, Processing, and Utilization, . H.T Stalker and R.F Wilson (eds)., AOCS Press, , pp. pp. 241- 252.
United Nations Department of Economic and Social Affairs. http://www.un.org/en/development/desa/news/population/2015-report.html
Urbano, L., 2011 Malthusian Growth, Retrieved November 26th, 2018, from Montessori Muddle: http://MontessoriMuddle.org/
USDA National Agricultural Statistics Service (NASS). https://www.nass.usda.gov
T.S.N., Varma, S.L., Swivedi, S., Pande, and M.V.C Gowda, (2005). SSR markers associated with resistance to rust (Puccinia arachidis Speg.) in groundnut (Arachis hypogaea L.). SABRAO J Breed. Genet 37: 107- 119.
Y Xu, (2010). In. Molecular Plant Breeding, Marson Book Services, Ltd, Didcot, Oxon, UK., pp. 18- 20.
Notes
- Research Biologist, USDA ARS Wheat, Peanut and Other Field Crops Research Unit, 1301 N. Western Rd., Stillwater, Oklahoma 74075 [^] *Corresponding author Email: Kelly.Chamberlin@ARS.USDA.GOV@uga.edu
Author Affiliations