Introduction
In 2017, Georgia producers harvested 333,865 hectares of peanut (Arachis hypogaea L.). This ranks Georgia as the leading producer of peanuts in the US (USDA-NASS, 2018). Currently, runner-type peanut cultivars are the most commonly grown in Georgia (Monfort, 2017). Peanut is characterized by having prostrate growth and indeterminate flowering. This growth habit limits peanut's ability to compete with weeds on its own (Buchanan et al., 1982). There is large variation in peanut yield loss due to weed interference. There are many factors, such as herbicide use, irrigation, and tillage practices that can influence weed populations. Because of this, producers must make sound decisions about weed control during the entire growing season (Hill and Santelmann, 1969; Hauser et al., 1975).
Dinoseb (2-sec-butyl-4,6-dinitrophenol) was heavily used in POST applications until its suspension. The loss of dinoseb in 1986 caused producers in the Southeastern US to rely heavily on paraquat (1,1'-dimethyl-4,4'-bipyridinium) as their staple POST herbicide in peanut (Wilcut et al., 1989). Paraquat is used to control broadleaf and grass weed species due to its nonselective nature (Wehtje et al., 1986). Paraquat must be applied no later than 28 d after emergence (DAE) in order to avoid significant foliar damage and yield loss to peanut (Wilcut and Swann, 1990). Peanut treated with paraquat past the 28 DAE mark may be injured with less time for recovery (Johnson et al., 1993). When paraquat is applied at the correct growth stage, foliar damage does not correlate with peanut yield loss (Wehtje et al., 1992; Wilcut et al., 1989).
Additionally, paraquat is tank-mixed with herbicides of different mechanisms of action to broaden the weed control spectrum, offset injury caused by paraquat, and provide longer weed control with residual herbicides (Wilcut et al., 1995). Producers tank-mix bentazon (3-(1-methylethyl)-1H-2,1,3-benzothiadiazin-4(3H)-one 2,2-dioxide) with paraquat to reduce injury and increase the flexibility of the application window (Wehtje et al., 1992). However, little information is available on the effects of these paraquat tank-mixtures on current runner-type peanut cultivars, which have varying growth characteristics, yield potential, and disease susceptibility. The main objectives for this research were to determine and quantify the level of injury and effects on vegetation, yield, and grade from POST herbicide tank-mixtures that include paraquat on runner-type peanut cultivars.
Materials and Methods
Peanut experiments conducted during 2016 and 2017 growing seasons were at the University of Georgia (UGA) Ponder Research Farm in Ty Ty (31.510624, -83.646659) and the Southwest Research and Education Center in Plains (32.046365, -84.366465). Trials in Ty Ty were conducted on a Tifton loamy sand (fine-loamy, Kaolinitic, thermic Plinthic Kandiudult) and trials in Plains were conducted on a Greenville sandy loam (fine, Kaolinitic, thermic Rhodic Kandiudult) (USDA-NRCS, 2018). The soils at these locations represent the southeastern US peanut production area. Two separate experiments were conducted at each location, one managed with supplemental irrigation and one entirely rainfed. Irrigation applied on an as-needed basis, in compliance with the UGA Peanut Production Guide Checkbook method (Porter, 2017; Stansell and Pallas, 1985; Stansell et al., 1976).
All trial sites were prepared by disk harrowing, moldboard plowing (30 cm deep) followed by rotary tilling to form 1.8 m wide soil beds. In Plains, plot length for 2016 was 12.2 m while in 2017 plot length was 9.1 m. In Ty Ty, plot length for 2016 was 10.7 m, 2017 irrigated plot length was 10.7 m, and 2017 non-irrigated plot length was 7.6 m. Plot length was determined by field dimensions for the given year and location. Fertilizer applications were based on UGA Extension recommendations and a pre-plant soil sample (Harris, 2018). Protective fungicide programs based on the high-risk management program from the Peanut Rx were followed (Kemerait et al., 2017). Fungicides applied to peanut at the R1 growth stage (Boote, 1982), and continued on 14-d intervals. Peanuts were planted on 11 May 2016 and 30 May 2017 in Ty Ty and 16 May 2016 and 2 May 2017 in Plains using a two-row Monosem air planter to a 5 cm depth at 19 seeds/m of row (Monosem-Inc., Edwardsville, KS). Seeding rate and depth remained constant across all site-year locations.
Trials were a split-plot design arranged in a randomized complete block with four replications. The whole plots (main effect) were a nontreated control (no herbicides added), a preemergence (PRE) control (flumioxazin at 0.107 kg ai/ha, plus pendimethalin at 0.90 kg ai/ha), PRE followed by (fb) paraquat (0.21 kg ai/ha) plus nonionic surfactant (NIS at 0.25% v/v) POST, PRE fb paraquat (0.21 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha) POST, PRE fb paraquat (0.21 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha) plus S-metolachlor (1.47 kg ai/ha) POST, and PRE fb paraquat (0.21 kg ai/ha) plus acifluorfen (0.28 kg ai/ha) plus bentazon (0.56 kg ai/ha) plus acetochlor (1.26 kg ai/ha) POST. The PRE application was made at planting and activated with 1.3 cm of irrigation. POST herbicide treatments were applied 28 d after planting. Plots were maintained weed free by handweeding. The sub-plot effect consisted of four cultivars assigned randomly within each whole-plot. Georgia-06G (Branch, 2007), Georgia-14N (Branch and Brenneman, 2015), TUF-Runner™ '511' (Tillman and Gorbet, 2017), and FloRun™ '157' were evaluated.
Data collection included visual foliar injury ratings, visual stunting ratings, yield (kg/ha), and grade as percentage of total sound mature kernels (%TSMK). Visual foliar injury ratings (% chlorosis/necrosis) were evaluated at 3, 7, 11, and 14 d after treatment (DAT). Visual stunting (%) was measured at 3, 7, 11, and 14 DAT. The visual injury ratings (foliar injury and stunting) were ranked on a percentage scale of 0 to 100, with 100% being complete necrosis/death while 0% represented no injury.
The hull scrape method established peanut maturity (Williams and Drexler, 1981). Peanut digging and inversion occurred with a 2-row digger for Ty Ty, and a 6-row digger (Kelley Mfg. Co., Tifton, GA) in Plains. Pods were allowed to desiccate to approximately 10 to 15% moisture before harvest with a 2-row KMC harvester (Kelley Mfg. Co., Tifton, GA) in Ty Ty and a Columbo harvester (Columbo North America, Adel, GA) in Plains. Yields were then adjusted to 7% moisture for uniformity. Grade was determined by the USDA-AMS grading standards by the USDA Federal-State Inspection Service in Tifton, GA (USDA-AMS, 1997).
For data analyses, SAS 9.4 utilized the PROC MIXED function. Location and year were treated as random effects (there were no interactions), while herbicide and cultivar, as well as their interactions, were fixed effects. Similar trends allowed for data to be combined across site-year locations. Data were analyzed by analysis of variance (ANOVA) and differences between means were determined using Tukey's honestly significant difference test (𝜶=0.05) (Tukey, 1949).
Results and Discussion
Irrigated experiments
There were no significant interactions between herbicide and cultivar (Table 1). Herbicide treatment was significant for all variables except grade. For cultivars, there were differences for yield and grade.
Herbicide effect
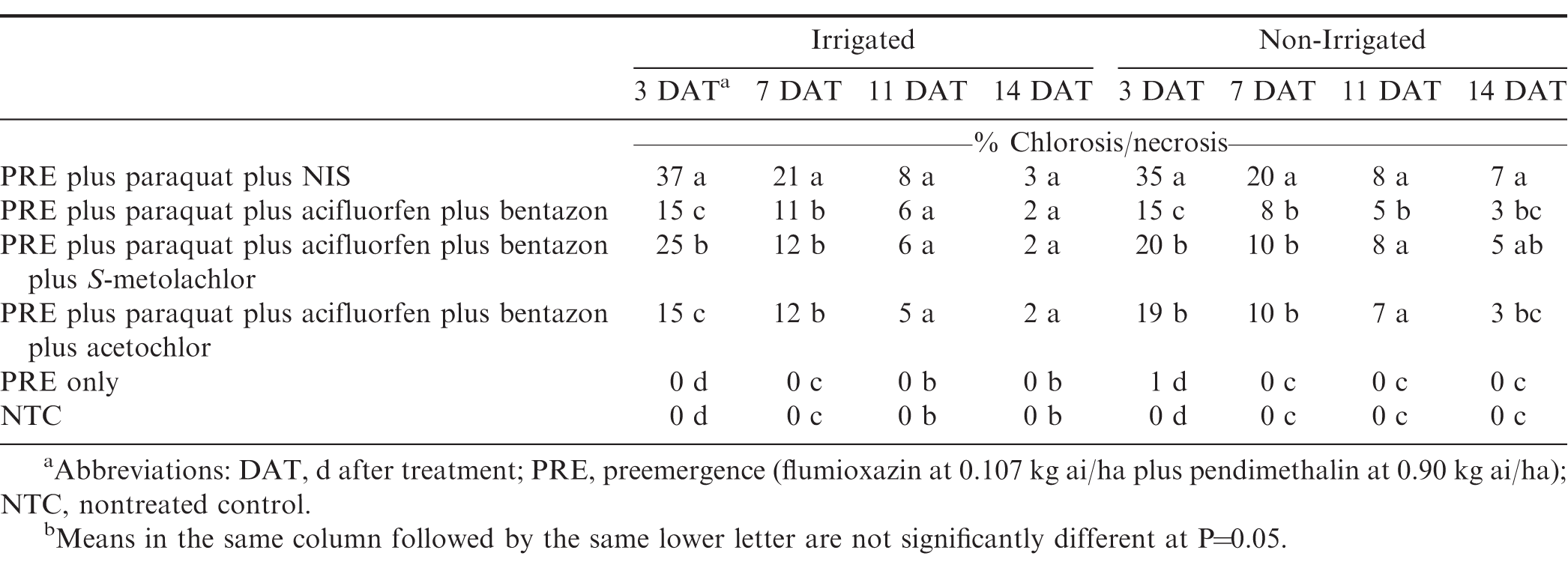
Leaf burn (% chlorosis/necrosis) and stunting (% stunting) decreased over time across all herbicide treatments (Table 2). At 3 DAT, paraquat plus acifluorfen plus bentazon, and paraquat plus acifluorfen plus bentazon plus acetochlor, resulted in the least amount of injury when compared to the other herbicide treatments. Including acifluorfen plus bentazon in tank-mixture with paraquat significantly reduced leaf burn at 3 and 7 DAT but subsequently had no influence on leaf burn. By 14 DAT all herbicide treatments exhibited less than 3% injury. Overall, there were negligible differences in vegetative injury between acetochlor and S-metolachlor, similar to previous research (Chaudhari et al., 2018).
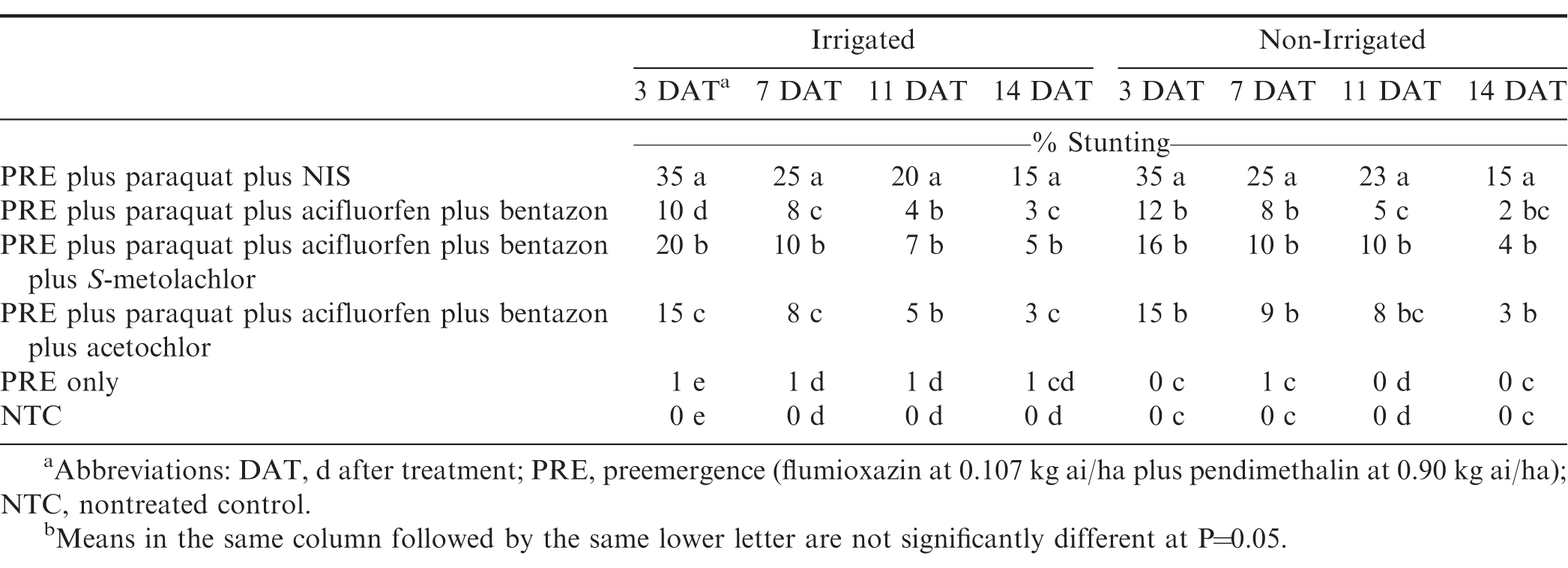
Paraquat alone caused the greatest amount of stunting injury (Table 3). The herbicide combinations that included acifluorfen plus bentazon all had less stunting than paraquat alone, for all ratings. Including bentazon in paraquat tank-mixtures is known to cause a reduction in stunting injury when compared to paraquat alone (Wehtje et al., 1992). S-metolachlor caused greater stunting at 3 and 7 DAT than acetochlor, but by 11 DAT both treatments were less than 7%. Stunting from acetochlor was similar to paraquat plus acifluorfen plus bentazon by 7 DAT.
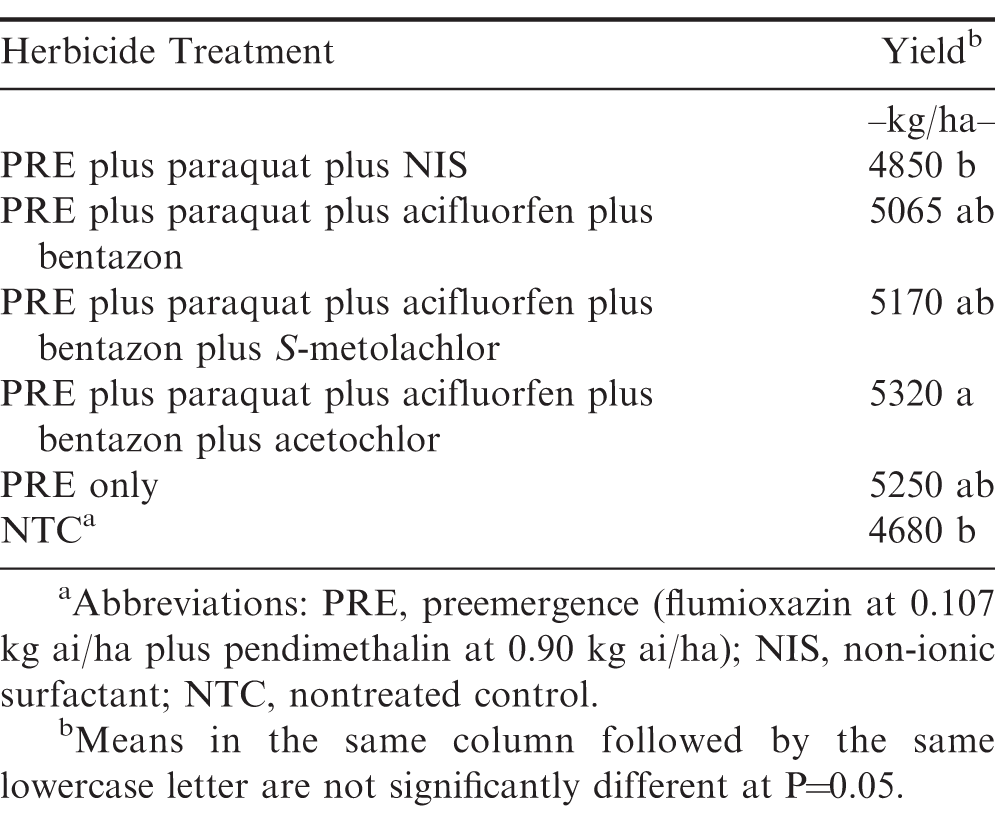
Although paraquat alone caused the greatest amount of peanut foliar leaf injury, there was no difference in yield from the nontreated control (Table 4). Paraquat plus acifluorfen plus bentazon plus acetochlor yielded greater than the nontreated control and paraquat alone. Herbicide treatment had no effect on grade (% TSMK) (Table 5).
Cultivar response
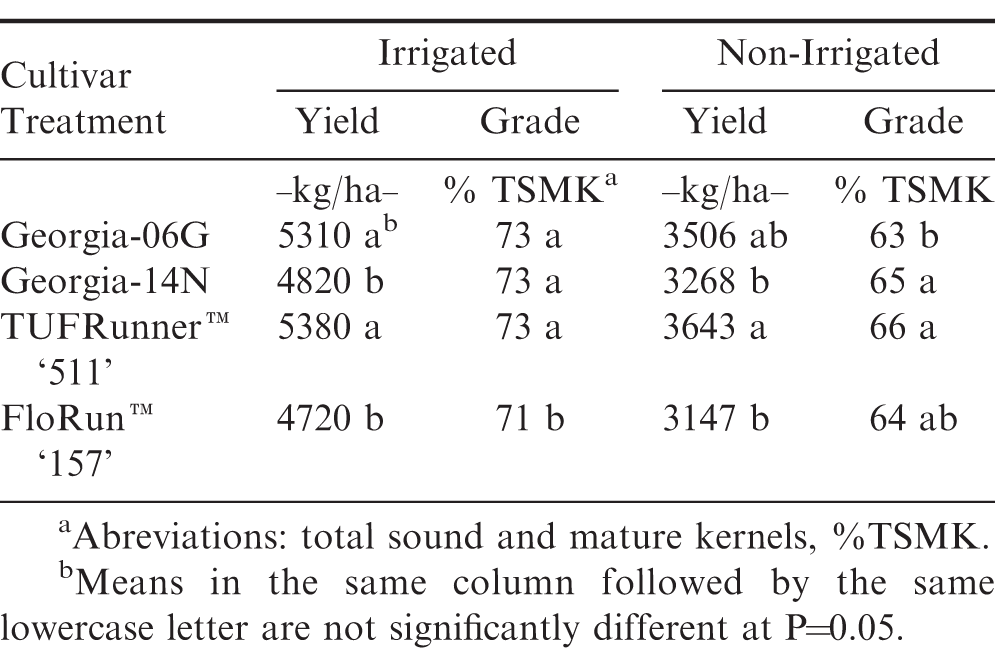
There were no differences in leaf burn or stunting injury for any of the tested cultivars (Table 1). Georgia-06G and TUFRunner™ '511' yielded greater than Georgia-14N and FloRun™ '157' (Table 5). Similar results were noted by Branch (2017). FloRun™ '157' had the lowest grade. Similar trends in yield and grade were noted by the UGA Statewide Variety Testing irrigated trials (Gasset et al., 2017) for the 2016 growing season.
Non-Irrigated Study
There were no interactions between herbicide and cultivar effects for any variable (Table 1). Herbicide treatment was significant for all injury (leaf burn and stunting) ratings but not yield or grade. No differences were observed for injury ratings for the cultivar treatment effect, but there were differences for yield and grade.
Herbicide effect
Overall, foliar injury decreased over time for all herbicide treatments (Table 2). Including acifluorfen plus bentazon in the tank-mixture resulted in a reduction in injury when compared to paraquat alone. Similar results were noted by Grey at al. (1995). S-metolachlor and acetochlor showed analogous results across all ratings, similar to previous research (Chaudhari et al., 2018).
Similar to the vegetative injury, stunting trended downward over time for all herbicide tank-mixtures (Table 3). Including acifluorfen plus bentazon reduced stunting from paraquat on peanut across all ratings. There were no differences among paraquat plus acifluorfen plus bentazon, paraquat plus acifluorfen plus bentazon plus S-metolachlor, and paraquat plus acifluorfen plus bentazon plus acetochlor. In non-irrigated peanut, S-metolachlor and acetochlor in tank-mixture with paraquat are not as injurious as paraquat alone, as long as they are used with acifluorfen plus bentazon. By the final injury rating (14 DAT), herbicide treatments including acifluorfen plus bentazon had less than 5% stunting while paraquat treated peanuts still had a 15% reduction in size. While herbicide treatment did influence injury, herbicide treatment had no effect on yield or grade (Table 1).
Cultivar responses
There were no differences in leaf burn or stunting injury for any of the tested cultivars (Table 1). TUFRunner™ '511' yielded more than Georgia-14N and FloRun™ '157', which is similar to the irrigated trial (Table 5). TUFRunner™ '511' and Georgia-14N had better % TSMK than Georgia-06G. These cultivars followed similar trends for grade as the results of the UGA Statewide Variety Testing non-irrigated trials (Gasset et al., 2017).
Summary and Conclusions
Leaf burn and stunting declined over time for both experiments across all herbicide treatments indicating peanuts plants ability to produce new tissue after paraquat injury. Including acifluorfen plus bentazon in the POST herbicide tank-mixture containing paraquat reduced visible injury to the peanut crop similar to previous research (Grey et al., 1995; Wehtje et al., 1992). Grade variation was due to cultivar differences for both the irrigated and non-irrigated studies. Similar variation in grade was noted previously by Wright et al. (1986) using different runner-type peanut cultivars. For the non-irrigated study, while herbicide injury did occur, herbicide treatment had no overall effect on peanut yield. Paraquat injury has been shown to have negligible effects on peanut yield (Carley et al., 2009; Johnson et al., 1993) previously as well.
With the given supporting data, the use of the herbicide tank-mixtures from these experiments can be recommended with the given runner-type peanut cultivars without fear of negative yield impact for irrigated and non-irrigated peanut. Future research is warranted to determine the effects of these herbicide tank-mixtures on different market-type peanut cultivars with other growth characteristics.
Acknowledgements
This research was supported by the National Peanut Board and the Georgia Peanut Commission, and seed donations were received from the Georgia Seed Development Commission. The authors would like to thank Kristen Pegues, Hunter Bowen, Hunter Hayes, Sarah Chance, Chris Cromer, Sidney Cromer, and the staff members of the UGA Ponder farm and UGA Southwest Research and Education Center for technical assistance.
Literature Cited
K. J Boote, (1982). Growth stages of Peanut (Arachis hypogaea L.). Peanut Sci 9: 35- 40.
B Branch, (2007). Registration of 'Georgia-06G' peanut. J. of Plant Reg 1: 120.
Branch, B 2017 Peanut breeding update, pp 6- 10 In 2017 Peanut Update Georgia Coop. Ext. Serv., Athens GA CS-17-018.
W. D., Branch, and T. B Brenneman (2015). Registration of 'Georgia-14N' peanut. J. of Plant Reg 1: 159- 161.
Buchanan, G. A., D. S Murray, and E. W Hauser 1982 Weeds and their control in peanuts, pp 206- 249 In H. E Pattee and C. T Young (eds) Peanut Sci. and Technol Am. Peanut Res. Educ. Soc.
D. S., Carley, D. L Jordan, R. L Brandenburg, and L. C Dharmasri (2009). Factors influencing response of virginia market type peanut (Arachis hypogaea L.) to paraquat under weed free conditions. Peanut Sci 36: 180- 189.
Gasset, J. D., L Day, D. G Dunn, H Jordan Jr., S. S LaHue 2017 Yield and grade performance, peanut variety trial, p 7 In Georgia 2016 Peanut, Cotton, and Tobacco Performance Tests Bulletin Georgia Coop. Ext. Serv., Athens, GA.
T. L., Grey, G. R Wehtje, R. H Walker, and K. P Paudel (1995). Comparison of imazethapyr and paraquat-based weed control systems in peanut (Arachis hypogaea L.). Weed Technol 9: 813- 818.
Harris, G 2018 Peanut fertility checklist, p 3 In UGA Peanut Production Quick Reference Guide Georgia Coop. Ext. Serv., Athens, GA.
E. W., Hauser, G. A Buchanan, and W. J Ethridge (1975). Competition of Florida beggarweed and sicklepod with peanuts. I. weed-free maintenance and weed competition. Weed Sci 23: 368- 372.
L. V Hill, and P. W Santelmann (1969). Competitive effects of annual weeds on Spanish peanuts. Weed Sci 17: 1- 2.
W. C., Johnson, J. R Chamberlin, T. B Brenneman, J. W Todd, B. G Mullinix, and J Cardina (1993). Effects of paraquat and alachlor on peanut (Arachis hypogaea L.) growth, maturity, and yield. Weed Technol 7: 855- 859.
S., Chaudhari, D Jordan, T Grey, E Prostko, and K Jennings (2018). Weed control and peanut (Arachis hypogaea L.) response to acetochlor alone and in combination with various herbicides. Peanut Sci 45: 45- 55.
Kemerait, R., A Culbreath, J Beasley, E Prostko, T Brenneman, S Tubbs, R Srinivasan, M Abney, S Monfort, A Rabinowitz, G. B Tillman, N Dufault, D Rowland, M Mulvaney, A Hagan, J Sarver, D Anco, and N Smith 2017 Peanut Rx, minimizing diseases of peanut in the southeastern United States Univ. of Georgia Coop. Ext. Serv, Athens, GA.
Monfort, W. S 2017 Peanut Cultivar Options, pp 4- 6 In 2017 peanut update Georgia Peanut Commission, Tifton, GA.
Porter, W 2017 Peanut irrigation update, pp 19- 24 In 2017 peanut update Georgia Peanut Commission, Tifton, GA.
J. R., Stansell, and J. E Pallas (1985). Yield and quality of florunner peanut to applied drought at several growth stages. Peanut Sci 12: 64- 70.
J. R., Stansell, J. L Shepherd, J. E Pallas, R. R Bruce, N. A Minton, D. K Bell, and L. W Morgan (1976). Peanut responses to soil water variables in the Southeast. Peanut Sci 3 ((1)): 44- 48.
B. L Tillman, and D. W Gorbet (2017). Registration of "TUFRunner '511'" peanut. J. of Plant Reg 11 ((3)): 235- 239.
J.W Tukey, (1949). Comparing individual means in the analysis of variance. Biometrics 5: 99- 114
USDA-AMS 1997 United States standards for grades of shelled runner type peanuts Available at http://www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5050496 (verified 20 April 2018) USDA Agric. Marketing Serv., Washington, D.C.
USDA-NASS 2018 Survey of peanut acres harvested in the United States USDA National Agricultural Statistics Service quick stats https://quickstats.nass.usda.gov/results/4D5FEDBA-6C1D-3D70-BAE1-6EA8AA74C437 (accessed 21 Mar. 2018).
USDA-NRCS 2018 Official soil series descriptions. Available at (accessed 20 April 2018) USDA Natural Resources Conversation Serv., Washington, D. C.
G., Wehtje, J. A McGuire, R. H Walker, and M. G Patterson (1986). Texas Panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat. Weed Sci 34: 308- 311.
G., Wehtje, J. W Wilcut, and J. A McGuire (1992). Influence of bentazon on the phytototxicity of paraquat to peanuts (Arachis hypogaea) and associated weeds. Weed Sci 40: 90- 95.
J. W Wilcut, and C. W Swann (1990). Timing of paraquat applications for weed control in virginia-type peanuts (Arachis hypogaea). Weed Sci 38: 558- 562.
J. W., Wilcut, G. R Wehtje, T. A Cole, T. V Hicks, and J. A McGuire (1989). Postemergence weed control systems without dinoseb for peanuts (Arachis hypogaea). Weed Sci 37: 385- 391.
Wilcut, J.W., A.C York, W.J Grichar, and G.R Wehtje 1995 The biology and management of weeds in peanut p 207- 224 In H. E Patteeand H.T Stalker (eds.) Advances in Peanut Science Am. Peanut Res. Educ. Soc.
E.J., Williams, and J. S Drexler (1981). A non-destructive method for determining pod maturity. Peanut Sci 8: 134- 141.
F. S., Wright, D. M Porter, N. L Powell, and B. B Ross (1986). Irrigation and tillage effects on peanut yield in Virginia. Peanut Sci 2: 89- 92.
Notes
- First, second, and third authors: Graduate Research Associate, Associate Professor, and Professor, Dept. of Crop and Soil Sciences, University of Georgia, Tifton, GA 31639; fourth author: Assistant Professor, Crop, Soil, and Environmental Sciences Dept., Auburn University, Auburn, AL 36849. [^] *Corresponding author's E-mail: kme51221@uga.edu
Author Affiliations