Introduction
Because of inherent low moisture content, low water activity (Aw) and non-animal origins, roasted peanuts, peanut butter and other nuts were considered low risk for Salmonella spp. contamination (Sheth et al., 2011). However, infective doses of Salmonella have been reported in fatty, low-moisture foods such as chocolate and cheese (Gill et al., 1983; D'Aoust, 1985). The first reported outbreak of Salmonella in peanut butter occurred in Australia in 1996 (Scheil et al., 1998). The source of the implicated Salmonella Mbandaka was traced back to a peanut roasting factory. Other outbreaks were associated with peanut-containing snacks in Israel and North American (Killalea et al., 1996) and Asian-style peanuts still in their shell (Kirk et al., 2004). From 2006 to 2007, Salmonella Tennessee in peanut butter was responsible for a major food-borne disease outbreak in the United States (Deng et al., 2013; MMWR, 2007). An epidemiological study identified 715 cases from 48 states, resulting in 93 hospitalizations (Sheth et al., 2011). In September 2012, Sunland Inc. of Portales, NM, announced a voluntary recall of almond and peanut butter due to a rare outbreak of Salmonella Bredeney associated with the products (MMWR, 2013). These reoccurring outbreaks of Salmonella from peanut products in recent years have caused processors to install new equipment, make roof repairs, sanitize facilities, and ensure separation of raw and finished product and hastened implementation of new Hazard Analysis and Critical Control Points plans to assure the safety of peanuts (Sheth et al., 2011).
New research indicates that Salmonella spp. are able to persist in foods under a variety of conditions. Burnett et al. (2000) found that Salmonella could survive for at least 24 wk in low-moisture foods such as peanut butter. It was able to habituate in high-fat, low water activity environments and survive high temperatures (Mattick et al., 2000). Aviles et al. (2013) reported that exposures to high fat content and low Aw in peanut butter caused a protective effect with increased survival of Salmonella Tennessee in gastric models. Stressed Salmonella enterica were found to have significantly more heat resistance than naive bacteria (He et al., 2011) and outbreak strains were shown to be more thermo-tolerant than other strains tested (Ma et al., 2009). Heat resistance can be influenced by many factors including previous growth and storage conditions with lower moisture levels generally producing greater heat resistance (Podolak et al., 2010). Likewise, increased Aw can reduce thermal resistance (He et al., 2013). Addition of 7% moisture to cocoa and hazelnut shells significantly reduced D values of two Salmonella strains (Izurieta and Komitopoulou, 2012). Corn syrup, soy protein, vitamin and mineral supplements in peanut butter formulations may increase the viability of Salmonella during the first weeks of storage (Burnett et al., 2000). The survival and heat resistance of S. enterica was significantly affected by peanut butter formulation and storage temperature. Due to its persistence, Salmonella contamination is especially troubling when it survives in products such as peanut butter that have a long shelf life (Sobel et al., 2001).
Samples of raw, shelled peanuts from 2008-2010 were examined for Salmonella contamination (Calhoun et al., 2013). Out of 944 samples, 2.33% were positive for Salmonella using the VIDAS® assay, an automated enzyme-linked fluorescent immunoassay (Crowley et al., 2011). Using a two-step enrichment, the VIDAS assay was deemed comparable to older, culture methods (Crowley et al., 2011). Another similar method using enrichment plus a lateral flow immunoassay, the Reveal Salmonella 2.0 test provided accurate results regardless of initial sample volume (Hoerner et al., 2011). Quantitative polymerase chain reaction (qPCR) kits have been evaluated (Balachandran et al., 2011; Li et al., 2012) and performed well against Bacteriological Analytical Manual (BAM 1998) methods. Other researchers have detected the Salmonella invA gene using isothermal methods, which require only a water bath and could be used on-site or in the field (Kim et al., 2011). While all these molecular detection methods are rapid, they fail to quantitate Salmonella cells and therefore have limited use in thermal process validation.
The destruction and fragmentation of mtDNA from high- and low-acid foods was highly correlated to surrogate spore destruction, cumulative F-values and time-temperature treatments in earlier studies (Caldwell et al., 2015a; Caldwell et al., 2015b). The goal with roasting peanuts was to evaluate mtDNA fragmentation as a time-temperature integrator (TTI) in a dry thermal process as opposed to previous “wet” processes. To this end, the effectiveness of mtDNA fragmentation, as determined by threshold cycle (Ct) value in qPCR, was examined using a solid, high-fat, low moisture and low Aw product. Also, to assess its usefulness as a quality parameter, intrinsic mtDNA fragmentation was compared to Hunter L color value, a quality indicator in roasted nuts. The Almond Board of California has recommended the use of Enterococcus faecium NRRL B-2354 as a surrogate for S. enterica in validation of dry roasting processes in almonds (ABC, October 2007). In recent studies, E. faecium was validated in oil and dry roasting of peanuts at various temperatures and found to have similar kinetics but proved more resistant to heat processing than Salmonella spp. (Sanders and Calhoun, 2014). Since these researchers provided documentation for the use of this microorganism as an acceptable surrogate for Salmonella in peanut thermal processes, fragmentation of intrinsic peanut mtDNA was compared to reduction curves of E. faecium . Finally, individual peanuts were roasted and dissected to determine the “cold spot” in processing using this non-obtrusive molecular method. Thermal fragmentation of ubiquitous and intrinsic mtDNA was evaluated as both a rapid measure of safety and quality in roasted peanuts.
Materials and Methods
Inoculation
E. faecium (ATCC 8459) was inoculated into BHI broth (Remel, Lenexa, KS) from freshly plated colonies and incubated statically overnight at 35 °C. Cultures were concentrated 2X by centrifugation (5810R, Eppendorf, Hamburg, Germany) at 6000 rpm for 10 min at 4 C and re-suspended in 0.5 X initial volume with sterile 0.9 % saline. Initial culture concentration was determined by A600 and a simplified agar plate technique (Jett et al., 1997) utilizing square Petri plates and the track-dilution method. For each temperature assay, Enterococcus-inoculated saline (110 ml) was added to the total weight of peanuts (2200 g) to be tested in a large plastic zipper bag, diluting the culture 1:20. The bag was closed and secured. Contents were mixed thoroughly, and then allowed to sit for 5 min to absorb the liquid. Target concentration was 108 CFU/g for inoculated peanuts. Inoculated peanuts were poured onto wire racks in 100 g aliquots and allowed to air dry for 20 min.
Roasting
A convection oven (Despatch Model LXD1-42-2; Despatch Industries; Minneapolis, MN) was set at 138, 153 or 167 C and allowed to equilibrate for 30 min. Three replicate samples of 100 g each were taken from the total 2200 g inoculated peanut test set for each of the following time points: 0, 6, 9, 12, 15, 18, and 21 min for each temperature. A metal rack was inserted in the oven and brought to temperature. This rack was designed to hold smaller racks made of hardware cloth through which peanuts would not pass. Each batch consisted of 100 g lots laid out on the small hardware cloth racks. Once the smaller racks are inserted into the larger rack, the peanuts were essentially suspended in the moving air inside the oven. For each roasting batch, the oven door was quickly opened, the tray with the peanuts slid into the large rack and the door of the oven quickly closed. There was a drop in the oven temperature due to opening the door. The lowest temperature reached and the number of seconds required for the oven to return to set point was recorded for each batch. At the appropriate time point, the oven was opened quickly and the peanuts removed in the small rack. This rack was placed over a box fan with sufficient flow to cool the peanuts to room temperature in 30 sec. To prevent cross contamination between reps, a new pair of gloves was used for loading each batch into the small rack and into the oven and a separate pair used to remove each batch. Between runs, the small rack and the cooler were sprayed with 70% ethanol and allowed to dry before the next batch came into contact with them. Cooled peanuts were placed in plastic bags for mtDNA and E. faecium plate count analysis.
Culture Enumeration
Ten grams were taken from each 100 g replicate and placed in a stomacher bag (Filtra-bag, Fisher, Pittsburg, PA) with 10 mL sterile 0.9 % saline (1:1 dilution). Peanuts were stomached in a Seward Stomacher 400 (Tekmar, Cincinnati, OH) for 2 min at normal speed. Filtrate was aseptically removed from the stomacher bag, serially diluted and plated as above using the simplified agar plate technique (Jett et al., 1997) with BHI agar (BD, Sparks, MD). Plates were incubated at 35 °C over night. Plates were counted manually and CFU/g peanuts were calculated, taking into account concentration and dilution factors.
Peanut Dissection
To determine if there were significant positional differences in mtDNA fragmentation after roasting, single peanuts were dissected and separated into the following parts: testa (skin), embryo, outer and inner endosperm. Georgia Green, medium grade size, runner peanuts were roasted at 167 °C as previously described and samples given the following time treatments: 0, 12 and 18 min. Three peanuts were chosen at random from each time point and dissected using a razor blade and cutting board, both of which were decontaminated between samples with 70% ethanol. The skin and embryo were removed and placed in microcentrifuge tubes. The endosperm was dissected in the following manner to differentiate between inner and outer portions: skin was removed and peanut cotyledons were separated, with the outer portion facing upward, two longitudinal cuts were made, dividing the cotyledon into three long sections, the middle section was turned on its side and cut in half, dividing the inner from the outer portion, the middle of each inner and outer portion was used for DNA extraction (∼ 5 to 10 mg). All samples were frozen at −80 C until DNA extraction with MasterPure DNA purification kit (Epicentre, Madison, WI) using the tissue sample portion of the protocol. Tissues were not ground but placed directly into first enzyme procedure.
DNA Extraction
Three peanuts from each replicate were ground under liquid nitrogen in a mortar and pestle. The mortar and pestle were thoroughly cleaned between samples with 70% ethanol to prevent cross contamination. DNA was extracted using ca. 2.5 mg or one inoculation loop of ground peanut in the MasterPure DNA purification kit (Epicentre, Madison, WI) using the tissue sample portion of the protocol. DNA was quantified and qualified with a spectrophotometer (Nanodrop, Wilmington, DE).
Quantitative PCR (qPCR)
qPCR was run in 25 μl total volume with 2X IQ SYBR Green supermix (SYBR Green I dye, 50 U/ml iTaq DNA polymerase, 0.4 mM each of dATP, dCTP, dGTP and dTTP, 6 mM MgCl2, 40 mM Tris-HCl, pH 8.4, 100 mM KCl, 20 nM fluorescein; BioRad, Hercules, CA), 300 nM final concentration each for forward and reverse primers, peanut DNA (5-10 ng/reaction) and real-time PCR (RT-PCR) water (Ambion, Austin, TX) to final volume.
Amplifications were performed in a MyiQ (BioRad, Hercules, CA) thermal cycler with the following conditions: 95 C for 3 min; 40 cycles of 95 C for 30 sec, 60 C for 30 sec, 72 C for 30 sec; with FAM channel optics on during annealing stage. No template and positive controls were used for all assays. The positive control was used to normalize data between assays. For a sample to be considered positive, its threshold cycle (Ct) value must be less than all negative control reactions and its corresponding amplification curve had to exhibit the three distinct phases of quantitative PCR: lag, linear and plateau. Ct value was evaluated as a time-temperature integrator in dry roast processes in peanuts. The lower the Ct value, the higher the copy number of the target mtDNA sequence. Therefore, an increase in Ct value indicates fragmentation of target by thermal mechanisms.
Hunter L Color Determination
The Hunter L color value was determined for each time point and replication. Testa were removed and 80 g of sample were placed in a glass petri dish and color was determined on a calibrated HunterLab DP 9000 with D25 sensor (Hunter Associates Laboratory; Reston, VA) utilizing the L, a, b scale. Color was determined three times per sample with the peanuts removed and resorted in the petri dish between readings with nuts placed outer-side down if broken. The color was expressed as the mean of three replications per sample.
Comparison of Processing and DNA Extraction Methods
Three different techniques were evaluated for DNA extraction and qPCR efficacy: mortar and pestle, commercial micro-centrifuge grinder tubes, no grinding but use of the removed embryo only. All extraction techniques were performed in duplicate. DNA concentrations, quality assessments (260/280 ratios) and Ct values were calculated twice for each sample to examine within sample variation. All were processed with Master Pure DNA extraction kit. Mortar and pestle procedure was used as previously described in the roasting assays. Grinder tubes were utilized with frozen peanut samples which were chopped into small pieces (5 -10 mg) with sterile razor blades and placed into micro-centrifuge grinding tubes (GE Healthcare, USA) then ground in 100 μl Master Pure tissue and lysis solution. Recovered solution from grinder tube centrifugation (60-70 μl) was brought up to 300 μl total tissue and lysis solution and proteinase K was added as the first step of the Master Pure DNA extraction kit. In the no-grind protocol the dissected peanut embryo was added to the first proteinase K enzymatic step of the Master Pure DNA extraction. After extraction, all DNA samples were quantified and quality evaluated by Nanodrop spectrophotometer. Quantitative PCR was run for mtDNA as before.
Statistical analyses
All ANOVA statistical analyses were performed using SAS (Version 9.3, SAS Institute, Inc., Cary, NC, U.S.A.). The treatments were considered to be statistically different if p<0.05 (Tables 1 & 2). Simple linear regressions were performed and goodness-of-fit regression correlations determined using Excel (Microsoft; Redmond, WA) (Figures 1-6, 8, 9).
Results and Discussion
mtDNA Fragmentation in Dry Roasting
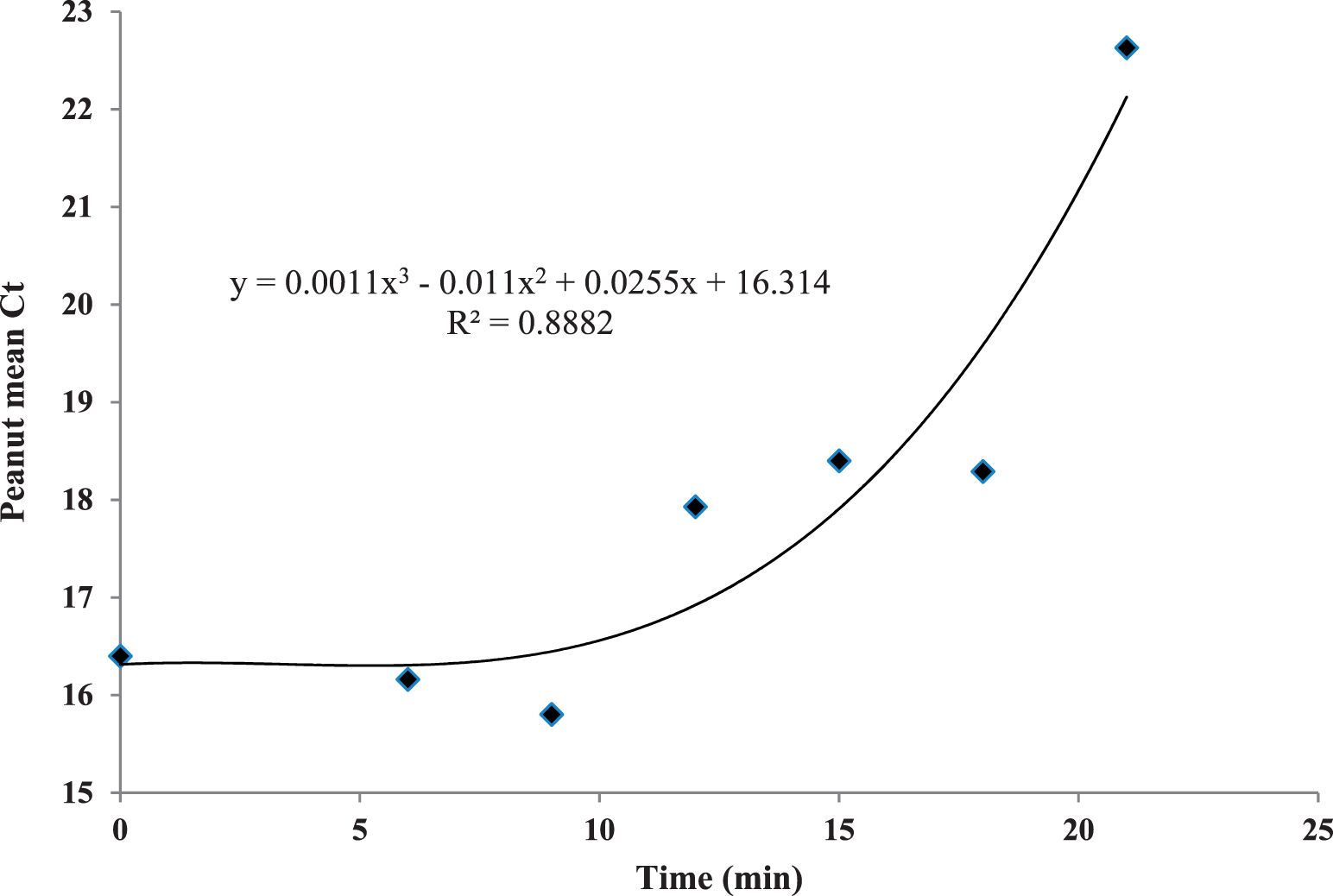
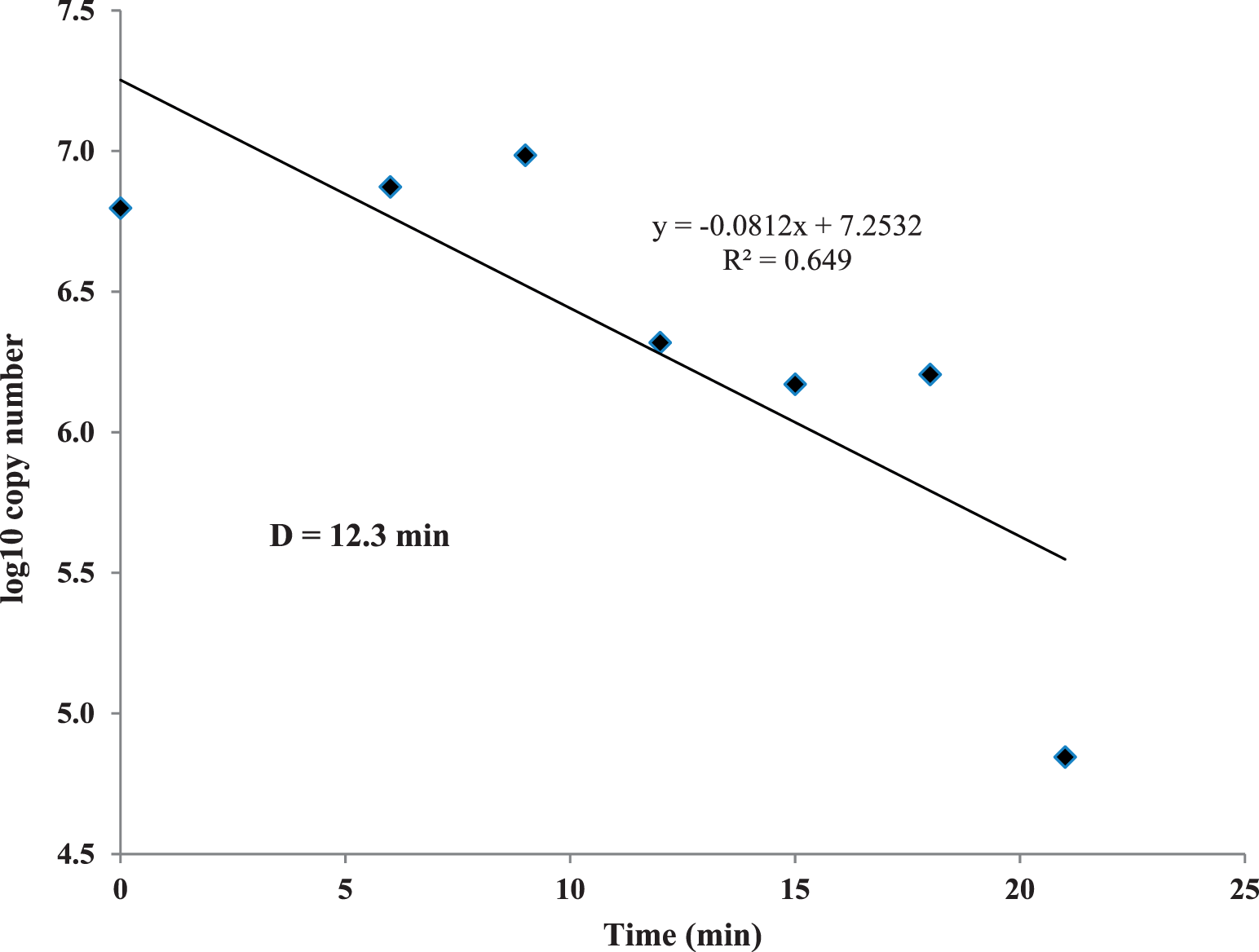
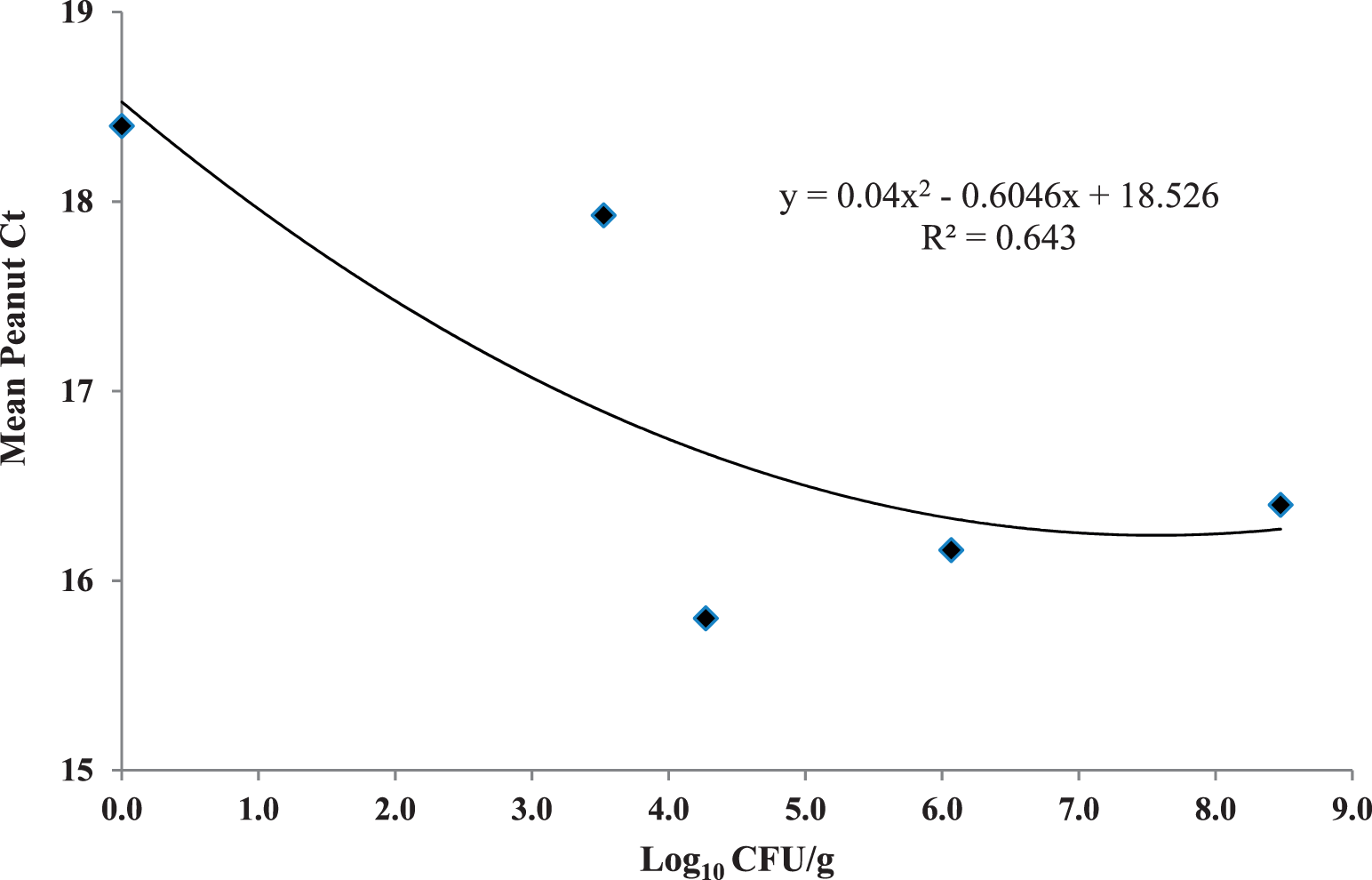
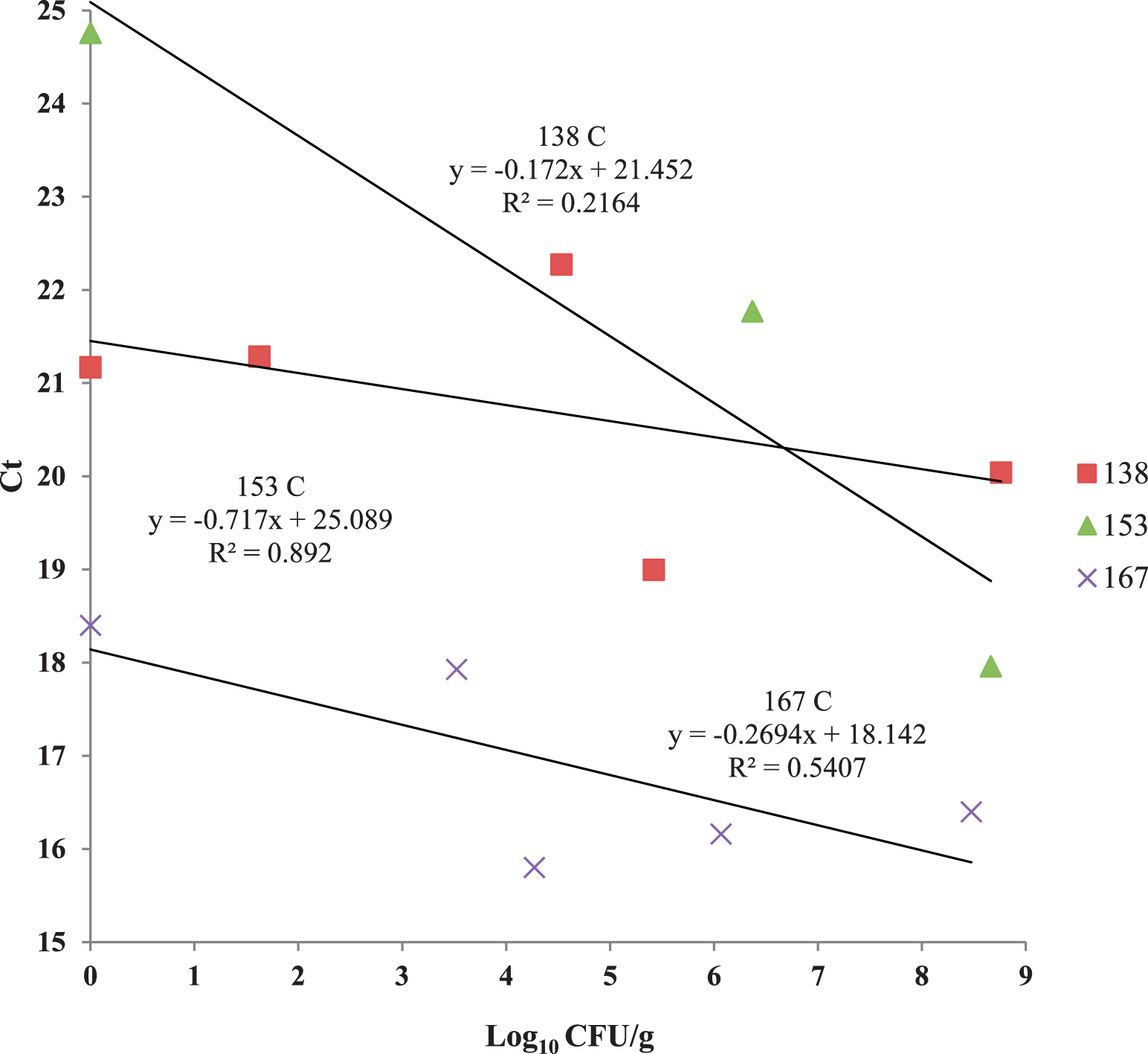
Peanut mtDNA fragmentation was assayed during a dry roast using a lab-scale convection oven. While this oven had convective airflow it was not comparable to industrial-scale, commercial ovens which utilize a conveyor belt, multi-directional air flows and more than one layer of peanuts. Ct values were initially steady, then increased after 12 min of roasting at 167 C to a mean value ca. 22 units after 21 min (Figure 1). This means that the copy number of the mtDNA target was decreasing due to fragmentation and destruction by dry heat. Thus, Ct was evaluated as a time-temperature integrator for the peanut dry roast process. However, Ct values did not increase in a linear fashion over time, perhaps due to low peanut Aw (0.40; Lee and Resurreccion, 2004) where water was sequestered in the solid matrix of the nut, causing an initial lag period. This phenomenon may possibly be explained that after ca. 10 min at 167 C, peanut cell membranes were broken and water was released, promoting mtDNA fragmentation and Ct values to increase rapidly in the last 2 min of the trial. The D value of mtDNA fragmentation was calculated used the log10 copy number as determined by the qPCR standard curve for the 174 bp amplicon (Caldwell et al. 2015a). The D value was 12.3 min at 167 C (Figure 2), which was slightly higher than values obtained for low- and high-acid thermal processes in previous reports (11.29 and 11.63 min, respectively) (Caldwell et al. 2015a, Caldwell et al. 2015b). However, the correlation between log10 copy number and time (min) was much lower (R2 = 0.65) than for these wet processes (low acid, R2 = 0.87; high-acid, R2 = 0.95). Peanut mtDNA fragmentation was correlated to surrogate death at 167 °C (Figure 3, R2 = 0.64) by a quadratic equation. Dry roasting at 167 C exhibited similar trends, such as initial Ct lag, to the other lower temperature treatments of 138 and 153 C (data not shown). All three temperature treatments were plotted and compared Ct values to E. faecium reduction (Figure 4), with the expectation that Ct values would increase as log10 CFU/g bacteria decreased. While this was the trend, the plotted lines had low correlation coefficients. The 153 C treatment had the highest correlation between Ct and log10 CFU/g (R2 = 0.89), possibly due to fewer time points than the other treatments. The 167 and 138 C treatments exhibited only weak correlations between Ct increase and E. faecium reduction (R2 = 0.54 and 0.22, respectively).
Peanut Salmonella Surrogate
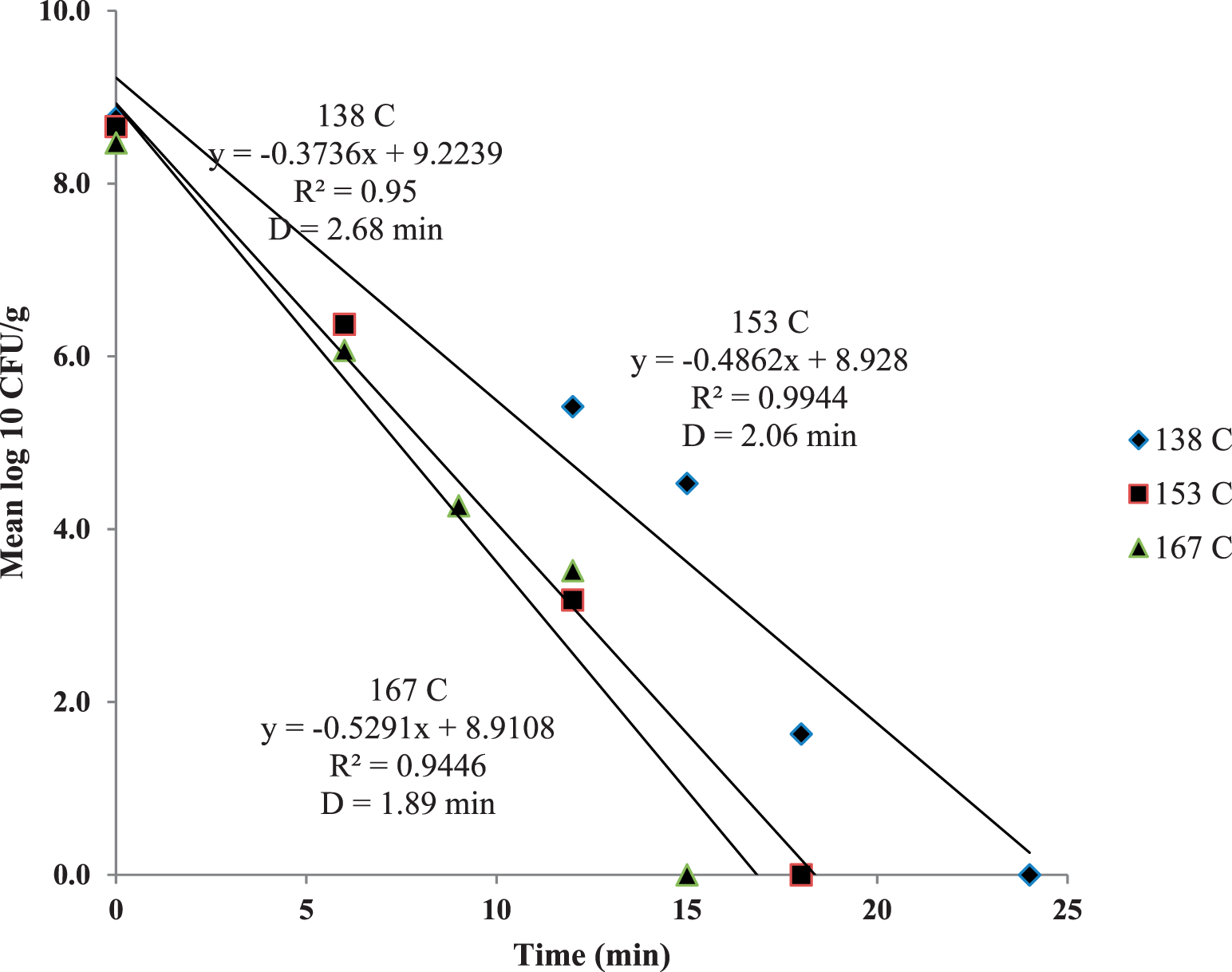
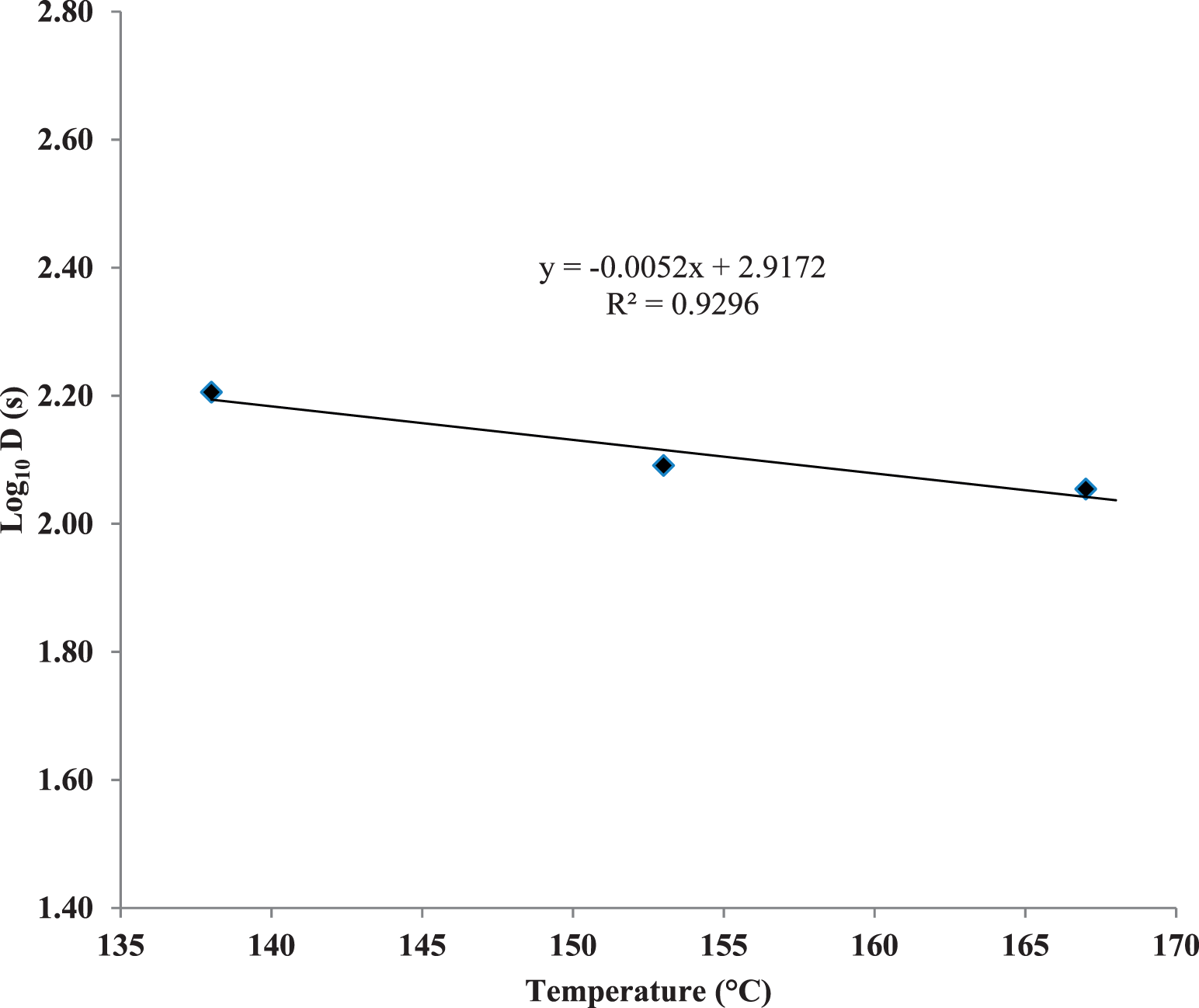
E. faecium D values were determined (Figure 5) and decreased only slightly with significant increases in temperature. D values for E. faecium were 2.68, 2.06 and 1.89 min for 138, 153 and 167 C dry roasting temperatures, respectively. The E. faecium z-value was very large (192 C) as microbial destruction in dry, roasted nuts does not exhibit true first-order kinetics, but has significant tailing (Figure 6; Harris, 2009). Podolak et al. (2010) stated that care must be taken when applying D and z-values from the literature in processes involving low-moisture foods and Salmonella contamination. The kinetics of E. faecium reduction in this study (4-log reduction after 10 min at 167 C) were similar to an earlier study (Sanders and Calhoun, 2014) which evaluated the usefulness of this organism as a Salmonella surrogate in peanut roasting.
Hunter L Color Values
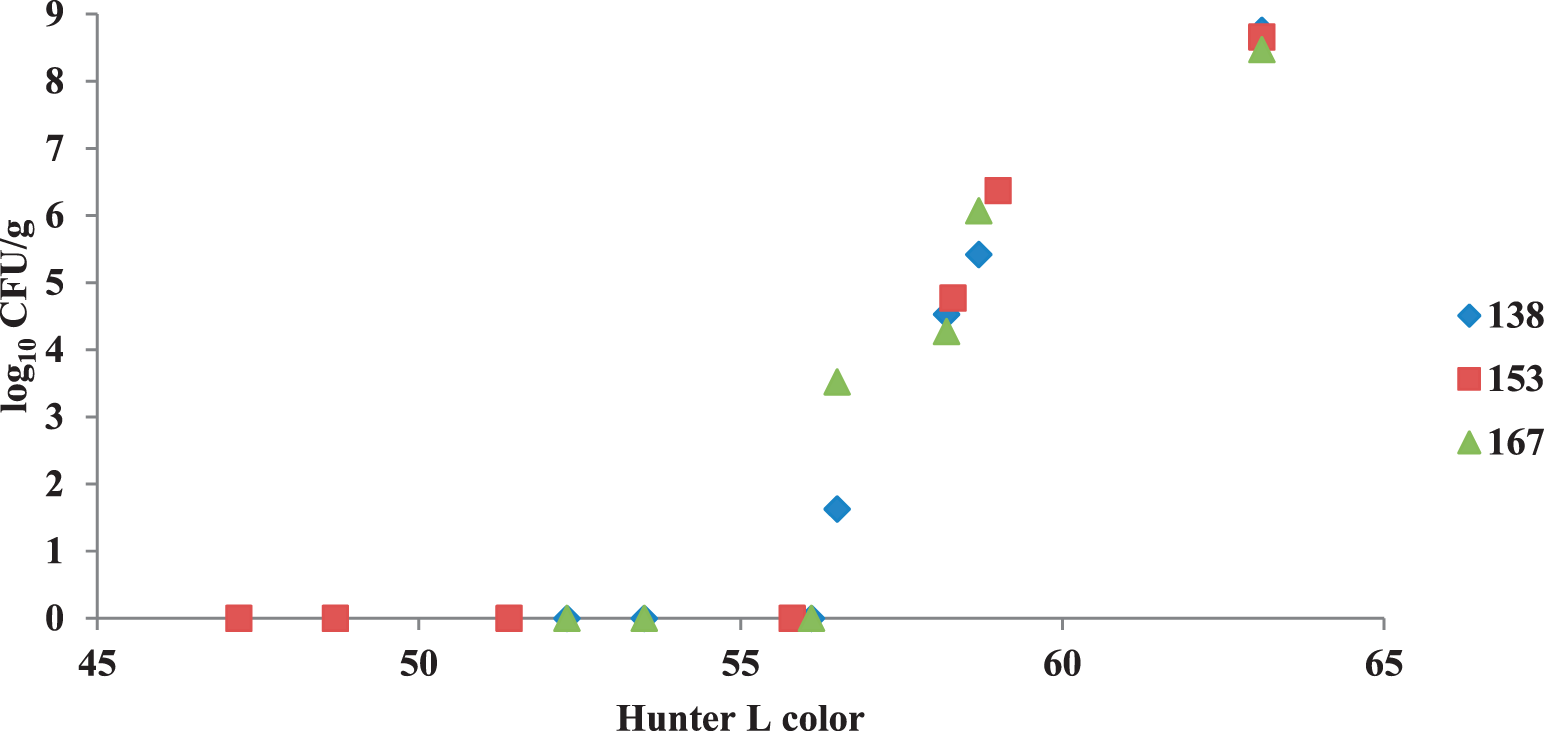
With this particular variety and lot of peanuts, one Hunter L color value (L=56) consistently corresponded to an apparent complete reduction of E. faecium regardless of time/temperature treatment (Fig. 7). Other researchers (McDaniel et al., 2012) have also noted the achievement of equivalent surface colors using different roast time-temperature combinations. They related Hunter L values to quality and nutrient parameters such as moisture content, sugars, amino acids, tocopherols and antioxidants. They concluded that peanuts roasted to equivalent Hunter L values using different time-temperature treatments were not equivalent in quality parameters. Further research with Hunter L values versus safety parameters may reveal a similar trend, since microbial destruction might not be a simple surface phenomenon and, like the quality parameters stated in the study above, involve the entire nut. Limits of detection for bacteria range from 400 to 1,000 CFU/g using culture methods described in this study. Since there is zero tolerance for Salmonella spp. in foods, Hunter L values corresponding to total bacterial kill would have to have a safety factor added to take limits of detection into account.
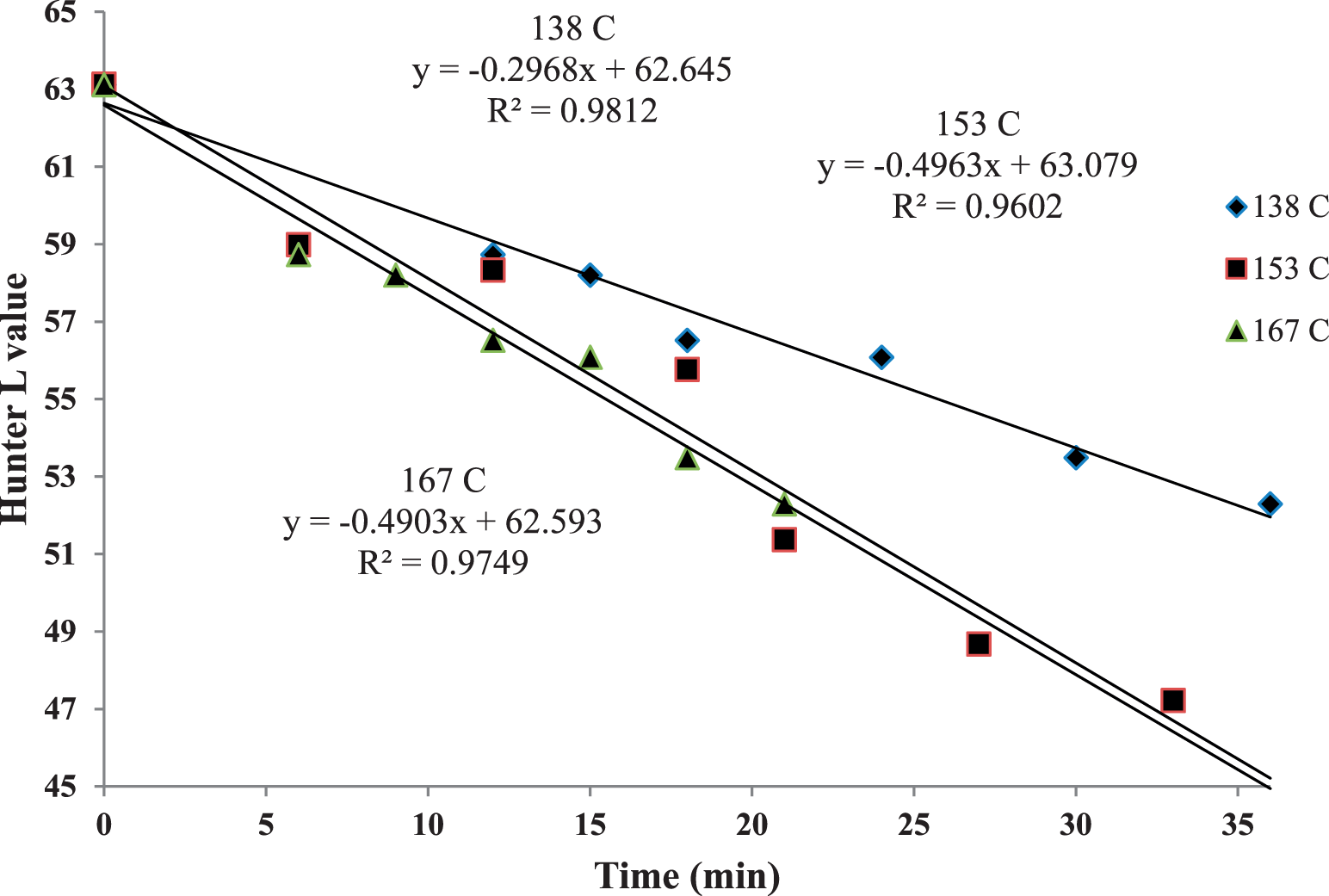
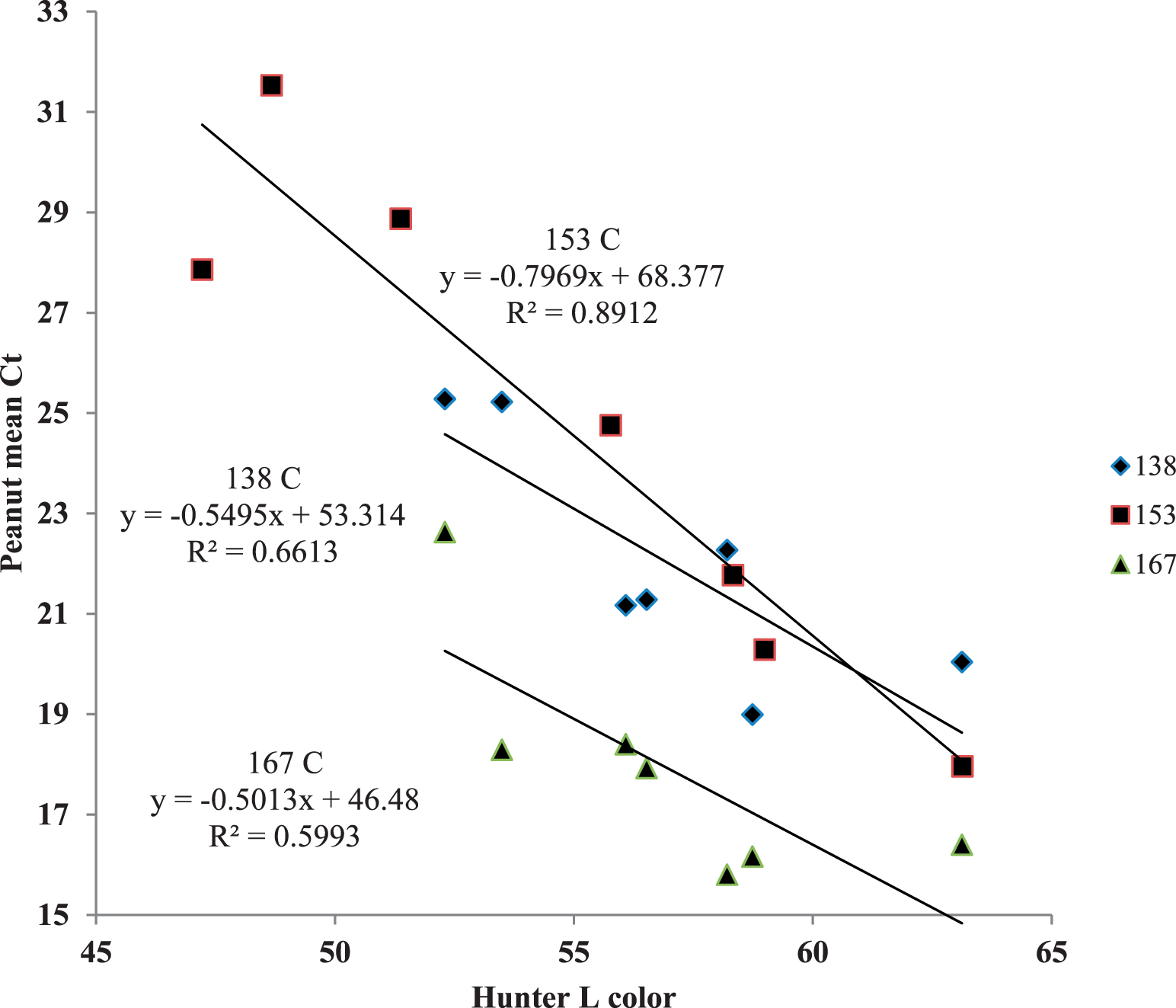
Hunter L values had an inverse linear relationship with high correlations (R2 > 0.96) to time-temperature in the three treatments studied (Figure 8). Peanut mean Ct values were compared to Hunter L values; the 153 C treatment had the highest correlation (R2 = 0.89) using simple linear regression (Figure 9). This indicates that surface browning was most closely associated with mtDNA fragmentation at that time-temperature treatment. Mitochondrial DNA fragmentation was more closely related to color change than E. faecium reduction. Bacteria reduction times in dry roasts are complex and do not exhibit first-order kinetics (Harris, 2009) compared to simple browning reactions. Therefore, mtDNA fragmentation is not a good predictor of indicator bacteria destruction.
Peanut Dissection
While peanut mtDNA Ct values increased with time of dry roast, there was wide variation at each time point (>1.0 Ct, data not shown). This was probably due to the part of the peanut that was chosen for extraction and to small sample size (n = 3). Our peanut dissection assay indicated large variation between individual peanuts (data not shown). A visual examination of individual peanuts also revealed large differences in roast color. Since Hunter L values use an average of 80 g, individual peanuts are not generally assessed. Log10 copy number, derived from Ct values, between different parts of the same peanut also varied (Table 1). Significant differences were found between the log10 copy numbers of the testa or skin and the embryo and both the inner and outer endosperm at all time points in the 167 C dry roast. Since the testa was desiccated compared to the other peanut parts, this was not unexpected. The log10 copy number of the embryo and the inner and outer endosperm (cotyledons) were not significantly different prior to roasting at time 0. After roasting for 12 min, the inner endosperm has a significantly higher log10 copy number compared to the embryo and outer endosperm, since the temperature inside the peanut was probably slightly lower than the outer portion of the nut, resulting in less mtDNA fragmentation. After 18 min of dry roasting, the inner and outer endosperm had equilibrated, exhibiting no significant difference in log10 copy number. However, at this time point, the embryo log10 copy number was significantly lower than either part of the endosperm. The embryo has a smaller mass than the cotyledons but because the area between the cotyledons is open it is exposed to hot air through the midline of the peanut. The embryo is easy to remove and peanut dissection confirmed the usefulness of the embryo as a reliable sample site. Presently, only surface temperatures of nuts are measured using thermocouple probes (Harris et al., 2012). In future studies, dissection and fragmentation of mtDNA could be combined with thermocouple probes to monitor internal peanut temperatures and find the cold spot in a complex solid. A non-invasive method could provide more accuracy.
Peanut Processing Prior to DNA Extraction
Other researchers have used dry ice and grinding in a mortar and pestle to process the nut samples prior to DNA extraction (Hird et al., 2003). However, prevention of DNA cross-contamination between samples in the mortar requires frequent cleaning. Different methods of peanut grinding and processing prior to DNA extraction did not significantly affect the quality of DNA (Table 2). The 260/280 ratio, indicative of DNA quality, across all treatments was between 1.7 to 2.1 with a mean and standard deviation of 1.92 +/- 0.13. Ratios above 1.8 are considered acceptable for downstream processes such as qPCR. No qPCR inhibition was seen due to fats, oils or other peanut components as tested by serial dilution (data not shown). DNA concentrations varied with grinding process. The mortar system had the most consistent DNA concentrations while both the grinding microcentrifuge tube and no-grind embryo process varied 1.5- to 3-fold. However, when DNA concentrations were normalized to 5-10 ng/well, Ct values were similar within and between grinding treatments, although Ct values for the no-grind procedure were ca. 0.8 units higher and significantly different. Using peanut embryos in the no-grind process produced less variation in Ct values within samples and between samples; an important factor in developing upstream DNA extraction protocols for qPCR. The no-grind process, while producing somewhat higher Ct values, was rapid, required no special equipment such as dry ice and reduced cross-contamination of samples while still providing robust Ct values. Future studies requiring DNA extraction of nuts can utilize the simplified, no-grind process without significantly effecting downstream protocols such as qPCR. More consistent Ct values within treatment samples might increase the power of mtDNA fragmentation as a TTI in dry roast processes.
Summary and Conclusions
Intrinsic mtDNA fragmentation, as measured by increase of Ct value in qPCR, occurs during dry roasting of peanuts. D values for these solids with low water activity are slightly higher than in low-acid, high-temperature and high-acid, low temperature processes (Caldwell et al. 2015a, Caldwell et al. 2015b). Threshold cycle (Ct) correlations to safety ( E. faecium reduction) and quality (Hunter L value) are low, due primarily to initial lag time of DNA destruction. Product contamination with Salmonella probably occurs after roasting during handling and further processing. This means that indicators of thermal process efficacy are of minimal value until equipment and employee cross-contamination after roasting are eliminated.
Intrinsic mtDNA fragmentation with dissection can be utilized to pinpoint the coldest spot in a solid food such as a peanut, providing a worst-case scenario for testing.
Acknowledgements
The authors would like to acknowledge Janet Hayes for lab management, ordering supplies and troubleshooting equipment; Sandra Parker for secretarial, grant and travel assistance; Bernard Eckhardt for IT support; and Joy Smith for statistical analysis. Mention of a trademark or proprietary product does not constitute a guarantee or warranty of the product by the U. S. Department of Agriculture or North Carolina Agricultural Research Service, nor does it imply approval to the exclusion of other products that may be suitable. An international and United States patent application has been filed for this process.
Literature Cited
Almond Board of California (ABC). Guidelines for validation of dry roasting processes. October 2007 , http://www.almonds.com/sites/default/files/content/attachments/dry-roast-validation-guidelines.pdf
B., Aviles, C Klotz, T Smith, R Williams, and M Ponder (2013). Survival of Salmonella enterica serotype Tennessee during simulated gastric passage is improved by low water activity and high fat content. J. Food Prot 76 : 333 - 337 .
P., Balachandran, Y Cao, L Wong, M.R Furtado, O.V Petrauskene, and R.S Tebbs (2011). Evaluation of applied biosystems MicroSEQ real-time PCR system for detection of Salmonella spp. in food. J. AOAC Int 94 : 1106 - 1116 .
BAM: Bacteriological Analytical Manual, 8th Edition, Revision A. Chapter 4. http://www.fda.gov/food/foodscienceresearch/laboratorymethods/ucm070149
S.L., Burnett, E.R Gehm, W.R Weissinger, and L.R Beuchat (2000). Survival of Salmonella in peanut butter and peanut butter spread. J. Appl. Microbiol 89 : 472 - 477 .
Caldwell J.M., Perez-Diaz I.M., Sandeep K.P., Simunovic J., Harris K., Osborne J., and H.M Hassan 2015 a Mitochondrial DNA fragmentation as a molecular tool to monitor thermal processing of Plant-Derived, Low-Acid Foods and Biomaterials J. Food Sci. Aug (8) 80 : M1804 - M1814 .
Caldwell J.M., Perez-Diaz I.M., Simunovic J., Harris K., Hassan H., and K.P Sandeep 2015 b Mitochondrial DNA fragmentation to monitor processing parameters in high acid, plant-derived foods J. Food Sci. Nov 10 doi: 10.1111/1750-3841.13139 .
S., Calhoun, L Post, B Warren, S Thompson, and A.R Bontempo (2013). Prevalence and concentration of Salmonella on raw shelled peanuts in the United States. J. Food Prot 76 : 575 – 579 .
E., Crowley, P Bird, K Fisher, K Goetz, M.J Benzinger, J Agin, D Goins, and R.L.A Johnson (2011). Evaluation of VIDAS Salmonella (SLM) easy Salmonella method for the detection of Salmonella in a variety of foods: collaborative study. J. AOAC Int 94 : 1821 - 1834 .
J.Y D'Aoust, (1985). Infective dose of Salmonella typhimurium in cheddar cheese. Am. J. Epidemiol 122 : 717 - 720 .
Deng, X., J.K Salazar, S Frezet, D MacCannell, E.M Ribot, P.I Fields, W.F Fricke, and W Zhang 2013 Genome sequence of Salmonella enterica serotype Tennessee strain CDC07-0191, implicated in the 2006-2007 multistate food-borne outbreak linked to peanut butter in the United States Genome Announc 1 (3) : e00260 – 13 . doi:10.1128/genomeA.00260-13 .
O.N., Gill, P.N Socket, C.L.R Bartlett, and M.S.B Vaille (1983). Outbreak of Salmonella napoli infection caused by contaminated chocolate bars. Lancet I : 574 - 577 .
Harris, L March. 2009 Process validation for low water activity foods IAFP http://www.foodprotection.org/files/rr_presentations/RR_07.pdf
L.J., Harris, A.R Uesugi, S.J Abd, and K.L McCarthy (). Survival of Salmonella Enteritidis PT 30 on inoculated almone kernels in hot water treatments. Food Res. International 45 : 1093 - 1098 .
Y., He, D Guo, J Yang, M.L Tortorello, and W Zhang (2011). Survival and heat resistance of Salmonella enterica and Escherichia coli O157:H7 in peanut butter. Appl. Environ. Microbiol 77 : 8434 - 8438 .
Y., He, Y Li, J.K Salazar, J Yang, M.L Tortorello, and W Zhang (2013). Increased water activity reduces thermal resistance of Salmonella enterica in peanut butter. Appl. Environ. Microbiol 79 : 4763 - 4767 .
H., Hird, J Lloyd, R Goodier, J Brown, and P Reece (2003). Detection of peanut using real-time polymerase chain reaction. Eur. Food Res. Technol 217 : 265 - 268 .
R., Hoerner, J Feldpausch, R.L Gray, S Curry, Z Islam, T Goldy, F Klein, T Tadese, J Rick, and M Mozola (2011). Reveal Salmonella 2.0 test for detection of Salmonella spp. in foods and environmental samples. Performance Tested Methods 960801. J. AOAC Int 94 : 1467 - 1480 .
W.P., Izurieta, and E Komitopoulou (2012). Effect of moisture on Salmonella spp. heat resistance in cocoa and hazelnut shells. Food Res. Int 45 : 1087 - 1092 .
B.D., Jett K.L., Hatter M.M., Huycker and M.S Gilmore (1997). Simplified agar plate method for quantifying viable bacteria. BioTechniques 23 : 648 - 650 .
D., Killalea, L.R Ward, and D deRoberts (1996). International epidemiological and microbiological study of out-break of Salmonella agona infection from a ready-to-eat savoury snack I. : England and Wales and the United States British Med. J 313 : 1105 - 1107 .
J.S., Kim, M.S Jahng, G.G Lee, K.J Lee, H.K Chae, J.H Lee, J.H Lee, and M.H Kim (2011). Rapid and simple detection of the invA gene in Salmonella spp. by isothermal target and probe amplification (iTPA). Lett. Appl. Microbiol 52 : 399 - 405 .
M.D., Kirk, C.L Little, M Lem, M Fyfe, D Genobile, A Tan, J Threlfall, A Paccagnella, D Lightfoot, H Lyi, L McIntrye, L Ward, D.J Brown, S Surnam, I.S.T Fisher (2004). An outbreak due to peanuts in their shell caused by Salmonella enterica serotypes Stanley and Newport – sharing molecular information to solve international outbreaks. Epidemiol. Infect 132 : 571 - 577 .
C.M., Lee, and A.V.A Resurreccion (2004). Descriptive profiles of roasted peanuts stored at varying temperatures and humidity conditions. J. Sens. Stud 19 : 433 - 456 .
J., Li, W.D Cheung, J Opdyke, J Harvey, S Chong, and C.G Moon (2012). Samsung Salmonella Detection Kit. AOAC performance tested method (SM). J. AOAC Int 95 : 1656 - 668 .
L., Ma G., Zhang P., Gerner-Smidt V., Mantripragada I., Ezeoke and M. P Doyle (2009). Thermal inactivation of Salmonella in peanut butter. J. Food Prot 72 : 1596 - 1601 .
K.L., Mattick, F Jorgensen, J.D Legan, H.M Lappin-Scot, and T.H Humphrey (2000). Habituation of Salmonella spp. at reduced water activity and its effect on heat tolerance. Appl. Environ. Microbiol 66 : 4921 - 4925 .
K.A., McDaniel, B.L White, L.L Dean, T.H Sanders, and J.P David (2012). Compositional and mechanical properties of peanuts roasted to equivalent colors using different time/temperature combinations. J. Food Sci 77 : C1292 - 1298 .
MMWR Morbidity and Mortality Weekly Report. Multistate outbreak of Salmonella serotype Tennessee infections associated with peanut butter – United States, 2006-2007. June 1; 56 ((21)) : 521 - 524 .
MMWR Morbidity and Mortality Weekly Report 2013 Notes from the field: Salmonella Bredeney infections linked to a brand of peanut butter – United States, 2012. Feb. 15; 62 (06) : 107 - 107 .
R., Podolak, E Enache, W Stone, D.G Black, and P.H Elliott (2010). Sources and risk factors for contamination, survival, persistence, and heat resistance of Salmonella in low-moisture foods. J. Food Prot 73 : 1919 - 1936 .
T., Sanders, and R.S Calhoun (2014). Effect of oil and dry roasting of peanuts at various temperatures and times on survival of Salmonella and Enterococcus faecium . Peanut Sci 41 : 65 - 71 .
W., Scheil, S Cameron, C Dalton, C Murray, and D Wilson (1998). A South Australian Salmonella Mbandaka outbreak investigation using a database select controls. Aust. N. Z. J. Public Health 22 : 536 - 539 .
A.N., Sheth, M Hoekstra, N Paatel, G Ewald, C Lord, C Clarke, E Villamil, K Niksich, C Bopp, T.-A Nguyen, D Zink, and M Lynch (2011). A national outbreak of Salmonella serotype Tennessee infections from contaminated peanut butter: A new vehicle for Salmonellosis in the United States. Clin. Infect. Dis 53 : 356 - 362 .
J., Sobel, D.L Swerdlow, and J Parsonnet (2001). Is anything safe to eat?. Curr. Clin. Top Infect. Dis 21 : 114 - 134 .
Notes
- Department of Food, Bioprocessing, and Nutrition Sciences, North Carolina State University, Raleigh, NC 27695; [^]
- USDA-Agriculture Research Service, SEA, Food Science Research Unit, 322 Schaub Hall-NCSU, Raleigh, NC 27695; [^]
- USDA-Agriculture Research Service, SEA, Market Research Unit, 236-Schaub Hall, NCSU, Raleigh, NC 27695. [^] *Corresponding author's E-mail: jane.caldwell@transagra.com
Author Affiliations