Introduction
Numerous studies have discussed the negative impact weeds have on peanut production (Buchanan et al. 1982; Cardina and Brecke 1989; York and Coble 1977; Young et al. 1982; Wilcut et al. 1994). Weed control during peanut growth and development is critical to reduce inter-species competition and maintain optimum pod yields (Bridges et al. 1992; Burke et al. 2007; Cardina and Brecke 1989; Walker et al. 1989; York and Coble 1977). Weeds can dramatically inhibit digging and inversion procedures in peanut, leading to harvest losses and harvest inefficiency (Wilcut et al. 1994; Young et al. 1982). Additionally, weeds may serve as hosts for nematodes and diseases (Bird and Hogger 1973; Clewis et al. 2001; Hogger and Bird 1976; Martin 1958).
Growers use a combination of cultural and chemical control tactics to manage weeds; such as, promoting crop health through fertility and crop rotation, as well as preventing weeds from going to seed each year using herbicides or hand-weeding (Buchanan et al. 1982). In Georgia, Palmer amaranth (Amaranthus palmeri S. Wats) is considered the most troublesome weed in peanut production (Webster et al. 2005). Populations of Palmer amaranth exist in Georgia that are resistant to acetyl-lactate synthesis, glycine, and/or triazine herbicides (Heap 2013; Wise et al. 2009). Managing the weed seed bank within cropping systems has become challenging due to increased incidence of herbicide resistant Palmer amaranth. Keeley et al. (1987) reported that a single female Palmer amaranth can produce up to 600,000 seeds. Weed control failures due to resistance can directly influence weed control in subsequent years due to a buildup of weed seed in the soil (Dieleman et al. 1999; Hartzler and Roth 1993; Sparks et al. 2003; Webster et al. 1998).
Weed management in Georgia peanut production commonly requires a combination of residual and postemergence (POST) herbicides to maintain season long weed control and prevent the production of weed seed (Wilcut et al. 1995). Due to fewer hectares of peanut than other row crops, agrichemicals are rarely developed specifically for peanut. Thus, it is critical to evaluate herbicides developed for use in other row crops for their potential use in peanut.
Pyroxasulfone, formerly KIH-485 is a soil applied herbicide labeled for use in corn (Zea mays L.) and soybean (Glycine max L.) for the control of annual broadleaf weeds and grasses. Pyroxasulfone applied at rates between 60 and 180 g ai/ha has been documented to control: Amaranthus spp., Lolium spp., Urochloa spp., goosegrass (Eleusine indica L.), crowfootgrass (Dactyloctenium aegyptium L), and Digitaria spp. (Geier et al. 2006; Hulting et al. 2012; King and Garcia 2008; Koger et al. 2008; Knezevic et al. 2009; Nurse et al. 2011). Pyroxasulfone inhibits very long chain fatty acid synthesis similar to chloroacetamide, oxyacetamide, and tetrazolinone herbicides (Tanetani et al. 2009). Previous research has determined that peanut is adequately tolerant to pyroxasulfone applied 44 to 51 days after emergence (Prostko et al. 2011). Pyroxasulfone applied PRE to peanut has been documented to cause minimal early season stunting but no yield loss (Prostko et al. 2011).
Pyroxasulfone use in corn, cotton (Gossypium hirsutum L.), soybean, and wheat (Triticum aestivum L.) tolerance has been thoroughly described (Cahoon et al. 2012; Eure et al. 2013; Geier et al. 2006; Hulting et al. 2012; King and Garcia 2008; Koger et al. 2008). However, very little is known concerning peanut response to pyroxasulfone following PRE applications. Previous research indicates differential peanut cultivar response to herbicides (Jordan et al. 1998; McLean et al. 1994). Therefore, research was conducted to evaluate peanut cultivar response to pyroxasulfone applied PRE.
Materials and Methods
Field experiments were conducted during 2012 and 2013 at the University of Georgia Ponder Research Station near Ty Ty, GA on a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults) soil with 93% sand, 3% silt, 4% clay, 1% organic matter, and pH 6.0. Trials were arranged in a split-plot design with main plots consisting of three peanut cultivars [‘Georgia-06G’(Branch 2007a), ‘TifGuard’ (Holbrook et al. 2008), and ‘Georgia Greener’ (Branch 2007b)] and sub-plots consisting of three pyroxasulfone rates (0, 120, or 240 g ai/ha). All treatments were replicated 4 times. Peanut cultivars were planted in freshly tilled seed beds at a rate of 15 plants/m-row, in twin rows spaced 23 cm apart on a 91 cm center. Plots were 1.8 m (two sets of twin rows) wide and 9 m in length. Herbicide treatments were applied immediately following planting using a CO2-pressurized backpack sprayer calibrated to deliver 140 L/ha at 275 kPa. Immediately following pyroxasulfone application, the trial area was irrigated with 1.3 cm of water using overhead irrigation. Plots were maintained weed-free throughout the season using commonly applied PRE herbicides (pendimethalin plus dimethenamid plus flumioxazin) in combination with cultivation between plots and hand-weeding. Production, irrigation, and pest management practices other than specific treatments were held constant over the entire experiment to optimize peanut growth and development (Anonymous 2013).
Visual estimates of peanut stunting were recorded 10, 80, and 120 d after planting (DAP) using a scale of 0 to 100% where 0 = no stunting and 100 = complete death. Peanut plant density was recorded 20 DAP from 1 meter of twin-rows. Additionally, canopy height was recorded prior to harvest at 120 DAP for 5 plants per plot. To determine treatment effects on peanut maturity, 100 pods per plot were randomly collected immediately after inversion. The hull-scrape method was used to remove the exocarp of the peanut pod. This practice is recommended to determine peanut maturity for harvest timing to ensure optimum peanut pod yield and grade (Johnson 1987; Williams and Drexler 1981). Peanut have indeterminate growth and commonly have pods with varying levels of maturity (Sholar et al. 1995). Peanut pod mesocarp darkens as pods mature. Pods with brown or black mesocarps are mature while pod mesocarps that are white, yellow, or orange in color are immature. The distribution of immature and mature peanut pods can serve as an indicator of delayed maturity caused by cultivar, irrigation, fertility, or herbicide injury (Johnson et al. 1987; Sholar et al. 1995; Mixon and Branch 1985; Mozingo et al. 1991). Once pod mesocarps were removed from each sample, pods were grouped by color: black, brown, and other (white, yellow, orange). These data were combined into two groups; immature (white, yellow, orange) and mature (brown and black).
Peanut were inverted and harvested 3 days later using commercial equipment. Peanut yields were recorded and adjusted to 10% moisture. Data for all parameters were analyzed as a split-plot design and subjected to ANOVA using the PROC MIXED procedure in SAS (SAS Institute Inc., Cary, NC 27513) with peanut cultivar and pyroxasulfone rate as fixed affects and years and replications as random effects. Means of significant main effects and interactions were separated using Fisher’s Protected LSD test at P ≤ 0.05.
Results and Discussion
Plant Density and Stunting
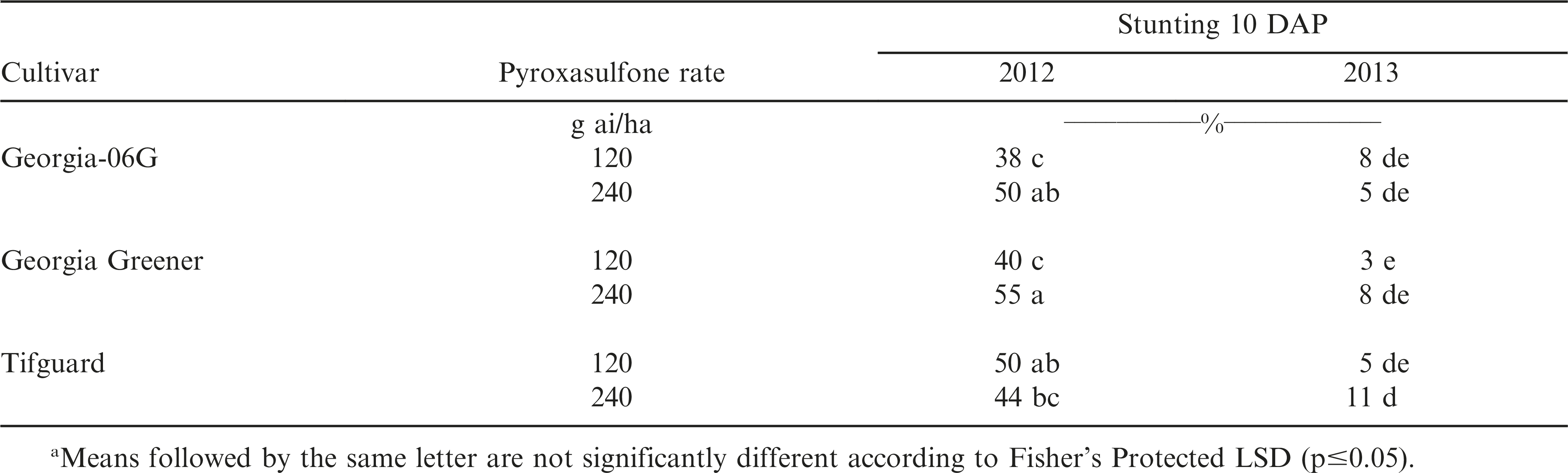
Peanut density was not influenced by the interaction of year, cultivar, and pyroxasulfone rate (P = 0.7783). Peanut plant density range from 14.3 to 14.4 plants per m of row and was not influenced by the main effect of pyroxasulfone rate (P = 0.3677) or peanut cultivar (P = 0.5590) (data not shown). Peanut stunting 10 DAP was influenced by the interaction of year, cultivar, and pyroxasulfone rate (Table 1). Greater stunting occurred during 2012 than in 2013. Peanut stunting 10 DAP during 2012 and 2013 ranged from 38 to 55% and 3 to 11%, respectively. During 2012, greater injury was observed in ‘Tifguard’ following 120 g ai/ha of pyroxasulfone applied PRE than in ‘Georgia-06G’. ‘Georgia Greener’ had greater stunting following 240 g ai/ha of pyroxasulfone applied PRE than in ‘Tifguard’. During 2013, pyroxasulfone applied at 240 g ai/ha resulted in greater stunting in ‘Tifguard’ than in ‘Georgia-06G’. Prostko et al. (2011) documented transient peanut stunting at one of two locations following pyroxasulfone applied PRE. Additionally, differential peanut cultivar response to herbicides has been observed in Virginia- and runner-type peanut cultivars (Jordan et al. 1998; McLean et al. 1994).
Several factors may have played a role in the differences observed in regards to early season peanut response to pyroxasulfone applied PRE. More rainfall occurred during peanut cracking in 2012 compared to 2013 (5 cm vs. 2.5 cm). Increased peanut stunting has been observed following application of PRE herbicide applications under cool, wet conditions (Grichar et al. 2004). Other research has shown that significant peanut injury from soil applied herbicides may occur if peanut emergence coincides with rain events (Johnson et al. 2006; Jordan 2007; Prostko 2013). Soil type can influence crop tolerance to pyroxasulfone (Anonymous 2012; Cahoon et al. 2012; Koger et al. 2008; Nurse et al. 2011; Odero and Wright 2013). Research in other crops has shown greater crop injury from pyroxasulfone applied PRE on course-textured soils than on fine-textured or organic soils (Cahoon et al. 2012; Eure et al. 2013; Nurse et al. 2011; Koger et al. 2008; Odero and Wright 2013). Sweet corn injury has been documented to be greater than 10% following pyroxasulfone applied at 250 g ai/ha on soil with 82% sand (Nurse et al. 2011). Pyroxasulfone applied PRE to sweet corn on soils high in organic matter has shown no visible injury (Odero and Wright 2013). In cotton, Koger et al. (2008) reported only transient injury on a silt loam soil following pyroxasulfone applied PRE. Others have reported significant cotton injury and stand loss following PRE application of pyroxasulfone on sandy soils (Cahoon et al. 2012; Eure et al. 2013).
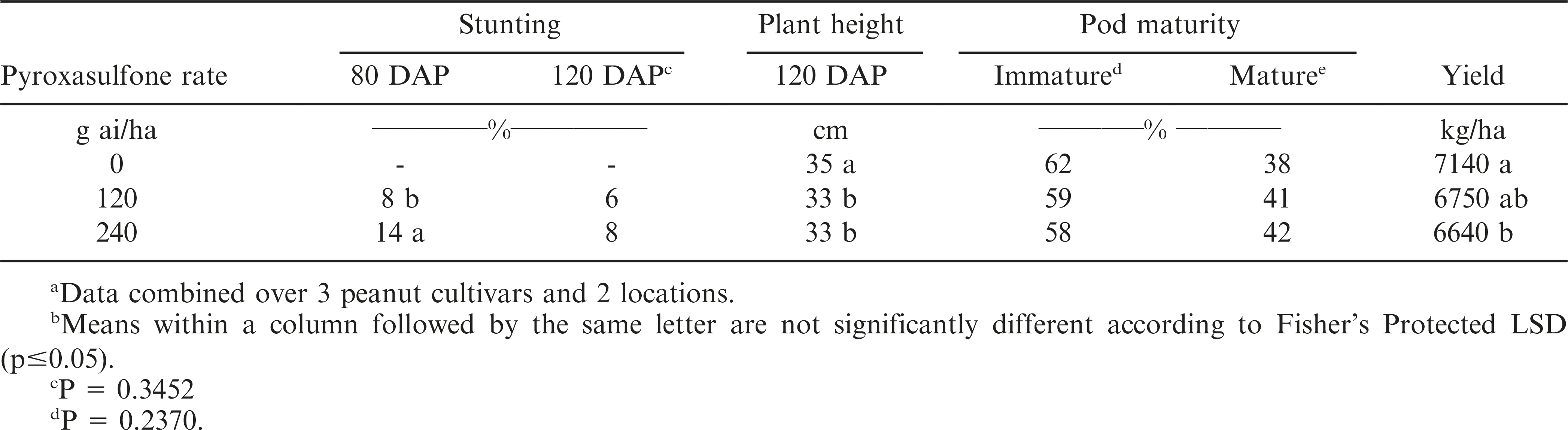
Peanut stunting 80 and 120 DAP was not influenced by the interaction of cultivar, pyroxasulfone rate, and year. However, peanut stunting at 80 DAP was influenced by the main effect of pyroxasulfone rate (Table 2). When pooled over cultivars and years, pyroxasulfone applied at 120 or 240 g ai/ha caused 8 and 14% stunting, respectively. By 120 DAP, peanut stunting ranged from 6 to 8% regardless of pyroxasulfone rate or cultivar (Table 2). Plant height was not influenced by the interaction of cultivar, pyroxasulfone rate, and year. The main effect of pyroxasulfone rate and cultivar did influence plant height 120 DAP. At this time, plant height was reduced from 35 cm in the non-treated control to 33 cm when pyroxasulfone was applied PRE (Table 2). When pooled over years and pyroxasulfone rates, ‘Georgia-06G’ and ‘Tifguard’ were 5 and 4 cm taller than ‘Georgia Greener’, respectively (Table 3).
Maturity and Yield
Peanut maturity and yield were not influenced by the interaction of cultivar, pyroxasulfone rate, and yield. Additionally, peanut maturity was not influenced by cultivar or pyroxasulfone rate (Tables 2 and 3). Peanut yield was influenced by the main effect of pyroxasulfone rate and cultivar. Yield was reduced following pyroxasulfone applied PRE at the 240 g ai/ha but not at the 120 g ai/ha rate (Table 2). When pooled over cultivars and trials, peanut yield was 7,140 kg/ha in treatments that did not include pyroxasulfone. Treatments that included pyroxasulfone applied at 120 g ai/ha yielded similar to treatments without pyroxasulfone. Pyroxasulfone applied at 240 g ai/ha reduced yield to 6,640 kg/ha (7%) as compared to the non-treated control. Previously, Prostko et al. (2011) did not observe yield loss following PRE application of pyroxasulfone in peanut. Peanut cultivar did influence peanut yield. ‘Georgia-06G’ yield was 630 to 635 kg/ha greater than ‘Georgia Greener’ and ‘Tifguard’ (Table 3). ‘Georgia-06G’ has historically produced greater pod yield than ‘Georgia Greener’ and ‘Tifguard’ (Branch 2012). Due to high yield potential, ‘Georgia-06G’ was planted on 77% of acreage in Georgia during 2012 (Beasley 2013).
In one of two trials, differential peanut cultivar tolerance was observed for stunting 10 DAP. ‘Tifguard’ was less tolerant to a 1x rate of pyroxasulfone than ‘Georgia Greener’ and ‘Georgia-06G’. Although peanut yield was not reduced following a 1x rate of pyroxasulfone applied PRE, the potential for early season stunting and less than a 2x safety margin on yield is concerning. Additionally, these trials were conducted under optimum conditions for plants to recover from early season herbicide injury. Results may have been different if late season conditions were not conducive for peanut recovery. Future research should be conducted to understand the influence of soil type and rainfall or irrigation timing on peanut injury from PRE applied pyroxasulfone.
Literature Cited
Anonymous 2012 Zidua herbicide label BASF Corporation Research Triangle Park, NC . Pages 12 .
Anonymous 2013 Peanut production update Cooperative Extension Service Series CSS-13-0110 . ed. J.P. Beasley, Jr., Athens, GA : University of Georgia . Pages 131 .
Beasley and J.P 2013 Cultivar options in 2013, Pages 55-56 In 2013 Peanut production update, Athens, GA : University of Georgia . Pages 108 .
Bird G.W and Hogger C.H 1973 Nutsedges as hosts of plant-parasitic nematodes in Georgia cotton fields Plant Dis. Rep 57 : 402 .
Branch W.D 2007a Registration of 'Georgia-06G' peanut J. Plant Reg 1 : 120 .
Branch W.D 2007b Registration of 'Georgia Greener' peanut J. Plant Reg 1 : 121 .
Branch W.D 2012 University of Georgia peanut breading program, Pages 12-17 In Peanut production update, Cooperative Extension Service Series CSS-12-0130 . ed. J.P. Beasley, Jr., Athens, GA : University of Georgia . Pages 108 .
Bridges D.C Brecke B.J and Barbour J.C 1992 Wild poinsettia (Euphorbia heterophylla) interference with peanut (Arachis hypogaea) Weed Sci 40 : 37 – 42 .
Buchanan G.A Murray D.S and Hauser E.W 1982 Weeds and their control in peanuts, Pages 209-249 In H.E. Pattee and C.T. Young (eds.) Peanut Science and Technology, Amer. Peanut Res. Educ. Soc. , Yoakum, TX . Pages 566 .
Burke I.C Schroeder M Thomas W.E and Wilcut J.W 2007 Palmer amaranth interference and seed production in peanut Weed Technol 21 : 367 – 371 .
Cahoon C.W York A.C and Jordan D.L 2012 Cotton tolerance and Palmer amaranth control with Zidua, Warrant, and Dual Magnum herbicides Page 1535 In 2012 Beltwide Cotton Conferences Orlando, FL January 3-6 , 2012 .
Cardina J and Brecke B.J 1989 Growth and development of Florida beggarweed (Desmodium tortuosum) selections Weed Sci 37 : 207 – 210 .
Clewis S.B Askew S.D and Wilcut J.W 2001 Common ragweed interference in peanut Weed Sci 49 : 768 – 772 .
Dieleman J.A Mortensen D.A and Martin A.R 1999 Influence of velvetleaf and common sunflower density variation on weed management outcomes Weed Sci 47 : 81 – 89 .
Eure P.M 2013 An assessment of cotton tolerance to pyroxasulfone, acetochlor, and metolachlor Page 660 In 2013 Beltwide Cotton Conferences San Antonio, TX January 7-10 , 2013 .
Geier P.W Stahlman P.W and Frihauf J.C 2006 KIH-485 and S-metolachlor efficacy comparisons in conventional and no-tillage corn Weed Technol 20 : 622 – 626 .
Grichar W.J Besler B.A Dotray P.A Johnson W.C and Prostko E.P 2004 Interaction of flumioxazin with dimethenamid or metolachlor in peanut (Arachis hypogaea L.) Peanut Sci 31 : 12 – 16 .
Hartzler R.G and Roth G.W 1993 Effect of prior years’ weed control on herbicide effectiveness on corn Weed Technol 7 : 611 – 614 .
Heap I 2013 The International Survey of Herbicide Resistant Weeds Online. Internet. Friday, October 20, 2013. Available www.weedscience.org.
Hogger C.H and Bird G.W 1976 Weed and indicator hosts of plant-parasitic nematodes in Georgia cotton and soybean fields Plant Dis. Rep 60 : 223 – 226 .
Holbrook C.C Timper P Culbreath A.K and Kvien C.K 2008 Registration of ‘Tifguard’ Peanut J. Plant Reg 2 : 92 – 04 .
Hulting A.G Dauer J.T Hinds-Cook B Curtis D Koepke-Hill R.M and Mallory-Smith Carol 2012 Management of Italian ryegrass in western Oregon with preemergence applications of pyroxasulfone in winter wheat Weed Technol 26 : 230 – 235 .
Johnson W.C III Prostko E.P and Mullinix B.J 2006 Phytotoxicity of delayed applications of flumioxazin on peanut Weed Technol 20 : 157 – 163 .
Johnson W.C and III 1987 The hull scrape method to assess peanut maturity Tifton, GA : Georgia Cooperative Extension Service Bulletin 958 . Pages 2 .
Jordan D.L 2007 Weed management in peanuts Pages 45-80 In 2013 Peanut Information. Cooperative Extension Service Series AG-331 Raleigh, NC : North Carolina State University . Pages 160 .
Jordan D.L Culpepper A.S Batts R.B and York A.C 1998 Response of Virginia-type peanut to norflurazon Peanut Sci 25 : 4 – 7 .
Keeley P.E Carter C.H and Thullen R.J 1987 Influence of planting date on growth of Palmer amaranth Weed Sci 35 : 199 – 204 .
King S.R and Garcia J.O 2008 Annual broadleaf control with KIH-485 in glyphosate-resistant furrow-irrigated corn Weed Technol 22 : 420 – 424 .
Knezevic S.Z Datta A Scott J and Porpoglia P 2009 Dose-response curves of KIH-for preemergence weed control in corn Weed Technol 23 : 420 – 424 .
Koger C.H Bond R Poston D.H Eubank T.W Blessitt J.B and Nandula V.K 2008 Evaluation of new herbicide chemistry: Does KIH-485 have a fit in the southern cotton producing region? Page 1738 In Proceedings of the Beltwide Cotton Conference New Orleans, LA January 2008 .
Martin G.C 1958 Root-knot nematodes in the federation of Rhodesia and Nyassland Nematologica 3 : 332 – 349 .
McLean H.S Richburg J.S Wilcut J.W Culbreath A.C Branch W.D and Kvien C.K 1994 Peanut variety response to norflurazon Proc. South. Weed Sci. Soc 47 : 33 .
Mixon A.C and Branch W.D 1985 Agronomic performance of a Spanish and runner cultivar harvested at six different digging intervals Peanut Sci 12 : 50 – 55 .
Mozingo R.W Coffelt T.A and Wright F.S 1991 The influence of planting and digging dates on yield, value, and grade of four Virginia-type peanut cultivars Peanut Sci 18 : 55 – 62 .
Nurse R.E Sikkema P.H and Robinson D.E 2011 Weed control and sweet maize (Zea mays L.) yield as affected by pyroxasulfone dose Crop Protect 30 : 789 – 793 .
Odero D.C and Wright A.L 2013 Response of sweet corn to pyroxasulfone in high-organic matter soils Weed Technol 27 : 341 – 346 .
Prostko E.P 2013 Weed control update Pages 47-65 In 2013 Peanut production update. Cooperative Extension Service Series CSS-12-0110, ed. J.P. Beasley, Jr., Athens, GA : University of Georgia . Pages 131 .
Prostko E.P Grey T.L Webster T.M and Kemerait R.C 2011 Peanut tolerance to pyroxasulfone Peanut Sci 38 : 111 – 114 .
Sholar J.R Mozingo R.W and Beasley J.P 1995 Peanut cultural practices, Pages 354-382 In H. E. Pattee and H.T. Stalker, eds. Advances in Peanut Science, Stillwater, OK ; American Peanut Research and Education Society . Pages 566 .
Sparks O.C Barrentine J.L Burgos N.R and McClelland M.R 2003 Effect of Palmer amaranth seedbank density on the performance of pendimethalin and fluometuron Sum. Ark. Cotton Res. Ser 521 : 167 – 172 .
Tanetani Y Kaku K Kawai K Fujioka T and Shiminizu T 2009 Action mechanism of a novel herbicide, pyroxasulfone Pestic. Biochem. Physiol 95 : 47 – 55 .
Walker R.H Wells L.W and McGuire J.A 1989 Bristly starbur (Acanthospermum hispidium) interference in peanuts (Arachis hypogaea) Weed Sci 37 : 196 – 200 .
Webster T.M 2005 Weed survey – southern states: broadleaf crops subsection Pages 291-306 In 2005 Proc. South. Weed Sci. Soc .
Webster T.M Cardina J and Loux M.M 1998 The influence of weed management in wheat stubble on weed control in corn Weed Technol 12 : 522 – 526 .
Wilcut J.W York A.C and Wehtje G.R 1994 The control and interaction of weeds in peanut (Arachis hypogaea) Rev. Weed Sci 6 : 177 – 205 .
Wilcut J.W York A.C Grichar W.J and Wehtje G.R 1995 The biology and management of weeds in peanut, Pages 207-224 In H. E. Pattee and H.T. Stalker, eds. Advances in Peanut Science, Stillwater, OK ; American Peanut Research and Education Society . Pages 566 .
Williams E.J and Drexler J.S 1981 A non-destructive method for determining peanut pod maturity Peanut Sci 8 : 134 – 141 .
Wise A.M Grey T.L Prostko E.P Vencill W.K and Webster T.M 2009 Establishing the geographic distribution level of acetolactate synthase resistance of Palmer amaranth (Amaranthus palmeri) accessions in Georgia Weed Technol 23 : 214 – 220 .
York A.C and Coble H.D 1977 Fall panicum interference in peanuts Weed Sci ., 43 – 47 .
Young J.H Person N.K Donald J.O and Mayfield W.H 1982 Harvesting, curing, and energy utilization, Pages 458-487 In H.E. Pattee and C.T. Young, eds. Peanut Science and Technology, Amer. Peanut Res. Educ. Soc. , Yoakum, TX . Pages 566 .
Notes
- First, second, and third authors: Graduate Research Assistant, Professor, Graduate Research Assistant Crop and Soil Sciences, University of Georgia, Tifton, GA 31793. *Corresponding author's E-mail: pmeure@uga.edu.
Author Affiliations