Introduction
In the Virginia-Carolina (VC) region, peanut (Arachis hypogaea L.) is an important cash crop grown annually on over 50,000 ha. The virginia-type is the predominant peanut market type grown in this region. Desirability of the virginia vs. other peanut types, e.g., runner, spanish, and valencia, is related to its large pod and kernel size, and the in-shell and gourmet industries that evolved in the VC region around this particular type of peanut. Virginia-type peanuts have almost twice the kernel size of the runner, spanish, or valencia-type for which farmers receive premiums. For example, for an average $355 per ton load of farmer stock, the farmer receives about $15–18 of premium based on the extra large kernels (ELK) of the virginia-type. However, the production of the large kernelled-type peanut requires additional costs as compared with runner, spanish, and valencia-type peanut. For example, virginia-type peanut requires more soil moisture for germination then runner and spanish-types (Pallas et al., 1977); this implies higher costs from extra seed to compensate for germination failure in dry years and when irrigation is not an option, or supplemental costs to irrigate for good stand establishment. Virginia-types also require more calcium than the runner (Gaines et al., 1991) and valencia peanuts (Zharare et al., 2009) to achieve high yield and grade factors, including high ELK content. Traditional production of the virginia-type peanut was first challenged in 1996 when farm legislation eliminated the quota system, reduced the price support, and removed the escalator provision (Brown, 1998). These modifications coupled with rising costs for fuel and pesticides appear to justify expansion of runner production in the VC region in the recent years. For example in 2011, South Carolina produced 91,189 tons of peanuts from which 30% were runners. In the same year North Carolina produced 170,907 tons and Virginia 31,543, from which 11% were runners (USDA-Federal State Inspection Service).
In addition to apparently lower production costs, there are several additional advantages for growing runner-type peanut. For example, it is well established that runner peanut are more productive than virginia-types on poorly drained soils (Batten, 1949), which appears to be an advantage on heavier soils, under conservational tillage, and in rainy years; recently, no-till and strip-till cultivation systems have expanded in Virginia to almost 50% of the peanut acreage. Newly developed runner cultivars have improved yield, high sound mature kernels (SMK) content, disease resistance, and high-oleic fatty acid oil chemistry (Branch and Brenneman, 2008; Branch, 2009, 2010; Culbreath et al., 2008; Gorbet and Tillman, 2009; Tillman and Gorbet, 2009; Tubbs et al., 2011); virginia-type cultivars with high-oleic fatty acid oil chemistry developed for the VC region are not yet available. Some of the recently developed runners have larger pods and kernels than the traditional runners and are comparable for this trait with the virginia-type peanuts (Gorbet and Tillman, 2009). However, the runner-type cultivars are known to have relatively late maturity when compared to the virginia-type for the northern parts of the VC region where early maturity is critical to avoid freeze damage. Their response to disease may also differ because of different disease pressures. Finally, in northern VC region runner-type cultivars may express different yield and grade potentials than in the environments in which they were developed. Therefore, their suitability for the northern VC region needs to be determined by comparison with the virginia-type cultivars that are traditionally grown in this region. Earlier research showed potential for similar economic returns from runners when compared to virginia-type peanut in the VC region (Jordan et al., 2000). However, direct comparison of the runner and virginia-type cultivars in northern VC region needs to be updated to include cultivars currently grown in this region.
The objective of this research was to compare four runner-type cultivars and four virginia-type cultivars for development and maturity, disease, yield, grade factors, pod brightness, and gross value with emphasis on Florida 07 runner and Bailey virginia-type, the most popular cultivars currently grown in the VC region.
Materials and Methods
In 2009 and 2010, four runner and five virginia-type peanut cultivars were evaluated at the Hare Road Farm (36° 39′ N, 76° 44′ W; 18.9 m elevation) of the Tidewater Agricultural Research and Extension Center near Holland, VA. These cultivars include old and new commercial cultivars currently grown in the Southeast and the VC peanut growing regions. Among the runner-type cultivars, Florida 07 (Gorbet and Tillman, 2009) is a widely grown cultivar in the VC region. Georgia Green (Branch, 1996) is an old check runner and it was included for comparisons with the newer cultivars and earlier studies. Cultivars AP-4 (Tillman and Gorbet, 2009) and Georgia-03L (Branch, 2004) are older runner-type cultivars but with excellent performance in earlier studies in Virginia (F. Shokes, personal communication). Also, they mature earlier than most of the runners and are more suited for shorter seasons of Virginia (Phipps and Telenko, 2011). Bailey (Isleib et al., 2011), CHAMPS (Mozingo et al., 2006), and Sugg (Copeland et al., 2010) are virginia-type cultivars predominantly grown in the VC region. Titan (Balota et al., 2011) and Spain (F. Garner, personal communication) are newer extra-large seeded virginia-type cultivars. Each year, cultivars were evaluated in two distinct fields for soil type and available water capacity. One field (F3.5) is classified as Eunola (fine-loamy, siliceous, thermic Aquic Hapludults) and has 3.5 cm of available water capacity in the first 25 cm of soil (USDA-Natural Resources Conservation Service, 14 April 2009). The second field (F2.5) is classified as Kenansville (loamy, siliceous, thermic Arenic Hapludults) and has 2.5 cm of available water capacity in the first 25 cm of soil. This last field was considered more drought prone than F3.5. In F2.5, peanut was grown after 10 yr of cotton monoculture. In F3.5, peanut followed corn in 2009 and cotton in 2010 in a 3 yr rotation of corn, cotton, and peanut. Each year, the same cultural practices were applied to both fields according to Virginia recommendations for production of high yield and quality (Faircloth and Shokes, 2008); and detailed information is described by Balota (2010, 2011). A maximum input approach was used in both fields and years. Thirty eight L/ha Metam sodium was applied 2 wk before planting. Aldicarb [2-methyl-2(methylthio) propionaldehyde] at 1.2 kg ai ha−1 was applied in-furrow at planting for tobacco thrips (Frankliniella fusca (Hinds)) control. Thrips control was maintained during vegetative growth by foliar sprays with 0.58 kg ai ha−1 acephate [O,S-dimethylacetyl-phosphoramidothioate] before flower when a 25% threshold leaflet damage was observed (Herbert, 1999). Chlorpyrifos [o, o-diethylo-(3,5,6,-trichloro-2-pyridinyl) phyosphorothioate] at 2.5 kg ai ha−1 was applied in late June for corn earworm (Helicoverpa zea (Boddie)) control. Fenpropathrin [alpha-cyano-3-phenoxybenzyl 2,2,3,3, -tetramethylcyclopropanecarboxylate] was used in mid August for spider mite (Tetranychus spp.) control at a rate of 0.21 kg ai ha−1. Biweekly applications of fungicides were started in late June to control foliar and soil-borne diseases using 0.15 kg ai ha−1 chlorothalonil (tetrachloroisophtalonitrite), 0.10 kg ai ha−1 prothioconazole 2-[2-(1-chlorocyclopropyl)-3-(2-chlorophenyl)-2-hydroxyprophyl]-1,2-dihydro-3H-1,2,4-triazole-3thione, and 0.21 kg ai ha−1 tebuconazole {alpha-[2-(4-chlorophenyl)ethyl]-alpha-(1,1-dimethylethyl-1H-1,2,4,-triazole-1-ethanol}. To prevent Sclerotinia blight produced by Sclerotinia minor, 0.30 kg ai ha−1 fluazinam {3choloro-N-[3-chloro-2,6-dinitro-4-trifuooromethyl)phenyl]-5-trifuoromethyl-2-pyridamide} was used in late August. Supplemental calcium was applied at 636 kg ha−1 rate as Gypsum 420 (USG Ben Franklin, Chicago, IL) at flowering. Boron liquid fertilizer (Helena Chemical Company, Collierville, TN) was applied pre-plant and at the beginning of seed set at a rate of 1.62 kg ai ha−1. In late July and August, 2.2 kg ai ha−1 manganese was also applied as a fertilizer. Pre- and post-emergence weed control was applied (Wilcut et al., 1995), and all plots received the same herbicide treatments each year. Cultivars were grown in plots of two-9.8 m rows planted on 0.9 m centers. Cultivars were planted on 29 April in 2009 and 4 May in 2010 at a seeding rate of 10.8 seeds m−2. Each year, weather parameters were recorded within 1.6 km from the plots. In each field and year, plots were replicated four times in a randomized complete block design.
The total seedling number within each plot was recorded and converted to the number of seedlings m−2 when all germinated seeds were considered fully emerged. Reproductive stages were determined using definitions by Boote (1982) from “beginning flower” (stage R1) to “beginning maturity” (stage R7) in all fields and years. This was done by visual observations every other day. Stage R1 was defined as one open flower on any plant within a plot. Stage R2 was “beginning peg”, when one elongated gynophore (peg) was observed. Stage R3 was “beginning pod”, when one peg within each plot was found in the soil with a swollen ovary twice the width of the peg. Stage R4 was “full pod”, when one fully expanded pod per plot was found in the soil. Stage R5 was “beginning seed”, when one fully expanded pod with visible cotyledon growth inside was found in the soil of each plot. Stage R6 was “full seed”, when one pod per plot was fully filled by the seeds. Stage R7 was “beginning maturity”, when one pod within each plot was showing natural coloration of the inner pericarp. The growth stage occurrence dates were recorded and transformed to the number of days after planting (DAP) for each growth stage. In 2010, pod maturity was determined by the pod mesocarp color developed by Williams and Drexler (1981). Five plants per plot were collected a week before harvest and approximately 150 pods removed. The pod exocarp was then removed through high pressure washing. Pods were placed on a maturity board and grouped by their mesocarp color into white, yellow, orange, brown, and black color groups and the number of pods within each group was counted and then converted to a percentage. The profile maturity classes by Williams and Drexler (1981) defines white and yellow as immature, and orange, brown and black as mature pods; orange is defined as incipient maturity, brown full maturity, and black over mature pods. Incidence of tomato spotted wilt virus (TSWV) (Bunyaviridae, Tospovirus) was evaluated in early September, and Sclerotinia blight close to harvest each year. Incidence for TSWV was determined by counting the number of symptomatic plants per plot (plot area was 17.6 m2). Incidence of Sclerotinia blight was determined by counting the number of infection centers in each plot. An infection center was a point of active growth by Sclerotinia minor. Peanut was harvested in the first week in 2009 and the second week of October in 2010 with conventional peanut harvest equipment. Pod yield was determined from the plot weight and adjusted for 7% seed moisture. A 500 g pod sample was then retained from each plot to determine pod brightness and grade characteristics, i.e., % ELK, % SMK, % damaged kernels (DK), % sound split kernels (SS), and % total meat (TM). Jumbo and fancy pod brightness was measured with a colorimeter (model D25LT, Hunter Associates Laboratory, Inc, Reston, VA) and expressed as Hunter L scores. Hunter L is a measure of pod albedo with a theoretically black pod scoring 0 and pure white as 100. Very bright peanut pods score over 50, bright pods range from 45 to 50, and dark pods score below 40. Usually, fancy pods are less bright than the jumbo pods. Growers do not receive premiums for jumbo and fancy pods, i.e., pods large enough to not pass a 13.5 mm spacing on the pre-sizer. However, pod brightness is very important for the in-shell peanut market. Extra large kernels are kernels that do not pass a 25.4 × 8.5 mm screen. There is a premium of 0.039¢ kg−1 for each % ELK. Sound mature kernels are defined as kernels that do not pass 25.4 × 6 mm screen for virginia and 19.05 × 6.4 mm screen for runner type peanut. Each % increase in SMK increases peanut value by 0.5¢ kg−1. Sound splits are undamaged halves of kernels for which there is no penalty up to 4%. Damaged kernels are inedible peanut kernels due to decay, mold, insect damage, sprouting, freeze damage, and skin discoloration. When DK exceeds 2.4%, seed is considered segregation 2 and growers receive only 35% of the gross value for that peanut. Total meat includes ELK, SMK, SS, and DK. Gross value was calculated from the federal formula and it is based on a combination of the grade factors and pod yield.
The effects of genotype, field, year, and their interaction on the measured traits were assessed with factorial ANOVA from the GLM procedure of SYSTAT® 10.2 (2002, SYSTAT Software Inc, Richmond, CA). Means were separated by Fisher's Protected LSD test at P = 0.05.
Results
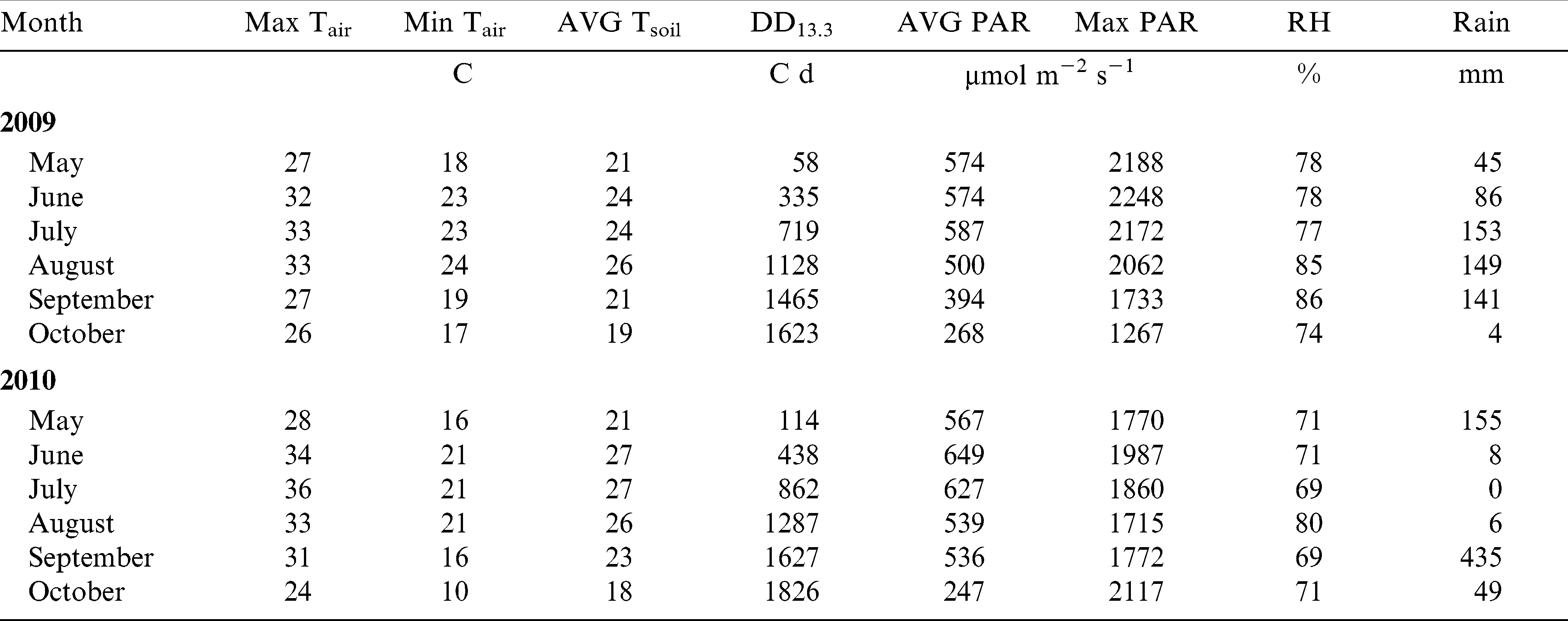
Years were contrasting for the weather conditions during peanut growing season (Table 1). For example, even though average air temperature was 24 C in both years, max monthly average temperatures in 2010 were 1 to 4 C higher than in 2009 from May through September. Air RH averaged 80% in 2009 and 72% in 2010. Almost no precipitation was available to the crop in June, July, and August in 2010; therefore 24.5 mm of irrigation was applied on 22 June, 7 July, and 21 July to both fields (73.5 mm total). In contrast, during the same period in 2009, rainfall totaled 388 mm. In summary, 2009 was wet and cool while 2010 was dry and hot. However, precipitation was adequate for germination and emergence in both years (Table 1). Both years had sufficient heat units for peanut maturity and optimum light conditions for photosynthesis (Pallas and Samish, 1974) and growth.
Air and soil temperature at 10 cm depth, peanut heat units (degree day – DD13.3) calculated based on a 13.3 C temperature base, daylight (photosynthetic active radiation – PAR), air relative humidity (RH), and precipitation at Tidewater AREC, Suffolk VA in the 2009 and 2010 growing seasons.
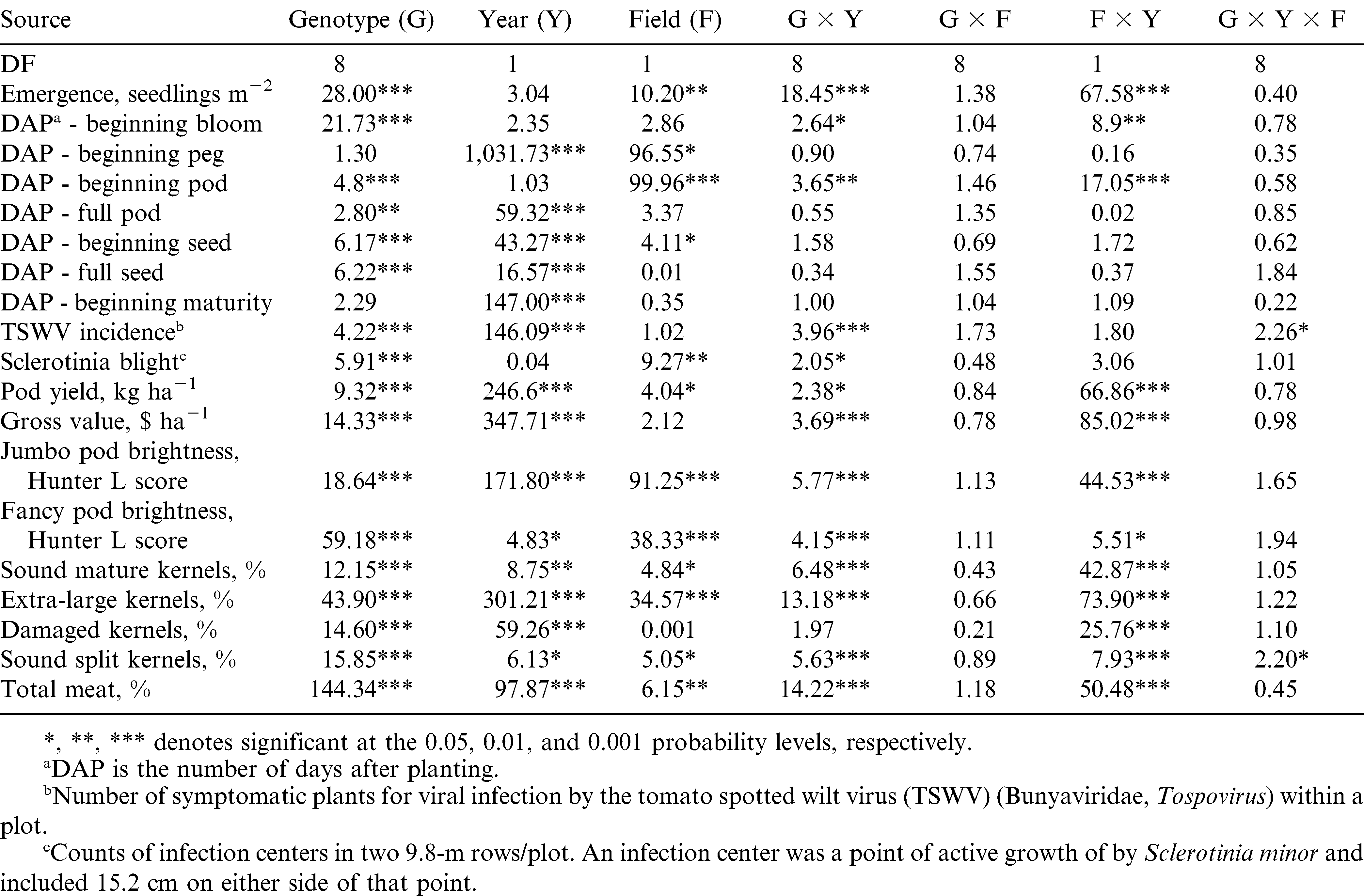
Combined analysis of variance for the effect of genotype, field, year, and their interactions showed that genotype had a significant (P ≤ 0.01) effect on disease, agronomic, and growth factors analyzed in this experiment excepting the number of DAP to beginning peg and maturity (Table 2). For the number of DAP until beginning maturity, genotype did not have a significant effect. Also, genotype had a significant (P ≤ 0.001) effect on the gross value. Year had a significant effect (P ≤ 0.05) on all traits excepting emergence, beginning bloom and pod, and Sclerotinia Blight incidence. The genotype × year interaction was significant (P ≤ 0.05) for seedling emergence, beginning bloom and pod, disease, gross value, and all agronomic and grade factors, excepting the content of DK. Field had a significant effect (P ≤ 0.05) on emergence and beginning bloom and pod, Sclerotinia blight incidence, and the majority of agronomic and grade traits, i.e., pod yield and brightness, and the content of SMK, ELK, SS, and TM; but it did not influence the gross value of the crop (Table 2). On average, F3.5 had improved yield and grade factors than F2.5, but incidence of Sclerotinia blight was higher in this field. In F2.5, peanut plants demonstrated more rapid development and produced brighter pods, and more jumbo and fancy pods than in F3.5. The interaction of genotype × field was not significant for any of the traits, for which data from both fields within each year were combined for further genotypic mean separation. The year × field interaction was significant for gross value, all agronomic and grade traits, emergence, and beginning bloom and pod development because in the wet year of 2009 plants performed better in the drier soil (F2.5) than in the wet soil (F3.5). The situation was reversed during the dry year of 2010. With the exception of TSWV incidence and the SS kernel content, the genotype × year × field interaction was not significant for any other trait (Table 2), suggesting a strong genetic effect on these traits.
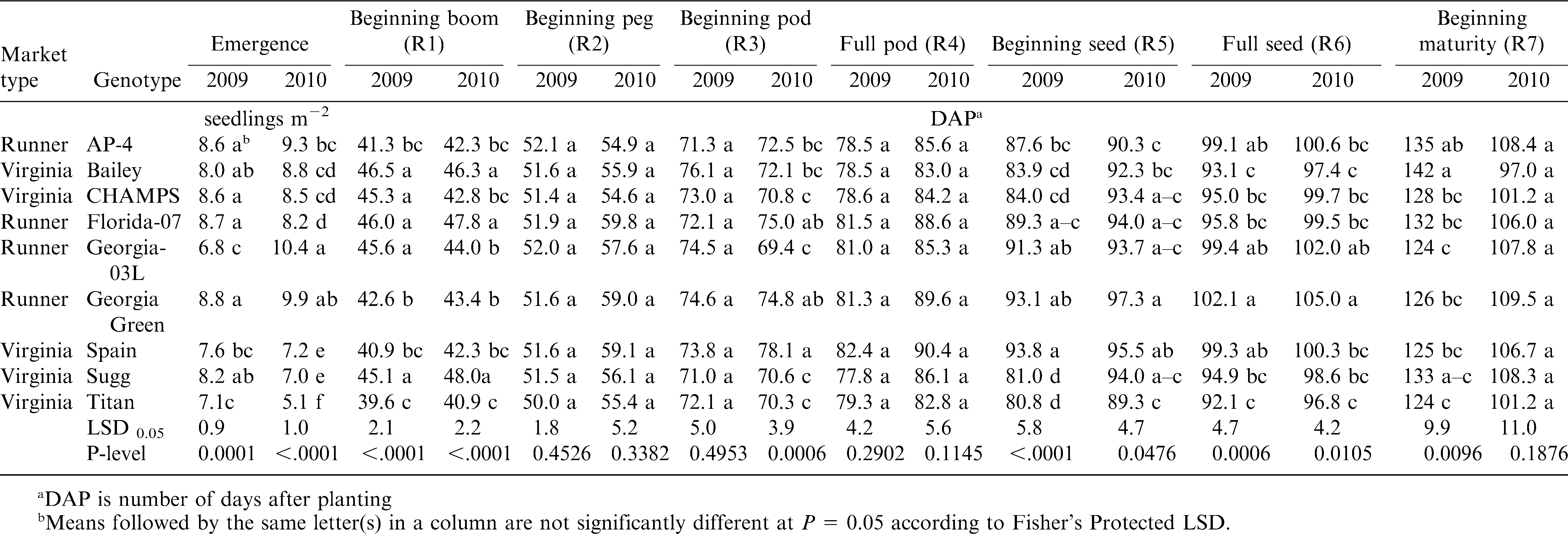
The influence of year and genotype on the growth stages of runner and virginia-type cultivars is presented in Table 3. The large seeded cultivars, Spain and Titan, had a significantly lower number of peanut plants m−2 than the smaller seeded cultivars. Because soil moisture at planting was good in both years and because all seed had over 85% germination, it is assumed that lower germination resulted from seed damage during planting. Indeed, planting very large kernelled cultivars requires special plates and adjustments to the planter (M. Balota, unpublished data). Sugg also showed a tendency for lower emergence and in particular during the dry year of 2010 when compared with the other runners and virginia-type cultivars. Beginning bloom was approximately 44 DAP in both years with insignificant differences due to the market type. Beginning peg was at 51.5 DAP in 2009 and 56.9 DAP in 2010, with no significant differences among genotypes. Beginning pod was approximately at 73 DAP in both years. Full pod growth stage was at 80 DAP in 2009 and 86.5 DAP in 2010. Beginning and full seed stages were also delayed by the dry year 2010 by 3 to 6 d depending on the genotype (Table 3). Beginning maturity was at 128.5 DAP in 2009 and 105 in 2010; in 2009 genotypes were significantly (P ≤ 0.01) different for this trait. Some pods showed earlier coloration of the inner pericarp, noted as beginning seed maturity, in 2010 vs. 2009. But in general, seed maturity was delayed in 2010 vs. 2009, probably as a result of drought and heat in 2010, and this was shown by a high percentage of orange pods, almost double compared with brown and black pods at a week from harvest when maturity was determined by the pod mesocarp color (data not shown).
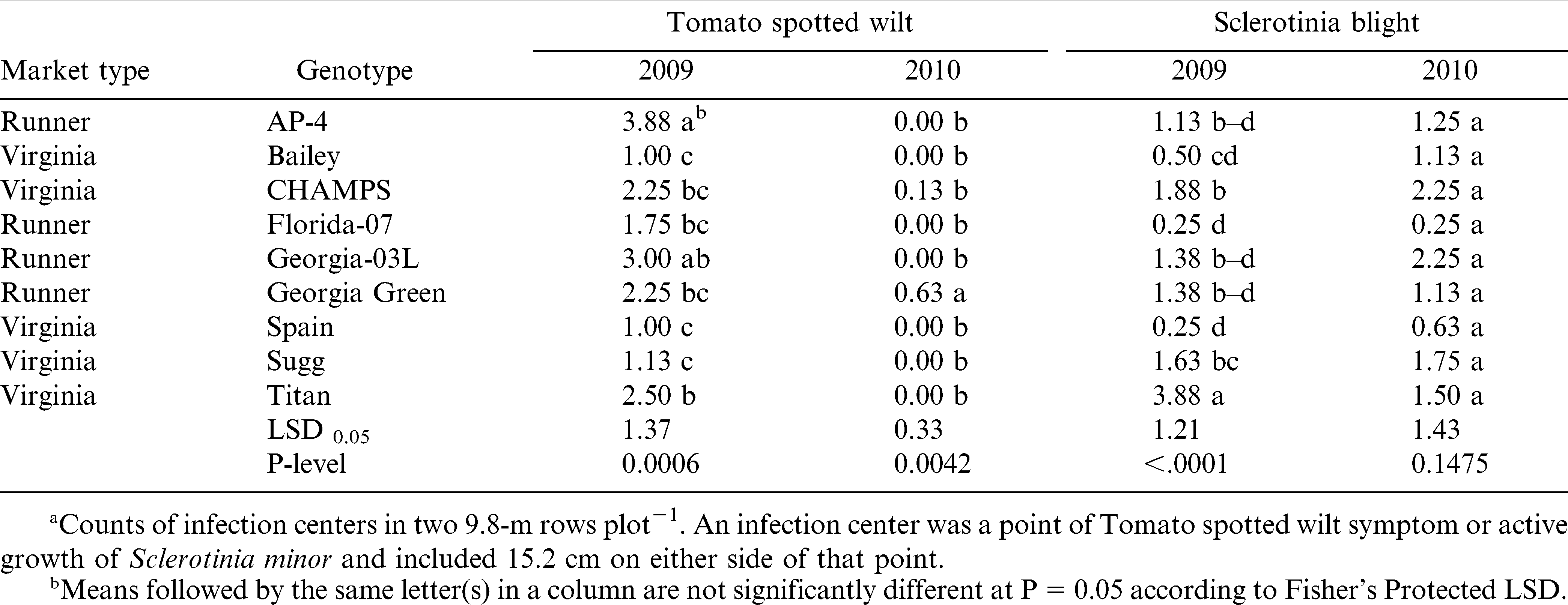
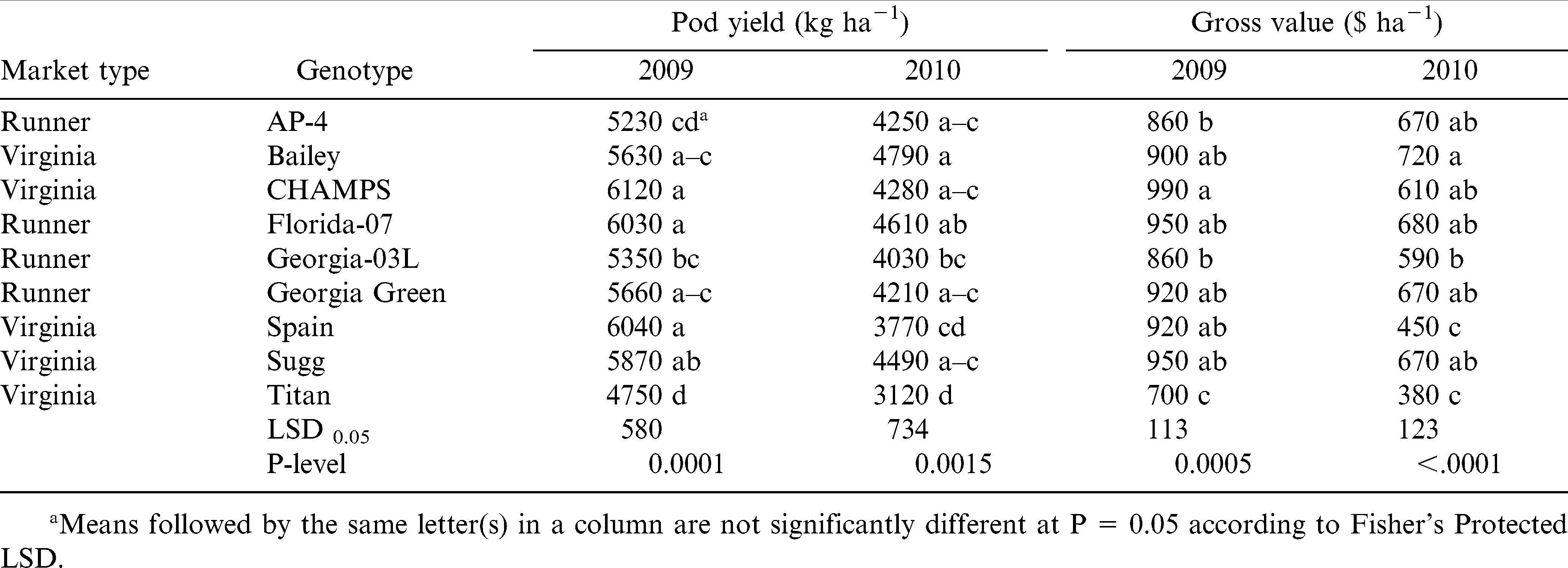
Disease information is presented in Table 4. Even though differences among genotypes were statistically significant, incidence of TSWV and Sclerotinia blight was low with less than 1% of the plants showing disease symptoms in both fields and years. This demonstrates good disease control by the fungicide program used in this experiment. Under low disease pressure, pod yield and gross value were significantly different among genotypes with no prominent effect by the market type (Table 5). The cultivar that performed consistently well in both years was Florida-07, whereas the worst performing cultivars were Titan and Georgia-03L. Sugg had high yields in both years, but value was high only in 2009. The highest yield and gross value in 2009 was for CHAMPS and in 2010 for Bailey. AP-4 ranked third for the gross value in 2010 even with low yield, which denotes good grading traits and suitability for northern Virginia environment; similarly in 2009, gross value of AP-4 was high when yield was among the lowest. This is in agreement with earlier studies by Phipps and Telenko (2011). Spain yielded similar to Florida-07 in 2009, but it was the second lowest for yield and value in 2010 (the dry year).
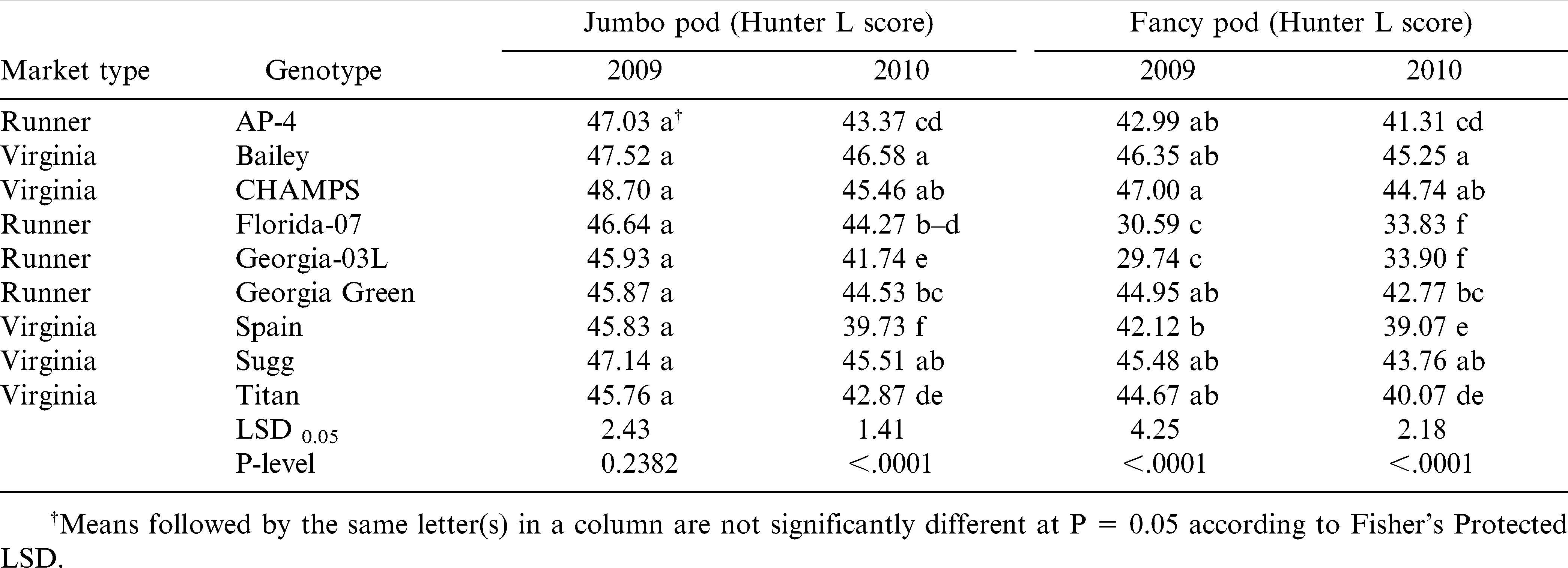
The jumbo pod brightness was not significantly different among genotypes in 2009 when all scored over 45, which denotes bright pods. However, genotypes were significantly different for the brightness of jumbo pods in 2010 and for brightness of fancy pods in both years (Table 6). Consistent over years, Bailey, CHAMPS, and Sugg were the brightest for the jumbo and fancy pods. Spain was less bright for the jumbo and fancy pods in 2010, while Florida-07 and Georgia-03L were the least bright for the fancy pods in both years (Table 6). Peanut market type had a significant effect on pod brightness with runners being less bright than virginia-type for the jumbo (P ≤ 0.01) in 2009 and fancy pods (P ≤ 0.001) in both years.
There were significant differences among genotypes for all grading characteristics, i.e., content of SMK, ELK, DK, SS, and TM, in both years (Table 7). CHAMPS, Bailey, and Sugg had the highest and Titan, Florida-07, and Spain the lowest content of SMK in 2009. In 2010, AP-4 and Georgia Green had the highest SMK content, and Spain, Sugg, and Florida-07 the lowest. As expected, the ELK content was greater for virginia then for runner cultivars in both years with top performers being Spain, Sugg, Titan, and CHAMPS, and lowest performers being Georgia Green, AP-4, and Georgia-03L. The content of DK was in excess of 2.4% for Spain and Titan in 2009 and 2010. In 2010, DK was over 2.4% for all virginia-type cultivars and Florida-07, a big seeded runner. High content of SS kernels were recorded each year for Georgia Green, AP-4, Florida-07, and Sugg; the least SS content was observed for Spain in both years. Finally, TM content was highest for the smallest seeded runners, AP-4 and Georgia Green, and lowest for the biggest seeded virginia-type, Titan and Spain (Table 6).
Discussion
Our data support earlier findings by Jordan et al. (2000) showing that runner-types can provide similar gross returns with virginia-type peanut cultivars when grown in the northern VC region. Florida-07, the most popular runner cultivar currently grown in the VC region (F. Garner, personal communication) had a comparable high yield (6034 kg ha−1) with CHAMPS (6115 kg ha−1) and Sugg (6042 kg ha−1), two virginia-type cultivars, in 2009; these yielded the highest among all genotypes in this experiment. Similarly in 2010, Florida-07 (4609 kg ha−1) was the second after Bailey (4793 kg ha−1) for peanut pod yield and gross value. Florida-07 had also large kernels and ELK content comparable with virginia cultivars (Table 7), but pod brightness was significantly lower (Table 6). This denotes that Florida-07 may be not as suitable as the virginia-type cultivars for the in-shell peanut markets. The lowest yielding cultivars were Titan (4745 kg ha−1) among virginia-type, and AP-4 (5231 kg ha−1) and Georgia-03L (5349 kg ha−1) among the runners. However, AP-4 exhibited good grading characteristics and gross values in both years in Virginia.
Runner and virginia-type cultivars were consistently different from each other only for three traits, including: total meat content (P ≤ 0.01, in both years), fancy pod brightness (P ≤ 0.001 in both years), and DAP to reach full seed (P ≤ 0.001 in 2009 and P ≤ 0.01 in 2010). Average total meat was 74.2% for runners and 70.4 for virginia, fancy pod brightness was 38% for runners and 44.3% for virginia, and the number of DAP to reach full seed was 100.4 d for runners and 96.1 d for the virginia-type cultivars. For all other traits differences among the two groups were dependent upon season. Indeed, runners appeared to have an advantage over virginia-type cultivars in dry years. For example in 2010, runners significantly out-performed virginia cultivars for all grading factors in both fields, therefore the gross return from runners ($652 ha−1) as a group was significantly higher than for virginia ($565 ha−1) cultivars. Runners AP-4 ($670 ha−1), Florida-07 ($682 ha−1) and Georgia Green ($666 ha−1), and virginia cultivars Bailey ($715 ha−1) and Sugg ($666 ha−1) had the highest gross value in 2010, the dry year. Both runner and virginia cultivars began blooming at approximately 43 DAP in 2009 and 44 DAP in 2010. There were no differences statistically until after full pod stage, approximately 80-85 DAP, when runners were from a few days to 2 wk later than virginia-type cultivars, depending on the season. For example, 2 yr average was 125 DAP to beginning maturity for AP-4, 129 for Florida-07, 119 for Georgia-03L, and 120 for Georgia Green. For virginia-type cultivars, these numbers were 119 DAP for Bailey, 116 for CHAMPS, 116 for Spain, and 114 for Titan; Sugg was as late as the runners with 125 DAP to beginning maturity. The pod mesocarp color evaluation close to harvest in 2010 revealed no significant differences among cultivars for maturity when orange, brown, and black pods were combined; the range was from 88.5% for AP-4 to 96% for Titan. However, when only the brown and black pods were considered, significant differences among cultivars (P ≤ 0.01) were observed. Ranges of black and brown pods were from 20 to 34% for runners and from 24 to 44% for virginia types with the exception of Spain, which appeared to be late maturing in 2010. Indeed, runners had significantly (P ≤ 0.05) higher percentage of immature pods (11.4%), i.e., white and yellow, than virginia-type cultivars (5.8%); the content of orange pods was also more for runners than for virginia-type cultivars.
These data were obtained under high input crop management in terms of pest control, micronutrients, and calcium applications and therefore reflect production potential of the genotypes for this experiment. Under conservative input production, results could change. None the less, testing for yield and grade potential, and maturity within the available heat units in the northern VC region, is an important step for a research-based selection of the most suitable runner and virginia-type cultivars for this region. Based on results from this experiment, Florida-07 was the most productive runner, comparable with Bailey, CHAMPS, and Sugg, but attention needs paid given to pod brightness deficiency and longer season requirement when choosing to grow this cultivar.
Acknowledgements
This work was supported with funding from the National Peanut Board, Virginia Peanut Board, Virginia Agricultural Council, and Virginia Crop Improvement Association.
Literature Cited
Balota M 2010 Peanut crop physiology related projects at Tidewater Agricultural Research and Extension Center - 2009 Virginia Coop. Ext. Publ. 3009-1460. http://pubs.ext.vt.edu/3001/3001-1432/3009-1460.html .
Balota M 2011 Peanut crop physiology related projects at Tidewater Agricultural Research and Extension Center - 2010 Virginia Coop. Ext. Publ. 3101-1532. http://pubs.ext.vt.edu/3101/3101-1523/3101-1523.html .
Balota M Mozingo W.R Coffelt T.A Isleib T.G Beahm B.R Pittman H.G Bryant F.S Copeland P.A Daughtrey C.J Kennedy B.C Shokes F.M Ashburn R.D Whitt D.L and Redd D.A 2011 Registration of ‘Titan’ peanut J. Plant Registrations 5 : 282 – 288 .
Batten E.T 1949 Peanut production Southern Planter 110 : 16 – 7 .
Boote K.J 1982 Growth stages of peanut (Arachis hypogaea L.) Peanut Sci. 9 : 35 – 40 .
Branch W.D 1996 Registration of ‘Georgia Green’ peanut Crop Sci. 36 : 806 .
Branch W.D 2004 Registration of ‘Georgia-03L’ peanut Crop Sci. 44 : 1485 – 1486 .
Branch W.D and Brenneman T.B 2008 Registration of ‘Georgia-07W’ peanut J. Plant Registrations 2 : 88 – 91 .
Branch W.D 2009 Registration of ‘Georgia-08V’ peanut J. Plant Registrations 3 : 143 – 145 .
Branch W.D 2010 Registration of ‘Georgia-09B’ peanut J. Plant Registrations 4 : 175 – 178 .
Brown B.A 1998 1998 outlook and situation, pp. 1 – 4 In 1998 Peanut Information. North Carolina Coop. Ext. Serv. Publ. Ag-331.
Copeland S Isleib T Milla-Lewis S Shew B Hollowell J Pattee H Sanders T Dean L Hendrix K Balota M and Chapin J 2010 Release of ‘Sugg’ Virginia-type Peanut Cultivar In: Proceedings of the American Peanut Research and Education Society, Vol 42, pp. 81 – 82 , Clearwater, FL .
Culbreath A.K Tillman B.L Gorbet D.W Holbrook C.C and Nischwitz C 2008 Response of new field-resistant peanut cultivars to twin-row pattern or in-furrow applications of phorate for management of spotted wilt Plant Disease 92 : 1307 – 1312 .
Faircloth J.C and Shokes F.M 2008 Agronomic recommendations and procedures In Virginia Peanut Guide Faircloth J.C VPI&SU/Va. Agric. Exp. Stn./TAREC, 74 pp.
Gaines T.P Parker M.B and Walker M.E 1991 Limestone and gypsum effects on calcium nutrition of ‘Florunner’ and ‘NC-7’ peanuts Comm. Soil Sci. and Plant Analysis 22 : 117 – 135.
Gorbet D.W and Tillman B.L 2009 Registration of ‘Florida-07’ peanut J. Plant Registrations 3 : 14 – 18 .
Herbert D.A 1999 Insect control in peanut, pp. 38 – 52 In 1999 Virginia Peanut Production Guide. Tidewater Agric. Res. and Ext. Center Info. Series 416, Suffolk, VA.
Isleib T.G Milla-Lewis S.R Pattee H.E Copeland S.C Zuleta M.C Shew B.B Hollowell J.E Sanders T.H Dean L.O Hendrix K.W Balota M and Chapin J.W 2011 Registration of ‘Bailey’ peanut J. Plant Registrations 2011, 5 : 27 – 39 .
Jordan D.L Swann C.W Spears J.F Brandenburg R.L Bailey J.E and Tucker M.R 2000 Comparison of virginia and runner market-type peanut (Arachis hypogaea) grown in the Virginia-Carolina production region Peanut Sci. 27 : 71 – 77 .
Mozingo R.W Coffelt T.A Phipps P.M Coker D.L Machado S and Petrie S.E 2006 Registration of ‘CHAMPS’ peanut Crop Sci. 46 : 2711 – 2712 .
Pallas J.E and Samish B.Y 1974 Photosynthetic response of peanut Crop Sci. 14 : 478 – 482 .
Pallas J.E Stansell J.R and Bruce R.R 1977 Peanut seed germination as related to soil water regime during pod development Agron. J. 69 : 381 – 383 .
Phipps P.M and Telenko D.E.P 2011 Comparison of virginia- and runner-type peanut cultivars, pp. 132 In Phipps M.P and Telenko D.E.P (eds) Applied research on field crop disease control VPI&SU/Va. Agric. Exp. Stn./TAREC, Information Series No. 500.
Tillman B.L and Gorbet D.W 2009 Registration of ‘AP-4’ peanut J. Plant Registrations 3 : 138 – 142 .
Tubbs R.S Beasley J.P Culbreath A.K Kemerait R.C Smith N.B and Smith A.R 2011 Row pattern and seeding rate effects on agronomic, disease, and economic factors in large-seeded runner peanut Peanut. Sci. 38 : 93 – 100 .
Wilcut J.W York A.C Grichar W.J and Wehtje G.R 1995 The biology and management of weeds in peanut (Arachis hypogaea), pp. 2007 – 244 In Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science Amer. Peanut Res. Educ. Soc. , Stillwater, OK .
Williams E.J and Drexler J.S 1981 A non-destructive method for determining peanut maturity Peanut Sci. 8 : 134 – 141 .
Zharare G.E Asher C.J and Blamey F.P.C 2009 Calcium nutrition of peanut growth in solution culture. I. Genetic variation in Ca requirements for vegetative growth J. Plant. Nutr. 32 : 1831 – 1842 .
Notes
- Dept. of Plant Pathology, Physiology, and Weed Science, Virginia Tech University, Tidewater Agriculture Research and Education Center, 6321 Holland Road Suffolk, Virginia USA 23437. * Corresponding author's e-mail: mbalota@vt.edu.
Author Affiliations