Introduction
In the southeast U.S., the most important and endemic foliar diseases of peanut (Arachis hypogaea L.) are tomato spotted wilt caused by Tomato spotted wilt virus (TSWV), early leafspot caused by Cercospora arachidicola Hori, and late leafspot caused by Cercosporidium personatum (Bert & Curt.) Deighton, respectively (Branch and Culbreath, 2013). The primary control for TSWV is resistant cultivars; whereas, fungicides are routinely used for leafspot and soilborne disease control. However, the total number of fungicide spray applications can vary from four to seven during a typical growing season depending upon low or high risk assessment of each field and specific fungicide recommendations (Kemerait et al., 2016.)
Irrigation and pesticides currently used in peanut production are very effective, but also expensive. During 2015, total cost for irrigated production in Georgia with an expected peanut pod yield of 5269 kg/ha was estimated at $2,388/ha, which includes both variable and fixed costs but excludes land and return to management (Smith and Smith, 2015). These values compare with $1,797/ha estimated total cost for nonirrigated or dryland peanut production in Georgia with an expected yield of 3811 kg/ha. Thus, irrigation adds $591/ha to peanut production cost and pesticides accounted for approximately 40%; of the total expense in material, fuel, maintenance, and labor. Consequently, Georgia peanut growers are seeking disease resistant and drought tolerant cultivars to substantially lower overall production cost in order to maintain relative economical return and enhance the sustainability of their farming operation.
The objective of this research was to evaluate the performance of runner and virginia-type peanut cultivars with minimum inputs and without irrigation. Drought tolerance has been defined as, "the ability of one genotype to be more productive with a given amount of soil moisture than another genotype" (Quizenberry, 1982). Since gross dollar values combine yield and grade, it was also considered an index criterion for assessing peanut genotypic performance under water-limiting environments when production cost is identical for each genotype in the experiment.
Materials and Methods
Fifteen runner and virginia-type peanut cultivars were evaluated for five-years (2011-15); whereas, seventeen runner and virginia-type cultivars were evaluated for four-years (2012-2015). Each year, field tests were used to evaluate cultivars for TSWV and total disease incidence, and gross dollar value, and were conducted on a Tifton loamy sand soil type (fine-loamy, siliceous, thermic Plinthic Kandiudult) at the Gibbs Research Farm (latitude: 31.43°;N and longitude: -83.59°W) near the Coastal Plain Experiment Station in Tifton and on a Greenville sandy clay loam soil type (clayey, kaolinitic, thermic Rhodic Kandiudult) at the Southwest Georgia Research and Education Center (latitude: 32.04°N and longitude: -84.37°W) near Plains, GA. At both locations, land preparation was the same, moldboard plowing and then bedding, prior to planting each year. Plots consisted of two rows spaced 1.8 m apart by 6.1 m long. At Tifton, planting dates were 11 April 2011, 9 April 2012, 10 April 2013, 4 April 2014, and 9 April 2015. At Plains, planting dates were 20 April 2011, 25 April 2012, 23 April 2013, 28 April 2014, and 5 May 2015.
Each test involved minimum inputs without irrigation. Three fungicide applications (tebuconazole plus chlorothalonil at 221 and 1,262 g a.i./ha, respectively) were included during each growing season beginning 37 d after planting and then applied at 28 d intervals. On-site rainfall was recorded monthly and compared to the long-term average for each location obtained from the Georgia automated environmental monitoring network (Flitcroft, 2015). These field tests were in a three-year crop rotation following corn (Zea mays L.) and cotton (Gossypium hirsutum L.). Individual entries were dug near optimum maturity based upon the hull-scrape method determined from adjacent border rows (Williams and Drexler, 1981).
Incidence of TSWV was first assessed at midseason approximately 70 d after planting, when TSWV is usually the only disease. Percentages of total disease were also scored prior to digging, which included primarily TSWV and any soilborne disease. A disease hit equaled one or more diseased plants in a 30.5 cm section of row (Rodriguez-Kabana et al., 1975).
After digging with a two-row digger and threshing with a plot combine, pods were dried with forced warm air to 6%; moisture. Pod samples were then hand-cleaned over a screen table before weighing for yield determinations. Market grades were determined according to federal state inspection service procedures for runner and virginia-type peanut, respectively (USDA-AMS, 1998). Gross dollar values were calculated from yield and grade based upon USDA-Farm Service Agency (FSA) peanut loan schedules for each crop year.
The experimental design was a randomized complete block with five replications. Data from each individual test was combined across years by location subjected to analysis of variances. Waller-Duncan's T-test (k-ratio = 100) was used for mean separation of significant differences (P ≤ 0.05).
Results and Discusson
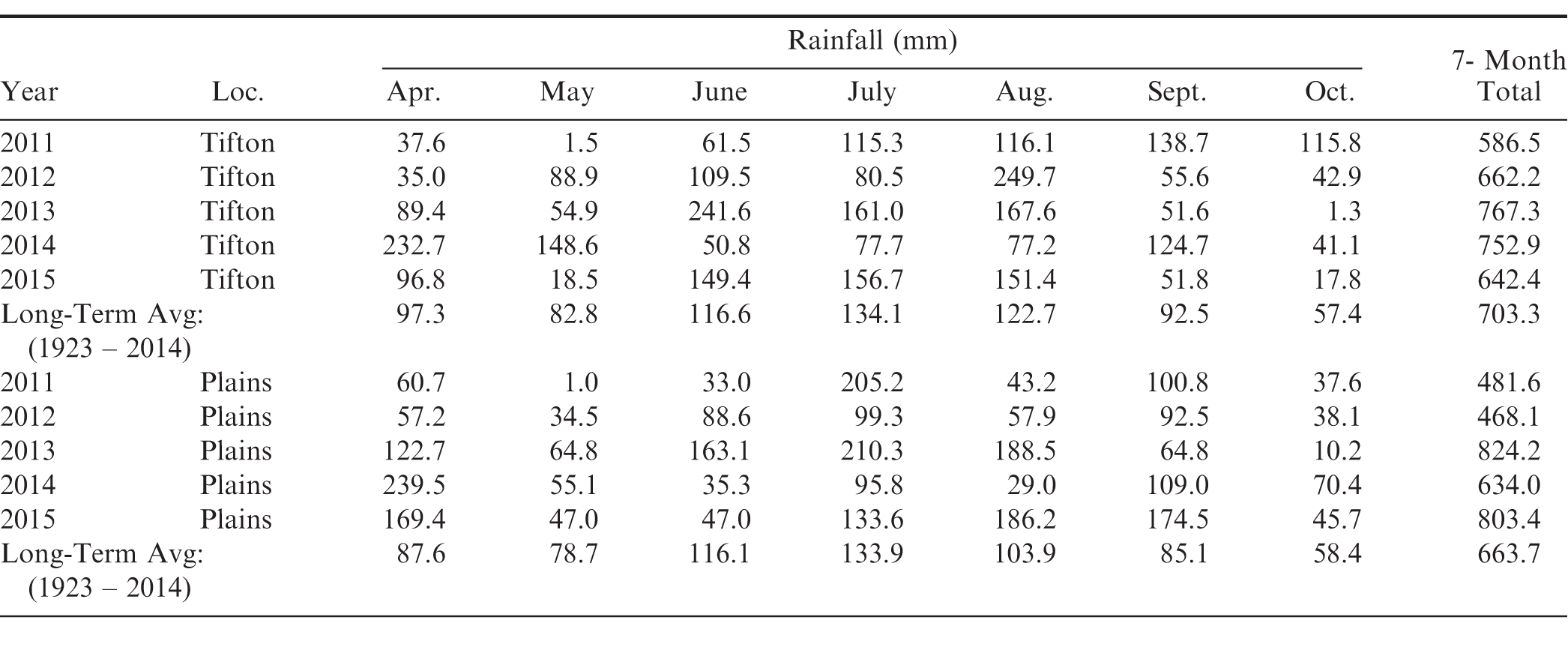
Total rainfall during the five growing seasons at both Georgia locations were below the long-term (91 yr) average for three out of the five years (Table 1). Likewise, approximately half of all monthly rainfall during the five growing seasons at both locations was also below the long-term average. Even during the higher rainfall seasons (2013 and 2014 at Tifton and 2013 and 2015 at Plains), there were at least two or more months where drought stress was a major limiting factor.
Leafspot was not severe until 2015, when rainfall was twice the long-term average at Plains during September (Table 1). Even with good rotation, environmental conditions were very favorable particularly for late leafspot development at this location in 2015. Significant cultivar differences (P ≤ 0.05) were observed among both runner and virginia-type cultivars for late leafspot on a visual rating scale of 0-9, where 0 = no leafspot and 9 = died defoliated plants. Overall, 'Georgia-08V' (Branch, 2009) had the highest late leafspot rating of 8.6 and 'CHAMPS' (Mozingo et al., 2006) was second highest at 8.2; whereas, 'Georgia-12Y' (Branch, 2013) had the lowest late leafspot rating of 4.8 followed by 'Bailey' (Isleib et al., 2011), 'Tifguard' (Holbrook et al., 2008) and 'Georgia-11J' (Branch, 2012) at 5.6, respectively.
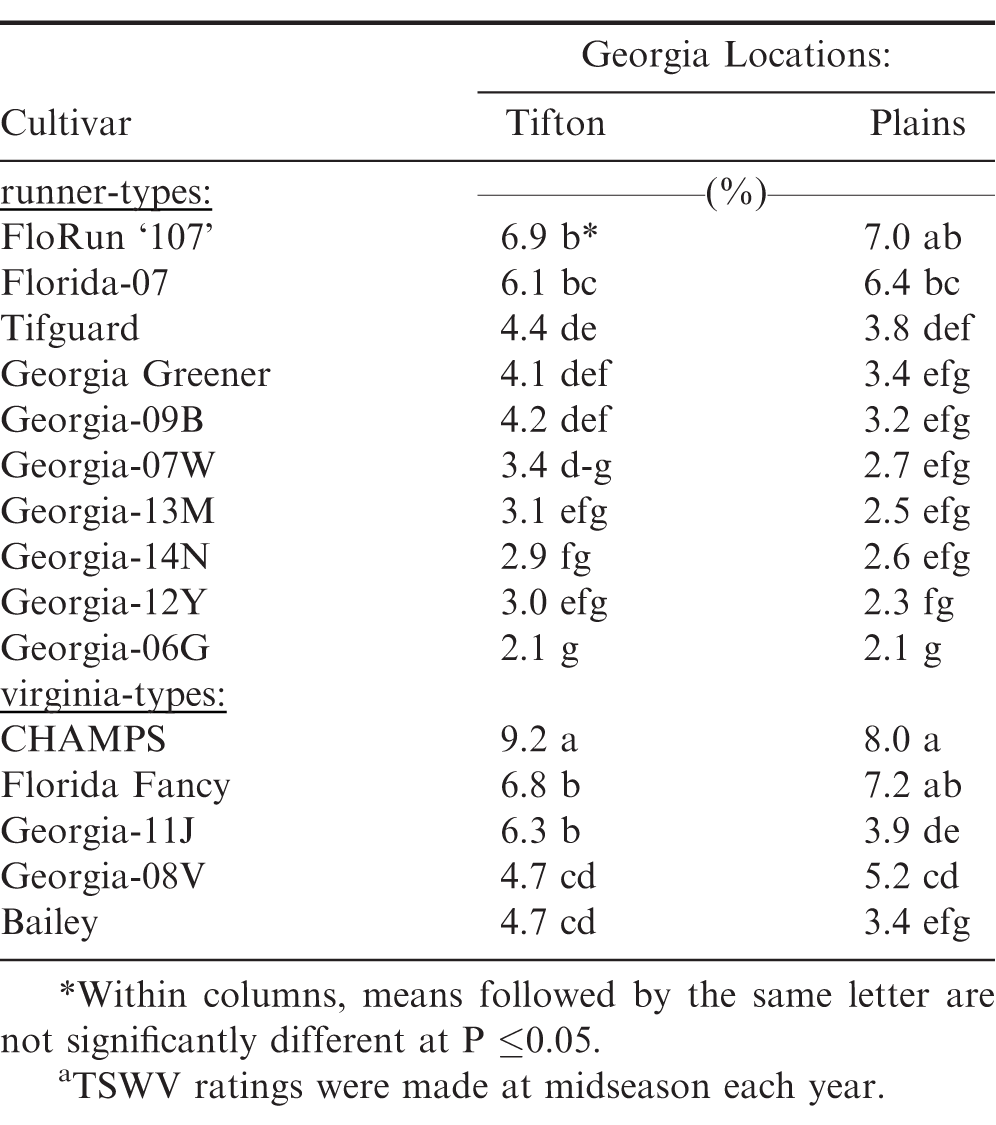
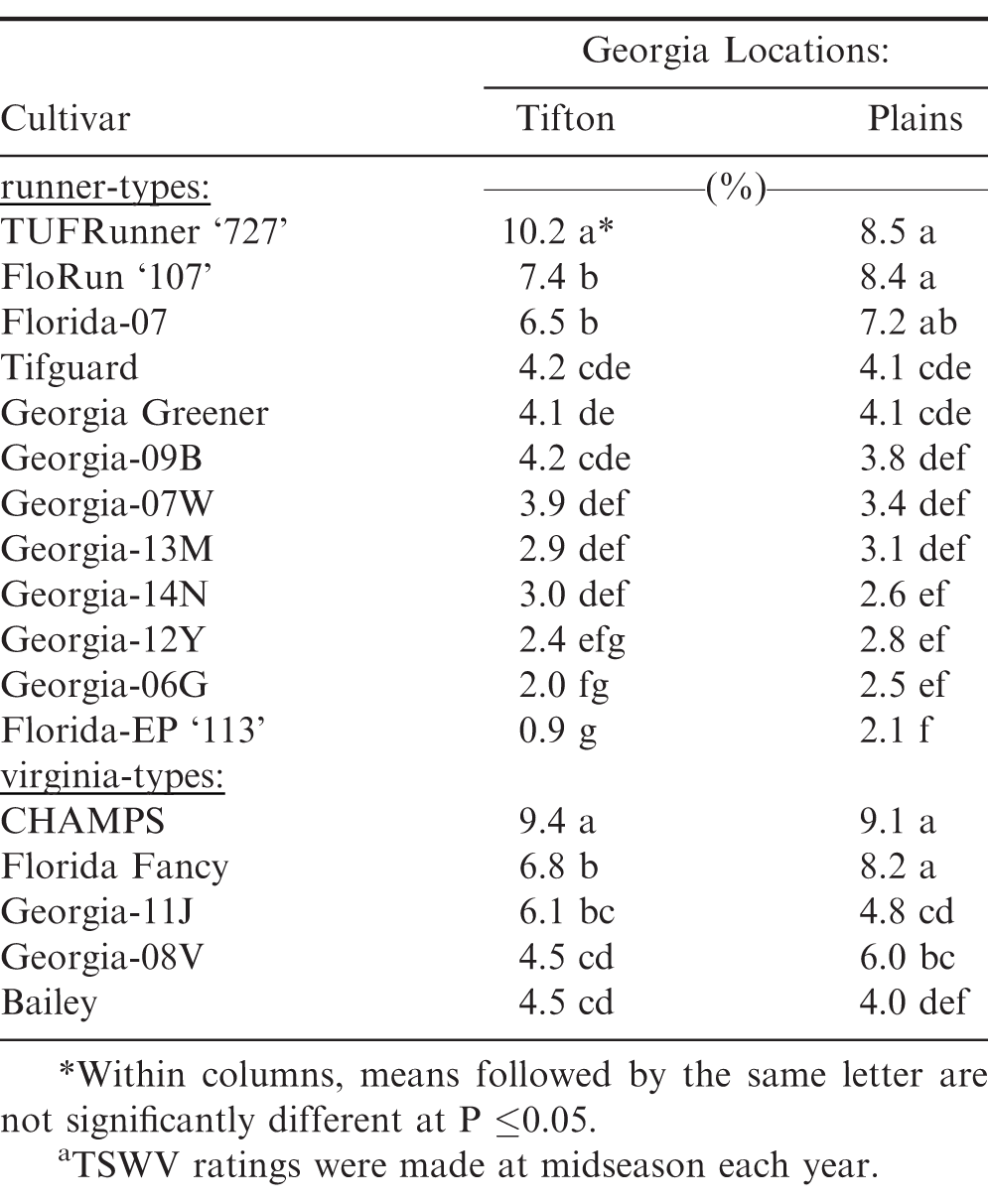
Significant differences were also observed among the 15 peanut cultivars for five-year average TSWV incidence at midseason (Table 2). 'Georgia-06G' (Branch, 2007) had the lowest percent TSWV incidence among both runner and virginia-type peanut cultivars. However, it was not different from the runner-type cultivars Georgia-12Y, 'Georgia-14N' (Branch and Brenneman, 2015), 'Georgia-13M' (Branch, 2014), and 'Georgia-07W' (Branch and Brenneman, 2008) at both locations; whereas, Bailey had the lowest TSWV among the viriginia-type cultivars at both Georgia locations. However, Bailey was not different from Georgia-08V at Tifton or Georgia-11J at Plains for TSWV incidence.
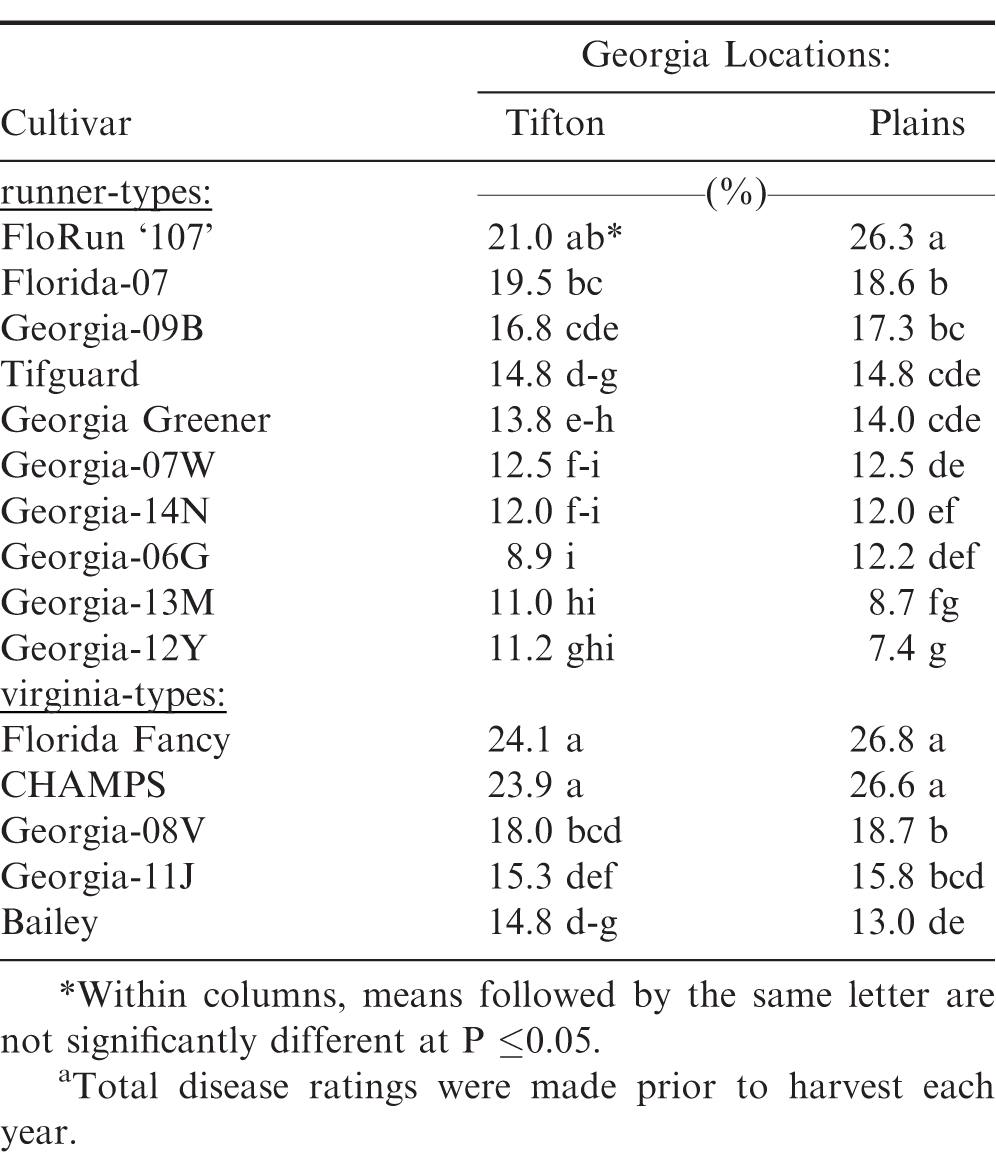
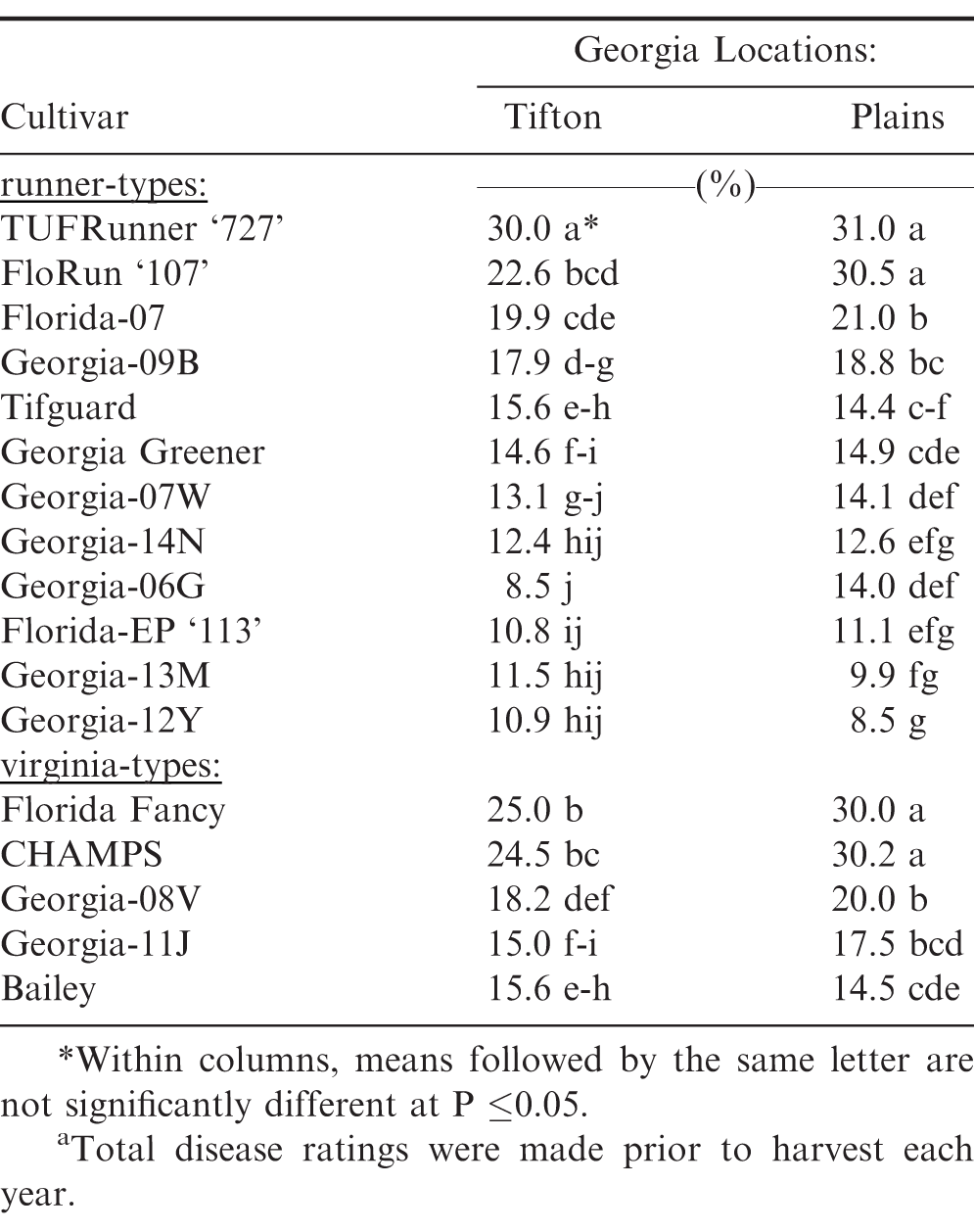
Significant differences were also observed among the peanut cultivars for five-year average total disease incidence prior to harvest (Table 3). Georgia-12Y, Georgia-13M, and Georgia-06G had the lowest percent of total disease incidence among runner-type cultivars; whereas, Bailey had the lowest total disease among the virginia-type cultivars at both locations. However, it was not different from Georgia-08V and Georgia-11J at Tifton and Georgia-11J at Plains.
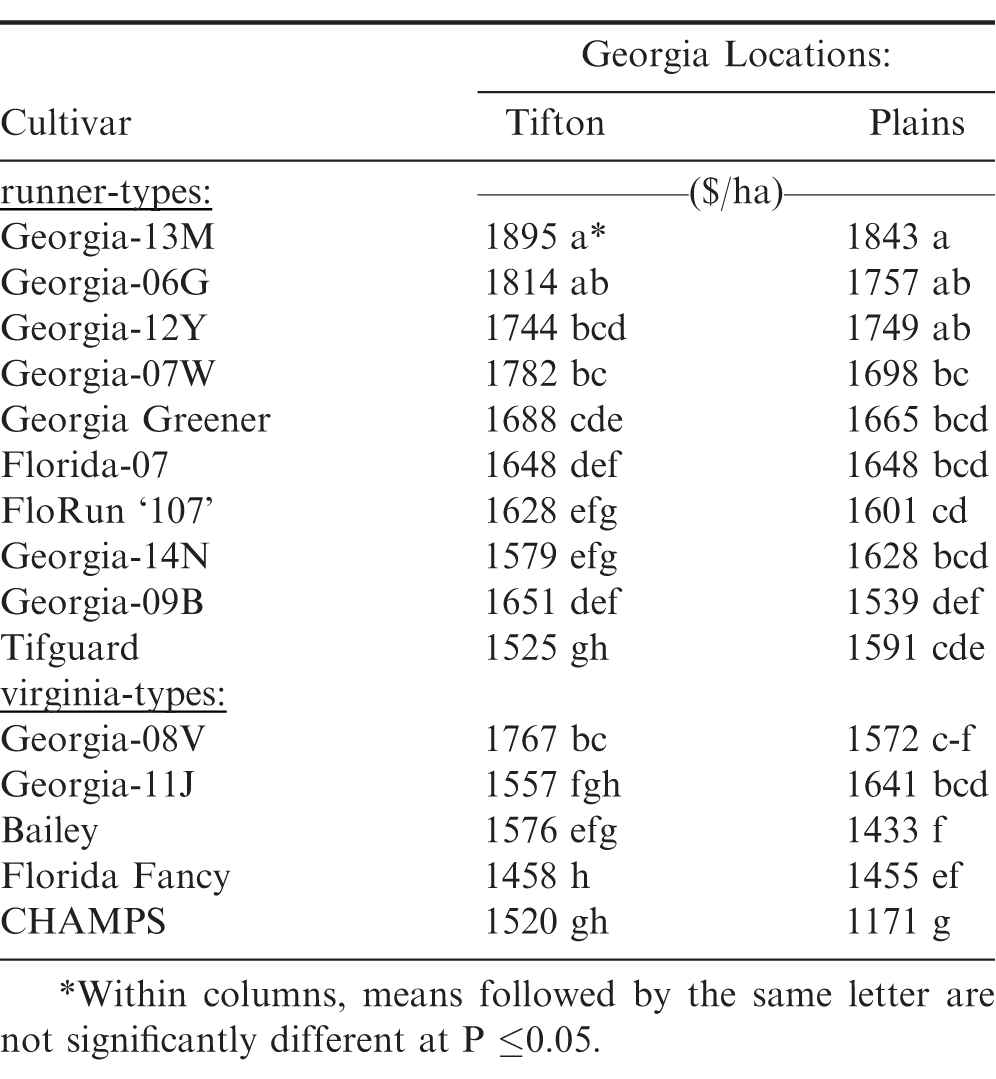
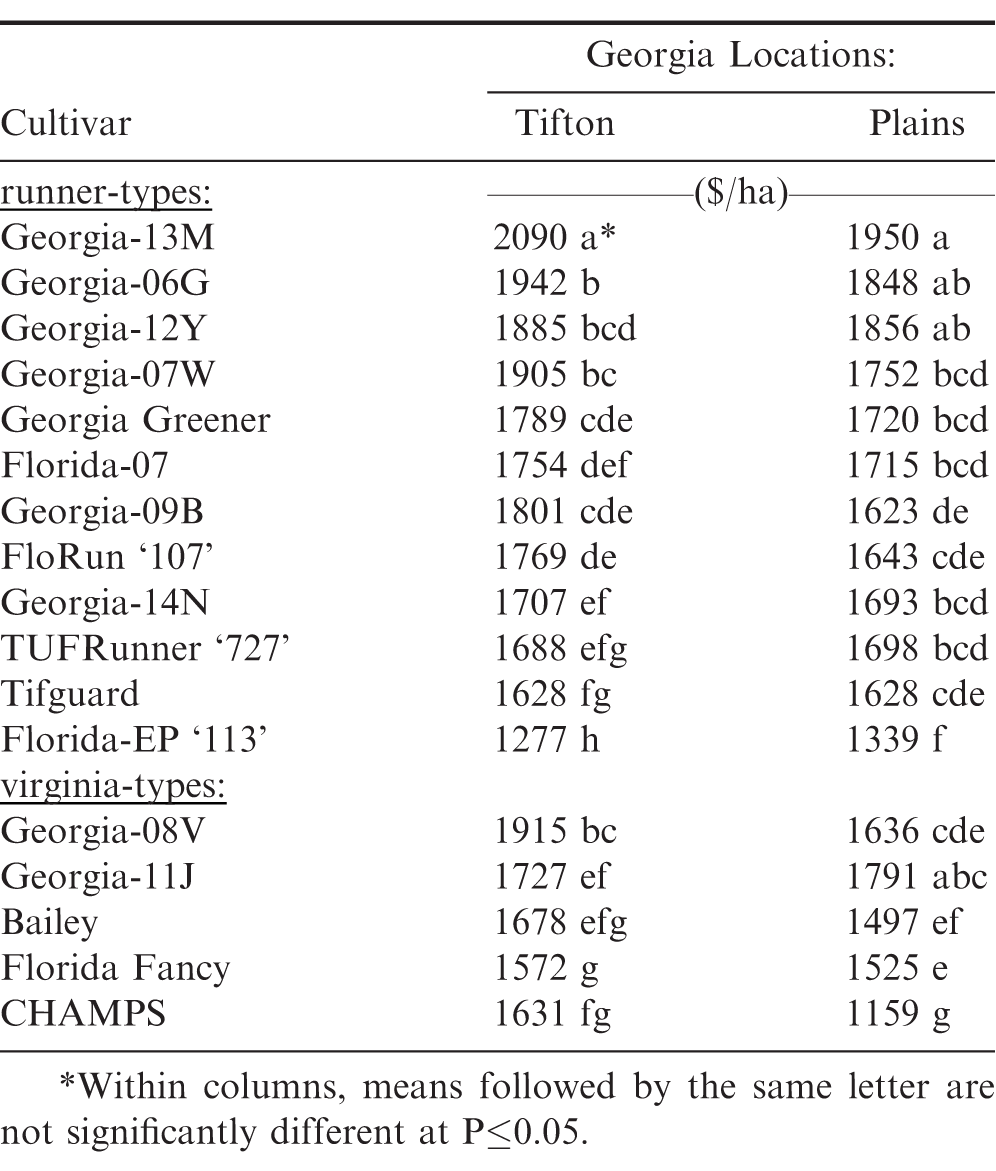
Likewise, significant differences were observed among these same 15 peanut cultivars for five-year average gross dollar value returns (Table 4). Georgia-13M had the greatest dollar value at the two Georgia locations among both runner and virginia-type cultivars. However, it was not greater than Georgia-06G at Tifton, and Georgia-06G and Georgia-12Y at Plains. Georgia-08V had the greatest dollar value among the virginia-type cultivars at Tifton; but Georgia-11J had the greatest dollar value at Plains. However at the Plains location, Georgia-11J was not different from Georgia-08V.
Two additional runner-type cultivars (TUFRunner '727' and Florida-EP '113') were included in the four-year averages (2012-15). Similarly, significant differences were observed for four-year average TSWV incidence (Table 5). Florida-EP '113' had the lowest percent TSWV incidence among both runner and virginia-types which agrees with another study involving Florida-EP '113 (McKinsey and Tillman, 2017); whereas, TUFRunner '727' had among the highest percent TSWV incidence. However, Florida-EP '113' was not different from Georgia-06G and Georgia-12Y at Tifton; and several other cultivars at Plains in TSWV incidence.
Significant differences were also observed among these 17 peanut cultivars for four-year average total disease incidence (Table 6). Georgia-12Y, Georgia-13M, Florida-EP '113', Georgia-06G, Georgia-14N, and Georgia-07W had the lowest total disease incidence among runner-types; whereas, Bailey, Georgia-11J, and Georgia-08V had the lowest total disease incidence among virginia-types at both Georgia locations.
Likewise, significant differences were found among these 17 peanut cultivars for four-year average gross dollar values (Table 7). Georgia-13M again had the greatest dollar value among both runner and virginia-type cultivars. However, it was not greater than Georgia-06G and Georgia-12Y at the Plains location. Georgia-08V had the greatest dollar value at the Tifton location; whereas, Georgia-11J and Georgia-08V had the greatest dollar values among the virginia-type cultivars at the Plains location.
The high-oleic, small-seeded, runner-type peanut cultivar Georgia-13M had the highest dollar values at the two Georgia locations for both the four and five-years (Tables 4 and 7). The results from this study would suggest that Georgia-13M, Georgia-06G, and Georgia-12Y each have better combined disease resistance and drought tolerance than the other runner and virginia-type cultivars which agrees with a previous report for Georgia-06G having the greater dollar value return in both maximum and minimum tests (Branch and Fletcher, 2010). Performance of these three cultivars when grown with minimum-inputs and without irrigation should have major potential impact for dryland peanut production, and demonstrates significant improvement in cultivar development over an earlier report by Branch and Fletcher (2004).
References
W. D Branch, (2007). Registration of 'Georgia-06G' peanut. J. Plant Reg 1: 120.
W. D Branch, (2008). Registration of 'Georgia-08V' peanut. J. Plant Reg 3: 143- 145.
W. D Branch, (2012). Registration of 'Georgia-11J' peanut. J. Plant Reg 6: 281- 283.
W. D Branch, (2013). Registration of 'Georgia-12Y' peanut. J. Plant Reg 7: 151- 153.
W. D Branch, (2014). Registration of 'Georgia-13M' peanut. J. Plant Reg 8: 253- 256.
W. D., Branch, and T. B Brenneman (2008). Registration of 'Georgia-07W' peanut. J. Plant Reg 2: 88- 91.
W. D., Branch, and T. B Brenneman (2015). Registration of 'Georgia-14N' peanut. J. Plant Reg 9: 159- 161.
W. D Branch, and A. K Culbreath (2013). Yield performance and pest resistance among peanut genotypes when grown without fungicides and insecticides. Crop Protection 52: 22- 25.
W. D., Branch, and S. M Fletcher (2004). Evaluation of advanced Georgia breeding lines with reduced-input and without irrigation. Crop Protection 23: 1085- 1088.
W. D., Branch, and S. M Fletcher (2010). Agronomic performances and economic return among peanut genotypes with maximum and minimum production inputs. Peanut Sci 37: 83- 91.
Flitcroft, I 2015 Georgia automated environmental monitoring network Griffin, GA University of Georgia Online at http://www.georgiaweather.net Accessed: March 14, 2017.
C. C, Holbrook, P Timper, A. K Culbreath, and C. K Kvien (2008). Registration of 'Tifguard' peanut. J. Plant Reg 2: 92- 94.
T. G., Isleib, S. R Milla-Lewis, H. E Pattee, S. C Copeland, M. C Zuleta, B. S Shew, J. E Hollowell, T. H Sanders, L. O Bean, K. W Hendrix, M Balota, and J. W Chaplin (2011). Registration of 'Bailey' peanuts. J. Plant Reg 5: 27- 39.
Kemerait, R., A Culbreath, E Prostko, T Brenneman, A Smith, S Tubbs, R Srinivasan, M Abney, S Monfort, B Tillman, N Dufault, D Rowland, M Mulvaney, A Hagan, J Sarver, D Anco, N Smith, 2016 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, In:: W.S Monfort (ed.), 2016 Peanut Production Update. Univ. of Georgia Coop. Ext. Service Rept. CSS-16-0115.
J. L McKinney, and B. L Tillman (2017). Spotted wilt in peanut as impacted by genotype resistance, planting date, and plant population. Crop Sci 57: 130- 136.
R. W., Mozingo, T. A Coffelt, P. M Phipps, and D. L Coker (2006). Registration of 'CHAMPS' peanut. Crop Sci 46: 2711- 2712.
Quizenberry, J. E 1982 Breeding for drought resistance and plant water use efficiency Pp 193- 212 In:: M. N Christiansenand C. F Lewis (eds.) Breeding Plants for Less Flavorable Environments John Wiley Sons, New York, NY.
R., Rodriguez-Kabana, P. A Backman, and J. C Williams (1975). Determination of yield losses to Sclerotium rolfsii in peanut fields. Plant Dis. Rep 59: 855- 858.
Smith, N. B and A Smith 2015 Peanut outlook and cost analysis Pp 3- 15 In:: E. P Prostko (ed) 2015 Peanut Update. Univ. of Georgia, Coop. Ext. Service Rept. CSS-15-0105.
USDA - Agricultural Marketing Service 1998 Farmer's stock peanuts inspection instructions U.S. Dept. of Agric.-Agric. Mkt. Ser. Fruit and Veg. Div USDA-ARS, Washington, D.C.
J. E Williams, and J. S Drexler (1981). A non-destructive method for determining peanut pod maturity. Peanut Sci 8: 134- 141.
Notes
- Professor, Department of Crop and Soil Sciences, University of Georgia, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793. [^]
- Professor Emeritus, Department of Agricultural and Applied Economics, University of Georgia, Georgia Experiment Station, Griffin, GA 30223. [^] *Corresponding author: Tel.: 229 386 3561; Fax: 229 386 7293
Author Affiliations
Email address: wdbranch@uga.edu (W.D. Branch).