Introduction
Peanut (Arachis hypogaea L.) is an economically important crop in the U.S. with an approximate value of 1 billion dollars (Anonymous, 2011). Several market types are grown in the U.S. and include runner, virginia, spanish, and valencia. Runner and virginia market types belong to subspecies hypogaea var. hypogaea, whereas valencia market types belong to ssp. fastigiata var. fastigiata and spanish market type to ssp. fastigiata var. vulgaris. Peanuts are grown in ten states in the U.S. Production can be divided into three main growing regions namely Southeast (Georgia, Florida, Alabama and Mississippi), Southwest (Texas, Oklahoma and New Mexico), and Virginia-Carolina, which account for 71, 15, and 14%, respectively, of the total acreage in the U.S. (AOSCA, 2011).
Peanut cultivation is constrained by foliar and soilborne diseases caused by several pathogens (Kokalis-Burelle et al., 1997). Pod rot is an economically significant disease which reduces yield and the market value of peanut (Kokalis-Burelle et al., 1997). Typically, soilborne mycelial pathogens associated with pod rot include Rhizoctonia solani, Sclerotium rolfsii, and Pythium spp. (Kokalis-Burelle et al., 1997). Under favorable environmental conditions, Rhizoctonia pod rot, caused by R. solani, can decrease yield by 22 to 28% (Besler et al., 2003). In fields surveyed in North Carolina for pod rot pathogens over a 3-year period, Hollowell et al. (1998) found that Rhizoctonia spp. and Pythium spp. were the most frequently isolated microorganisms from rotted pods.
In New Mexico, peanut (Valencia type) is predominantly produced in the eastern part of the state with an area of 4,047 ha harvested producing 16,000 tons and is valued at approximately 13.3 million dollars (NMDA, 2010). Pod rot is an economically important disease in peanut production in the state. Valencia peanut is sold in the in-shell market and any discoloration due to pod rot can reduce the premium to the growers by $50 per ton. It is not known whether pathogens reported as causal agents of pod rot in other regions are the same type of microorganisms associated with pod rot in New Mexico. Elucidation of the composition of pathogenic microorganisms associated with pod rot has implications in breeding programs and the management of this disease. Efforts in breeding peanut for resistance to pod rot will be strengthened if the composition of pathogenic populations associated with pod rot is known. Such information will enable off-season screening of germplasm under greenhouse conditions.
The primary focus of this study was to isolate and identify microorganisms associated with pod rot of Valencia peanut in New Mexico. The secondary focus of this study was to characterize the microorganisms from root and stem from the same plants selected for pod rot assessment. The merit of this secondary focus is the determination of whether microorganisms associated with pod rot are similar to those found in the roots and stems of plants with pod rot symptoms. Although plants with pod rot do not typically display any rotting symptoms on the stem or root, microorganisms associated with pod rot may be present in these asymptomatic tissues. Therefore, these tissues may represent additional reservoirs for pod rot pathogens. Characterization of the microorganisms present in roots and stems from plants displaying pod rot has not been done previously. Such characterization is important and should result in an increased understanding of the ecology of pod rot pathogens.
Materials and Methods
Sample collection and isolation of microorganisms
To characterize the populations of microorganisms associated with pod rot, peanut fields with known history of pod rot were identified. Fields were arbitrarily selected and surveyed in Curry and Roosevelt counties in eastern New Mexico, with 10 and 4 fields surveyed in 2005 and 2006, respectively. Surveys were conducted in different fields in each of the two years. Each field is approximately 48.6 ha, and is divided into seven sections or spans under a center-pivot irrigation system. Fifteen plants (three plants from each of five sectors) were sampled arbitrarily from each selected field, approximately 10 m away from the field perimeter. During the surveys, several above-ground symptoms were visible on plants in each field at different levels which included leaf spot with incidence of 10 to 20%, and stem blight and wilt with incidence of 1 to 3% of field area. For pod rot assessment, attempts were made to collect plants that did not display any of these symptoms.
Plant samples were stored in plastic bags on ice in a cooler, and transported to the laboratory. Samples were kept in a cold room at 5 °C and processed within 3 to 5 d. For isolation, each collected plant was rinsed free of soil under running water, and pods were harvested. The incidence of pod rot was recorded for each sample as the percentage of mature pods with rot or hull discoloration symptoms. Similarly, severity of pod rot or hull discoloration was visually estimated as the area of individual pods covered with symptoms of pod rot or hull discoloration.
From each plant, 5-cm segments were cut from the lower stem portion beginning at the crown. Similarly, 5-cm segments were cut from the upper portion of tap root starting at the crown. All fine roots were removed from the 5-cm tap root segments. All mature pods from each plant were removed and placed in a brown paper bag. Each bag was shaken for 30 to 45 sec, and 3 to 5 symptomatic pods with hull discoloration were randomly withdrawn from the bag for assessment. Pods were opened to separate seeds from hulls. Segments of stem and root, hull, and seeds were washed free of soil, submerged separately in 0.5% sodium hypochlorite for 3 min, rinsed once in sterile distilled water, and blot dried on sterile paper towels. For each stem or root segment, 0.5 cm of tissue was trimmed at both ends of the segment with a plant clipper. The remaining segment (4 cm in length) was sectioned into 1-cm portions to provide four 1-cm pieces per segment of stem or root. Similarly, four 1-cm pieces were cut from hulls. Seeds and pieces of hull, stem, and root were plated separately on acidified potato dextrose agar (APDA) and incubated at room temperature (20 to 23 °C) for two to three weeks. Colonies emerging from the plated seeds, pieces of hull, root, and stem were transferred to APDA or potato dextrose agar (PDA). Colonies were identified based on colony morphology and characteristics of somatic and reproductive structures using taxonomic keys described by Watanabe (2002) and other mycological resources (Schroers et al., 1999). The total number of seeds, and pieces of hull, root, and stem plated per field was 240 (60 per tissue type per field), and was 2,400 across all 10 fields surveyed in 2005, and 960 across all four fields surveyed in 2006. The percentage of isolation (frequency) of each microorganism was computed as the number of seeds and pieces of tissue from which each microorganism was recovered divided by the total number of seeds or pieces of tissue plated (60 per tissue type per field).
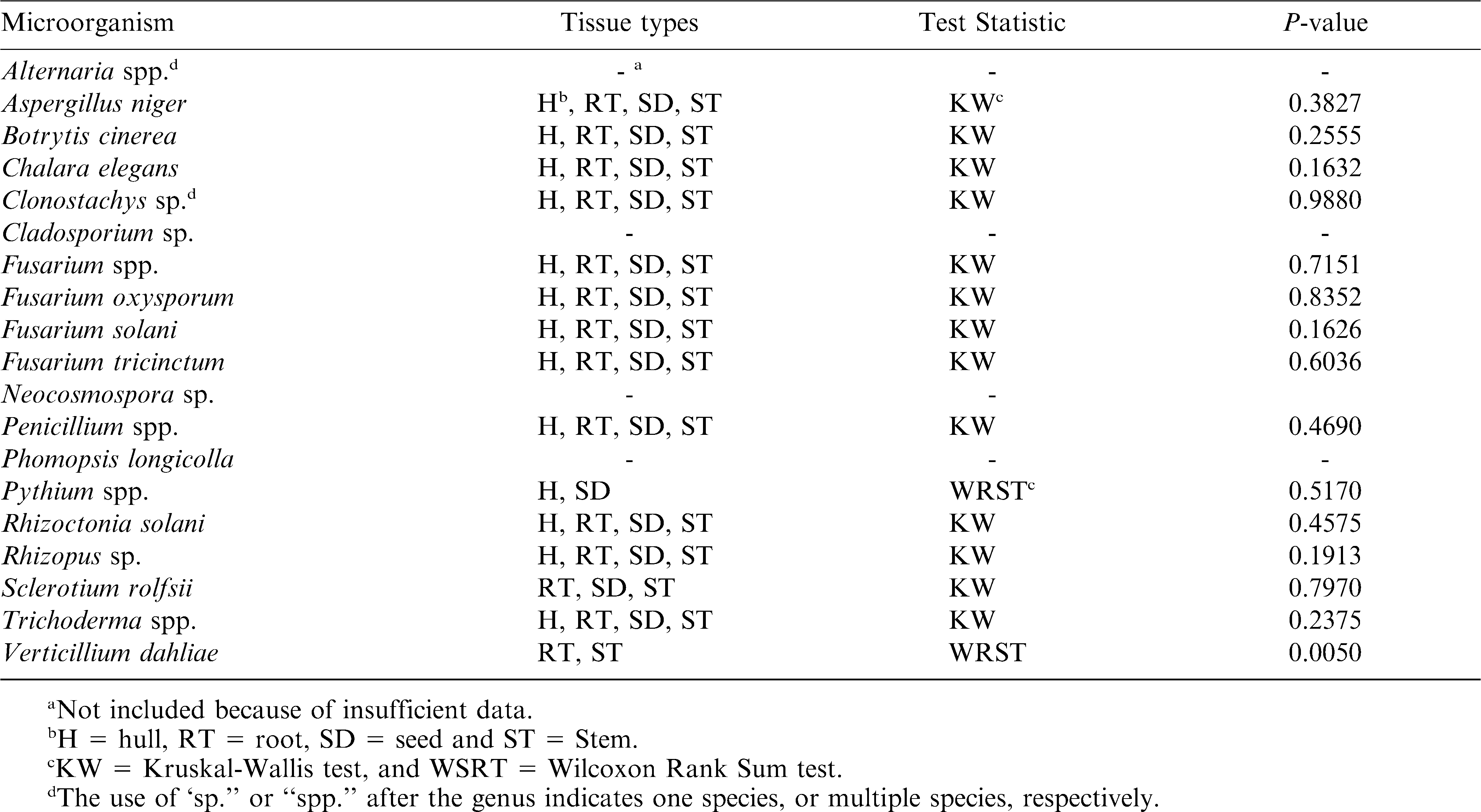
Within each year of the survey, the types of microorganisms recovered were generally identical with similar range in frequency of isolation among fields. Therefore, an average frequency of isolation of each microorganism was calculated using pooled data across both years for each tissue using SAS software (SAS Institute, Cary, NC). For each microorganism, frequency of isolation was compared across tissue types by the non-parametric Kruskal-Wallis test using the NPAR1WAY procedure in SAS. This test was used because the frequency of isolation data did not meet the normality requirement for parametric analysis. Only data for microorganisms isolated in at least four fields and at least three tissue types were included in the analysis. Data based on only two tissue types were analyzed using Wilcoxon rank sum test. Spearman rank correlation analysis was conducted to determine the association between frequencies of isolation from different tissue types for microorganisms isolated from each field.
Pathogenicity of isolates
Pathogenicity of microorganisms from genera with known pathogenic species was evaluated on peanut grown in styrofoam cups and test tubes as described below. The two systems were designed to test for consistency of pathogenicity under differential conditions of soil volume, inoculum amount, and seeding stage. Two to five isolates of each of the following microorganisms were tested: R. solani, F. oxysporum, F. tricinctum, V. dahliae, S. rolfsii, Pythium spp., Botrytis cinerea, and A. niger. These microorganisms were included in the pathogenicity as representatives of a broad range of pathogens known to cause pod, root, and stem diseases of peanut. Each microorganism was grown on PDA in 9-cm diameter Petri dish until the mycelium reached the edge of the dish. Then, the content of each dish was mixed in a blender with 100 mL sterile water, and the slurry was used as inoculum (Franke et al., 1999).
Seeds of cultivar Valencia-C, which is the most widely grown Valencia cultivar in New Mexico, were surface sterilized as described by Abd El-Rehim et al. (1981). Briefly, seeds were wrapped in one-layer cheesecloth and immersed for 20 min in calcium hydroxide solution (1.58 g/L) heated to 50 C. Treated seeds were placed on two layers of moistened sterile filter paper (Whatman #1) in 9-cm diameter Petri dishes, and incubated at 25 C for 48 hr. Seeds displaying no mycelial growth were placed in styrofoam cups or test tubes. Seeds were placed at a depth of 2 cm in styrofoam cups (with a diameter of 9.2 cm on top and 6 cm on bottom, and a depth of 13.5 cm) filled with sterilized Terra-Lite Metro Mix 360. All cups were maintained in a growth chamber set at 26 C under a 14-hr photoperiod. Plants at the 2nd to 3rd leaf-stage were inoculated by adding 50 ml of mycelium slurry to each cup in a hole (approximately 1-cm width × 2-cm depth) around the base of each seedling. The slurry was overlaid with sterilized fine vermiculite to a thickness of 5 mm. A clear plastic cup (with a diameter of 10.5 cm on top and 7.5 cm on bottom, and a depth of 17.5 cm) was placed over the inoculated plant, and secured to the rim of the styrofoam cup, to generate a high humidity environment. In each of two trials, 8 plants were inoculated per isolate, and 3 non-inoculated plants served as controls in each trial. All plants were maintained in a growth chamber as described above.
Evaluation of pathogenicity in test tubes was conducted using procedures described by Bell (1967). Approximately 20 mL of vermiculite was dispensed into glass test tubes (15-cm height × 2-cm diameter) and moistened with 10 mL water. The test tubes were plugged with cotton and autoclaved twice over two consecutive days. Seeds of peanut (cultivar Valencia-C), surface sterilized as previously described, were placed in each tube at a depth of 2 cm and the tubes were maintained in a growth chamber under conditions described above. Seedlings were inoculated when cotyledons were opened by adding 3 mL of mycelium slurry to each test tube around the base of each seedling. In each of two trials, 8 plants were inoculated per isolate of microorganism, and 3 non-inoculated plants served as controls in each trial.
All plants were monitored for symptom development for a period of 4 to 7 wk. Re-isolation of pathogens from symptomatic plants was completed using 2-cm segments cut from the lower stem portion beginning at the crown. Similarly, 2-cm segments were cut from the upper portion of tap root starting at the crown. Segments of stem and root were washed free of soil, submerged separately in 0.5% sodium hypochlorite for 3 min, rinsed in sterile distilled water, and blot dried on sterile paper towels. For each stem segment or root segment, 0.5 cm of tissue was trimmed at both cut ends of each segment. The remaining 1-cm segment was cut into four pieces and plated on APDA.
A third pathogenicity test was conducted on peanut seeds of Valencia-C, using two isolates (one from seed and one from root) of R. solani, S. rolfsii, and Pythium spp., which are species reported as typically associated with pod rot. A 1-cm diameter mycelium plug of each isolate was placed centrally on PDA medium contained in deep Petri dishes (10-cm diameter × 2-cm depth). Four seeds of peanut cultivar Valencia-C, surfaced sterilized as previously described, were placed around each mycelium plug. Seeds serving as control treatment were placed on PDA medium with no mycelium plug added. For each isolate and control treatment, 10 deep Petri dishes were prepared and carefully sealed with parafilm so as to prevent movement of seeds from their placement position on PDA medium surface. All Petri dishes were incubated at 25 °C in an incubator under a 14-hr photoperiod. Isolates were pathogenic if the seeds were totally colonized by an isolate and did not sprout or sprouted but were colonized and necrotized.
Results
Field surveys and isolation of microorganisms
In 2005 and 2006, 14 fields were surveyed and 15 fifteen plants were collected in each field. The number of mature pods harvested from each plant varied from 32 to 94 across both years. The average incidence of pods with hull discoloration or pod rot symptoms varied from 55 to over 90%, and disease severity (area of individual pods covered with symptoms) ranged from 5 to over 90% across all fields surveyed. Hull discoloration and pod rot symptoms were as follows: a) small black speckles on the hull, b) light tan to dark brown discoloration or dry decay on hull with or without the presence of indentations on the hull, c) dark brown dry decay on hull with or without visible mycelium on decayed areas, and d) dark brown to black water-soaked or greasy-like decayed areas on hull. Based on the literature on hull discoloration and pod rot (Kokalis-Burelle et al., 1997), these four categories of symptoms were determined as symptoms of blackhull, Rhizoctonia pod rot, Sclerotium pod rot, and Pythium pod rot, respectively. More than 60% of symptomatic pods had symptoms of blackhull and/or Rhizoctonia pod rot.
The two predominant microorganisms were Rhizoctonia solani and Fusarium oxysporum, which were recovered from all fields with an average frequency of isolation greater than 15% across all tissue types (Table 1); all other microorganisms were recovered from variable number of fields from 1 to 13 out of 14 fields, with an average frequency less than 13% across all tissue types. For each microorganism isolated from at least three fields and at least three tissue types, there was no difference among average frequencies of recovery across tissue types according to Kruskal-Wallis (P > 0.05; Table 2). Among microorganisms recovered from at least three fields and two tissue types, the only difference was recorded between average frequencies of isolation of V. dahliae from root and stem tissues (P = 0.005; Table 2).
Microorganisms isolated from seed (Table 1) at an average frequency greater than 5% included R. solani (18.5%), Fusarium oxysporum (17.0%), Rhizopus sp. (12.0%), F. tricinctum (9.7%), and F. solani (9.5%), Chalara elegans (7.4%), Clonostachys sp. (7.6%), Sclerotium rolfsii (7.2%), and Pythium spp. (5.7%). From hull tissue (Table 1), microorganisms recovered at average frequency greater than 5% were R. solani (21.8%), F. oxysporum (17.4%), Rhizopus sp. (9.6%), F. tricinctum (11.5%), F. solani (8.9%), Chalara elegans (7.4%), Clonostachys sp. (7.0%), Pythium sp. (6.1%), and S. rolfsii (5.5%).
Similar to the scenario observed with pod tissues (hull and seed), several microorganisms were isolated from root and stem tissues (Table 1). Microorganisms recovered from root tissues at an average frequency greater than 5% included R. solani (24.0%), F. oxysporum (19.5%), Rhizopus sp. (13.3%), S. rolfsii (11.0%), F. tricinctum (9.0%), Phomopsis longicolla (8.3%), Clonostachys sp. (7.1%), B. cinerea (6.2%), and Chalara elegans (5.3%). From stem tissues, microorganisms recovered at an average frequency greater than 5% were R. solani (20.8%), F. oxysporum (17.0%), V. dahliae (11.7%), F. tricinctum (10.8%), S. rolfsii (9.2%), Rhizopus sp. (8.3%), Phomopsis longicolla (6.7%), F. solani (6.6%), and Clonostachys sp. (6.4%). The percentage of plant tissue with multiple genera or species of microorganisms, and unidentified microorganisms ranged from 4.8 to 6.5% across all tissue types and are shown under the “Others” category in Table 1.
Among microorganisms reported as typically associated with pod rot (R. solani, S. rolfsii, and Pythium spp.), R. solani was isolated at the highest frequency in all fields (18.5 to 24.0%). In contrast, S. rolfsii and Pythium spp. were recovered at low frequencies of 1.7 to 6.1%, and 7.2 to 11%, respectively. Rhizoctonia solani was found in all fields with an average frequency of isolation of 24.0, 18.5, 21.8, and 20.8%, from root, seed, hull, and stem tissues, respectively. There was no difference among the frequencies of isolation of R. solani across tissue types using Kruskal-Wallis test (P = 0.4575; Table 2).
Pythium spp. were recovered at an average frequency of 5.7% from seed, 6.1% from hull, and 1.7% from stem and root tissues. However, Pythium spp. were recovered in more fields from seed and hull tissues than from root and stem tissues. Recovery of Pythium spp. was not different between seed and hull tissues using Wilcoxon rank sum test (P = 0.517; Table 2).
Sclerotium rolfsii was isolated from all tissue types in variable number of fields from 3 to 6, with an average frequency of 11.0% for root, 9.2% for stem, 7.2% for seed, and 5.5% for hull tissues. There was no statistically significant difference in frequency of isolation among tissue types using Kruskal-Wallis test (P = 0.7967; Table 2).
Spearman rank correlation analyses were conducted only for R. solani, F. oxysporum, and F. tricinctum. Data for other microorganisms were insufficient to be amenable to correlation analyses. For R. solani, correlation analysis revealed a significant association between average frequency of isolation from stem and root (r = 0.73, P = 0.004), and stem and hull (r = 0.67, P = 0.010). With respect to F. oxysporum, there was a statistically significant association between average frequency of isolation from stem and root (r = 0.71, P = 0.006), stem and seed (r = 0.72, P = 0.005), stem and hull (r = 0.70, P = 0.006), root and seed (r = 0.61, P = 0.024), root and hull (r = 0.77, P = 0.002), and seed and hull (r = 0.57, P = 0.037). Correlation analysis for F. tricinctum indicated a statistically significant association between average frequency of isolation from stem and root (r = 0.62, P = 0.032).
Pathogenicity of microorganisms on peanut
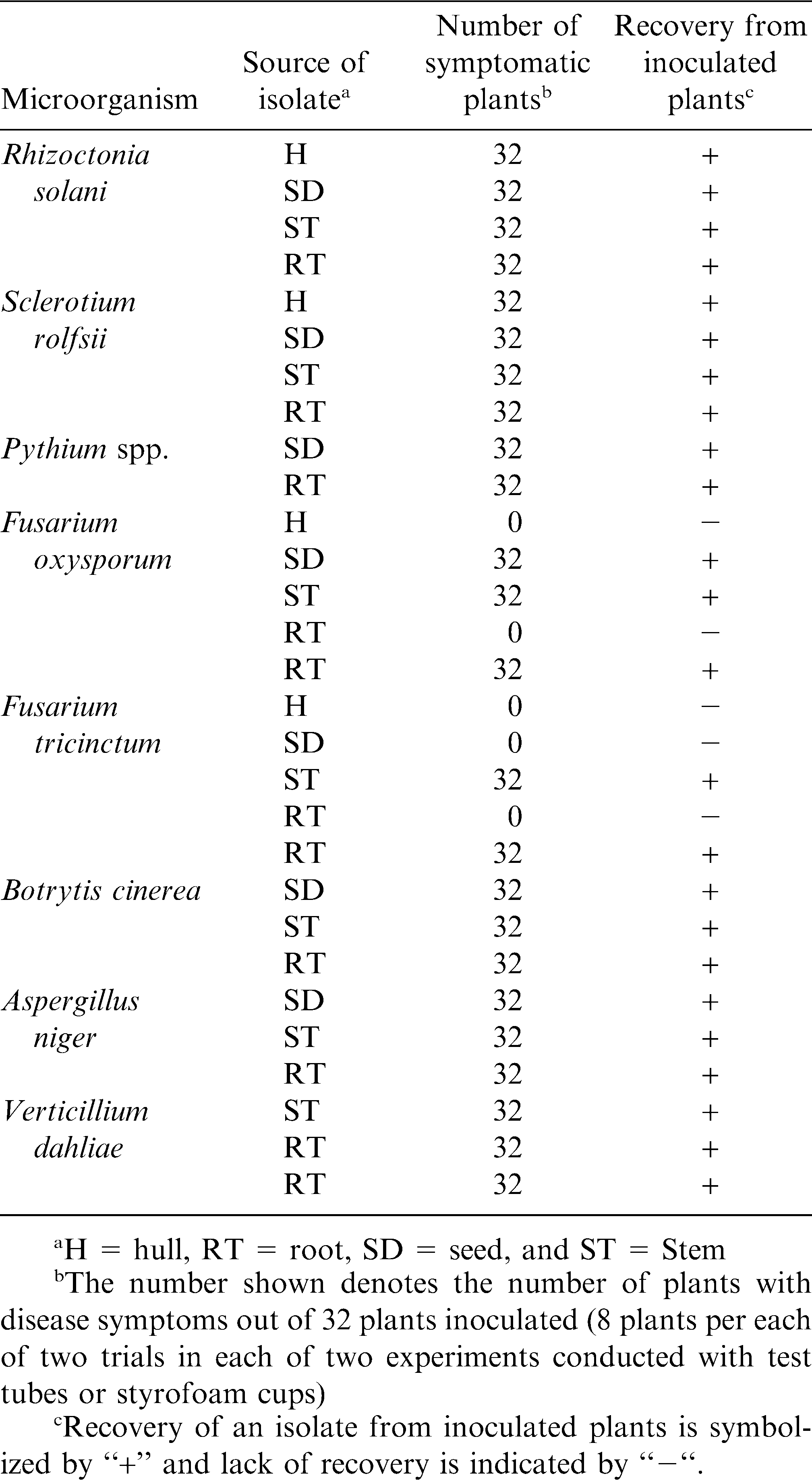
Pathogenicity of selected microorganisms on peanut was similar under both the styrofoam cup and test tube systems. All isolates of microorganisms tested by inoculation of peanut were pathogenic (Table 3), except for F. oxysporum and F. tricinctum. Five isolates of F. oxysporum were tested and only three were pathogenic. Fusarium tricinctum yielded two pathogenic isolates from a pool of five isolates tested. Symptoms expressed on inoculated plants included root and stem necrosis, leaf yellowing, marginal leaf necrosis, plant wilting, root rot, and plant death (Table 4). Of the five isolates of F. oxysporum tested for pathogenicity, three were pathogenic causing stem necrosis and wilt of seedlings. Similarly, of the five isolates of F. tricinctum tested for pathogenicity, two were pathogenic causing stem and root necrosis. All microorganisms were re-isolated from symptomatic plants (Table 3). None of the non-inoculated plants displayed any symptoms.
All isolates of R. solani, S. rolfsii, and Pythium spp. tested were pathogenic and completely colonized seeds causing necrosis of radicles and seed death (Fig. 1). Seeds serving as controls produced vigorous radicles with no necrosis. All isolates of R. solani, S. rolfsii, and Pythium spp. were recovered from colonized seeds. Plating of control seeds yielded no growth of R. solani, S. rolfsii, and Pythium spp.
Seeds of peanut cultivar Valencia-C non-inoculated (CTL) and inoculated with Rhizoctonia solani (RS), Sclerotium rolfsii (SR), and Pythium sp. (PY). Notice complete colonization of seeds by each microorganism and failure of colonized seeds to establish any vigorous radicle compared with control seeds.
Discussion
Peanut pod rot is known to occur in New Mexico; however, there has not been a systematic study on the causal microorganisms associated with pod rot. This study is the first to provide a synopsis into the composition and predominance of mycelial microorganism populations associated with pods and other plant part tissues of Valencia peanut affected by pod rot in New Mexico. The methodological approach used in this work was similar to that adopted in other studies (Hollowell et al., 1998; Osuinde and Daibo, 1999; Wheeler et al., 2005), with the exception that sodium hypochlorite was used in this survey, which could have greatly reduced the frequency of isolation of microorganisms, such as species of Pythium (Davison and McKay, 2003; Denman et al., 1993; Stanghellini and Kronland, 1985). Rhizoctonia solani was shown to be the predominant microorganism associated with Valencia peanut displaying pod rot. This study provides new knowledge on assemblage of similar microorganisms on tissue of different types, and correlation analysis revealed strong association among tissue types with regard to R. solani.
Although results from this study were consistent in both years, it must be pointed out that the predominance of microorganisms may be influenced by environmental conditions and field-specific crop management practices. This is illustrated in the work Hollowell et al. (1998) who showed that Rhizoctonia and Pythium species were the most frequently isolated microorganisms from peanut in North Carolina. In two of the three years during which the surveys were conducted, Rhizoctonia spp. predominated, whereas in one of the three years Pythium spp. were the predominant microorganisms. Specific crop management information (rotation, fertilization, and pesticide application) was not collected for each field during this study. Given the consistency of the results in this study, it is reasonable to state that such factors were of little or no influence on the observed frequency of isolation.
Frequency of isolation of microorganisms from plant tissues may be influenced by several factors including plant age. In this study, plant samples were collected two to three weeks before harvest. This period was selected because the frequency of isolation of microorganisms such as Pythium spp. has been shown to increase with pod age (Soufi and Filonow, 1992). The study was conducted over two years using different sets of fields in each year to enable plant sampling and to ascertain consistency in frequency of isolation of microorganisms under different environmental conditions. Across both years, R. solani was clearly shown to be the most predominant microorganism isolated from peanut tissues in this study.
The number of fields surveyed was reflective of the small acreage of Valencia peanut production in New Mexico in contrast to the large production acreage of major peanut market types in other states. The uniqueness of this study is its comprehensive approach in assessing microorganisms associated with Valencia peanut. In many other surveys, work was limited to only peanut pods (Hollowell et al., 1998; Wheeler et al., 2005). However, this study examined not only pods, but also roots and stems. The benefit of this approach is that it enables an assessment of the variability in frequency of isolation along tissue type, and allows an inference to be made on the preferential niche or tissue type for each microorganism. Whereas R. solani was isolated at comparable levels across all tissue types, Pythium spp. were mostly isolated from pods. The presence of R. solani in all tissue types could indicate that this microorganism may inflict damages to root, stem, and pods if conditions in the environment are favorable. Recovery of R. solani and other microorganisms from stem and root of non-symptomatic plants signify a subclinical infection which could be visibly manifested under favorable environmental conditions.
Although studies similar to this work have been conducted (Hollowell et al., 1998; Wheeler et al., 2005), no pathogenicity has been carried out. In this study, three pathogenicity tests were conducted using seedlings and seeds. The microorganisms included in the pathogenicity test on seedlings were selected as representatives of a broad range of pathogens known to cause pod, root, and stem diseases of peanut. Seedlings have been shown as appropriate materials for testing microorganisms isolated from various peanut plant parts including pods (Bell, 1967; Bell and Minton, 1973). The two pathogenicity tests on seedlings were used to ascertain consistency of plant response to selected microorganisms under varying conditions of plant age and size (plant inoculated at cotyledon stage or at 2nd to 3rd leaf-stage). All isolates of R. solani, S. rolfsii, and Pythium spp., associated with pod rot symptoms, were pathogenic on peanut seedlings. Furthermore, these microorganisms were demonstrated to be pathogenic on seeds as gauged by the extent of radicle necrosis, seed colonization, and seed death. Severe pod rot may be accompanied by infection of seeds by pathogens (Kokalis-Burelle et al., 1997), therefore, pathogenicity tests using seeds provides a realistic representation of the ability of pathogens to infect pod tissues including seeds.
Characterization of microorganisms, such as R. solani and S. rolfsii, may include determination of mycelial compatibility among isolates to establish population genetic structure (Cubeta and Vilgalys. 1997; Punja and Sun, 2001). This determination was deemed unnecessary in this study, because the main interest was to assess the pathogenicity of isolates, which provides an important gauge for potential impact of the microorganisms on peanut production.
A diverse range of microorganisms were recovered from pods in this work, and such diversity was also reported in the survey study conducted by Hollowell et al. (1998). However, in this work, microorganisms isolated from pod tissue represented more than 10 fungal and Oomycete genera, whereas, the study by Hollowell et al. (1998) microorganisms isolated from pod tissue represented only five genera, Rhizoctonia, Pythium, Cylindrocladium, Sclerotiorum, and Sclerotinia. This difference could be ascribed to various factors, such as cropping practices and environmental conditions. It should be noted that species of microorganisms in the genera Rhizoctonia, Sclerotium, and Pythium, which are typically reported as associated with pod rot, were encountered in this study as well as in the work by Hollowell et al. (1998).
Besides R. solani, Pythium spp., and S. rolfsii, which are typically involved in pod rot, many of the microorganisms isolated in this study are known to cause various diseases in peanut in New Mexico and in other peanut growing regions within and outside the U.S. (Kokalis-Burelle et al., 1997) such as blackhull caused by C. elegans (Liddell, 1997), Botrytis blight caused by B. cinerea ( Porter, 1997), seedling diseases and wilt caused by species of Fusarium, Pythium, Rhizoctonia, and Rhizopus (Bell and Minton, 1973; Brenneman, 1997; Hollowell et al., 1998; Osuinde and Daibo, 1999), Verticillium wilt caused by V. dahliae (Melouk et al., 1983; Smith, 1960), Neocosmospora foot rot caused by Neocosmospora sp. ( Porter, 1997), Aspergillus crown rot caused by A. niger (Melouk and Damicone, 1997), Phomopsis stem blight caused by P. longicolla (Sanogo and Etarock, 2009), stem canker caused by F. oxysporum (Mullen et al., 1996), Rhizoctonia limb rot by R. solani (Barnes et al., 1990; Brenneman, 1997; Franke et al., 1999), and southern stem blight caused by S. rolfsii (Backman and Brenneman, 1997). Other isolated microorganisms, such as Trichoderma spp. and Clonostachys rosea, belong to fungal groups known for their antagonistic activity against several plant pathogens (Papavizas, 1985; Schroers et al., 1999).
Surveys such as this are inherently confronted with several logistical issues, two of which are the choice of media for isolation of microorganisms from plant tissues and the use of surface sterilization of plant tissues prior to isolation. These issues were recognized long ago by Garren (1964) who showed that isolation procedures may influence the spectra of terrestrial microflora from peanut pods. Also, recent work on isolation of Pythium from other plant hosts such as carrot (Daucus carota L.) and lucerne (Medicago sativa L.) has addressed these issues (Davison and McKay, 2003; Denman et al., 1993). Selective or semi-selective and non-selective have all been employed across studies. The fundamental problem lies in the level of resource needed for isolation from plant tissue, and exclusion or underestimation of microorganisms. In studies focused on specific microorganisms, selective or semi-selective media have proven useful in isolating targeted microorganisms while minimizing the isolation of undesired microorganisms. Such studies could become prohibitively unmanageable in terms of time and financial expenditures if several microorganisms are being targeted. Non-selective media enable a wide array of microorganisms to be isolated; however, these media may provide a competitive edge to fast-growing microorganisms. As a result, isolation frequency data could exclude or underestimate the level of slow-growing microorganisms. In this study, acidified potato dextrose agar, a non-selective medium, was used because it allows a rapid visual assessment of types of mycelial growth and color.
The centrality of this work was the use of sodium hypochlorite for surface sterilization of tissues prior to plating onto growth media. Surface sterilization of plant tissue is a widely used phytopathological procedure to ensure aseptic isolation of targeted causal agents from infected plant tissues. However, this procedure may affect the isolation of some microorganisms highly sensitive to sodium hypochlorite including species of Pythium (Davison and McKay, 2003; Denman et al., 1993; Stanghellini and Kronland, 1985). Despite the possible harsh treatment with sodium hypochlorite, Pythium spp. were recovered from pod tissues, but was not eliminated in this study. Although it is plausible that the low level of isolation of Pythium spp. was due, in part, to surface sterilization with sodium hypochlorite, it is important to note that this low level of isolation is reflective of the low incidence of pods with Pythium-like symptoms. Indeed, as previously indicated, more than 60% of symptomatic pods had symptoms of blackhull and Rhizoctonia pod rot symptoms. Besler et al. (2003), working on Rhizoctonia pod rot, used a 10% mixture of sodium hypoclorite for pod surface sterilization for an unspecified duration. Similarly, Garren (1964) working on rotted and sound peanut pods, dipped pieces of peanut pods in 70% ethyl alcohol followed by soaking in 1% sodium hypochlorite for 2 min. Likewise, a protocol using sodium hypochlorite was implemented in this study.
Summary and Conclusions
Information on frequency of isolation and predominance of microorganisms could be used to design a management strategy aimed at reducing populations of predominant microorganisms. This study provides important information from at least two perspectives. First, it will be useful in breeding effort, because it will allow off-season screening to focus more on major microorganisms. Second, other management practices could be primarily directed at reducing the impact of the identified predominant pathogens. Based on the results from this study, pod rot management in New Mexico should be focused on R. solani since it has been shown to be the most predominantly isolated mycelial microorganism from peanut affected by pod rot. A two-pronged management may be established through the utilization of (a) resistant Valencia cultivars, which may be identified through off-season screening of Valencia germplasm for resistance to R. solani, and (b) control methods such as application of fungicides efficacious against R. solani.
Acknowledgements
The authors thank Carmen Sanogo, Johnny Maruthavanan, and Fekede Workneh for reviewing this manuscript. This research was funded in part by New Mexico Agricultural Experiment Station, New Mexico Peanut Research Board, and National Peanut Board.
Literature Cited
Abd El-Rehim M.A Backman P.A Rodriguez-Kabana R and Crawford M.A 1981 Peanut seed treatment with hot calcium hydroxide solutions Peanut Sci. 8 : 36 – 39 .
Anonymous 2011 Peanut crop estimate for 2011, Alabama peanut producers association http://www.alpeanuts.com/news/news/273-2011-peanut-crop-estimate.html. 18 October 2009 .
AOSCA, Association of Official Seed Certifying Agencies 2009 http://aosca.org/ index.htm.
Backman P.A and Brenneman T.B 1997 Stem rot, pp. 36 – 37 In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Barnes J.S Csinos A.S and Branch W.D 1990 Sensitivity of Rhizoctonia solani isolates to fungicides and evaluation of peanut cultivars to Rhizoctonia limb rot Peanut Sci. 18 : 62 – 65 .
Bell D.K 1967 Pathogenicity of fungi to peanut seedlings in known fungal culture at four temperatures Oléagineux 6 : 373 – 375 .
Bell D.K and Minton N.A 1973 Postemergence damping-off of peanut plants caused by Pythium myriotylum Phytopathology 63 : 1544 – 1545 .
Besler B.A Grichar W.J Brewer K.D and Baring M.R 2003 Assessment of six peanut cultivars for control of Rhizoctonia pod rot when sprayed with azoxystrobin or tebuconazole Peanut Sci. 30 : 49 – 52 .
Brenneman T.B 1997 Rhizoctonia diseases, pp. 30 – 31 In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Cubeta M.A and Vilgalys R 1997 Population biology of the Rhizoctonia solani complex Phytopathology 87 : 480 – 484 .
Davison E.M and McKay A.G 2003 Host range of Pythium sulcatum and the effects of rotation on Pythium diseases of carrots Australas. Plant Path. 32 : 339 – 346 .
Denman S Knox-Davies P.S and Calitz F.J 1993 Evaluation of some media and methods for isolating Pythium species from Lucerne seedlings Phytophylactica 25 : 149 – 156 .
Franke M.D Brenneman T.B and Holbrook C.C 1999 Identification of resistance to Rhizoctonia limb rot in a core collection of peanut germplasm Plant Dis. 83 : 944 – 948 .
Garren K.H 1964 Isolation procedures influence the apparent make-up of the terrestrial microflora of peanut pods Plant Dis. Rep. 48 : 344 – 348 .
Hollowell J.E Shew B.B Beute M.K and Abad Z.G 1998 Occurrence of pod rot pathogens in peanuts grown in North Carolina Plant Dis. 82 : 1345 – 1349 .
Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) 1997 Compendium of Peanut Diseases, 2nd Edition APS Press .
Liddell C.M 1997 Black hull, pp. 8 – 10 In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Melouk H.A and Damicome J.P 1997 Aspergillus crown rot, pp. 7 – 8 In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Melouk H.A Wadsworth D.F and Sherwood J.L 1983 Effect of Verticillium wilt on root and top weight of peanut cultivar Tamnut 74 Plant Dis. 67 : 1349 – 1350 .
Mullen J.M Hagan A.K and Nelson P.E 1996 A new stem canker of peanut in Alabama caused by Fusarium oxysporum: a wound-dependent disease Plant Dis. 80 : 1301 .
NMDA 2010 New Mexico Agricultural Statistics, Peanuts: acreage, yield, production, price and value. 52 pp.
Osuinde M.I and Daibo O.O 1999 Wilt disease of Arachis hypogaea L. in Ekpoma, Edo State, Nigeria Austr. J. of Exp. Agr. 39 : 39 – 42 .
Papavizas G.C 1985 Trichoderma and Gliocladium: biology, ecology, and potential for biocontrol Ann. Rev. Phytopathol. 23 : 23 – 54 .
Porter D.M 1997a Botrytis blight, pp. 10 – 11 In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Porter D.M 1997b Neocosmospora foot rot, 23 pp. In Kokalis-Burelle N Porter D.M Rodriguez-Kabana R Smith D.H and Subrahmanyam P (eds.) Compendium of Peanut Diseases, 2nd Edition APS Press , St. Paul, MN .
Punja Z.K and Sun Li-J 2001 Genetic diversity among mycelial compatibility groups of Sclerotium rolfsii (teleomorph Athelia rolfsii) and S. delphinii Mycol. Res. 105 : 537 – 546 .
Sanogo S and Etarock F.B 2009 First Report of Phomopsis longicolla causing stem blight of Valencia peanut in New Mexico Plant Dis. 93 : 965 .
Schroers H-J Samuels G. J Seifert K. A and Gams W 1999 Classification of the mycoparasite Gliocladium roseum in Clonostachys as C. rosea, its relationship to Bionectria ochroleuca, and notes on other Gliocladium-like fungi Mycologia 91 : 365 – 385 .
Smith T.E 1960 Occurrence of Verticillium wilt on peanuts Plant Dis. Rep. 44 : 6 .
Soufi R.K and Filonow A.B 1992 Population dynamics of Pythium spp. in soil planted with peanut Plant Dis. 76 : 1203 – 1209 .
Stanghellini M.E and Kronland W.C 1985 Detrimental effect of surface sterilization on isolation of Pythium spp. from feeder roots (Abstr.) Phytopathology 75 : 1334 .
Watanabe T 2002 Pictorial atlas of soil and seed fungi: morphologies of cultured fungi and key to species. 2nd edition CRC Press , Boca Raton, FL .
Wheeler T.A Howell C.R and Porter D 2005 Pythium species associated with pod rot on West Texas peanuts and in vitro sensitivity of isolates to mefenoxam and azoxystrobin Peanut Sci. 32 : 9 – 13 .
Notes
- Department of Entomology, Plant Pathology, and Weed Science, New Mexico State University, Las Cruces, NM 88003
- Agricultural Science Center at Clovis, New Mexico State University, Clovis, NM 88101 *Corresponding author: ssanogo@nmsu.edu
Author Affiliations