Introduction
Timely overhead sprinkler irrigation following an application of systemic fungicide can play a role in integrated management of stem rot (caused by Sclerotium rolfsii) by redistributing fungicide residuals from the upper to the lower peanut (Arachis hypogaea) canopy, where S. rolfsii infection and disease development occur. Previous research by Woodward (2006) showed that an 18-h period between fungicide application and an overhead sprinkler irrigation event would provide effective control of both foliar diseases and stem rot, as irrigating very early after a fungicide application could compromise control of early leaf spot (caused by Cercospora arachidicola) and late leaf spot (caused by Cercosporidium personatum). Another effective management strategy for stem rot is night applying peanut fungicides. Recent research has shown that applying systemic fungicides at night not only increased fungicide residual activity in the lower canopy, but improved stem rot control and augmented yield (Augusto et al., 2010a; Augusto et al., 2010b). Peanut plants open their leaves during the day but fold them at night resulting in a sparser canopy (Augusto et al., 2010a). The interaction of applying fungicides at night when peanut leaves are folded and applying irrigation after those sprays have been made has not been investigated.
In Georgia, approximately 55% of the peanut acreage is irrigated (Kemerait, 2008), primarily utilizing overhead center-pivot irrigation systems. Although the peanut growing region in Georgia is relatively humid (Stansell et al., 1976; Truman et al., 2007), the distribution of rainfall is not always predictable and severe drought periods may occur during the season. In addition, the soils have a relatively sandy texture (Truman et al., 2007) with low water-holding capacity and irrigation is often needed to ensure high yield and seed quality. Drought stress sensitivity varies with growth stage of the plant, and this may vary with peanut market type as well. Based on water response curves for the typical runner peanut cultivars grown in Georgia, 78% of the 584.2 mm of water required per season is needed between 70 and 120 days after planting (DAP) of the 140-day season (Beasley, Jr., 2010; Harrison, 2009). Much of this time period corresponds with the timing of key fungicide applications for stem rot management that occur from 60 to 100 DAP. Irrigation scheduling methods have evolved with time and range from computer-based models, in-field water balance, to rainfall and soil moisture monitoring (Beasley, Jr., 2007; Harrison, 2009). However, the amount and timing of irrigation are often reliant upon grower experience and costs (Harrison, 2009).
Other aspects of the relationship between irrigation water and peanut diseases have been documented (Alderman and Beute, 1987). Overhead irrigation has been shown to increase humidity around the leaf surface, prolong leaf wetness from natural dew, and promote sporulation of early and late leaf spots (Lyle, 1964; Smith and Crosby, 1973), two damaging peanut diseases. The spores of C. arachidicola on the upper leaf surface are predisposed to washing off by sprinkler irrigation from the upper leaves to the lower canopy where, with high humidity, they increase infection and disease intensity. The resulting disease lesions are then an important source of inoculum for secondary infections of the whole plant. Stem rot is also favored by high soil moisture from irrigation. This disease has surpassed Tomato Spotted Wilt Virus as the most important peanut disease since 2006 based upon damage and cost of control (Kemerait, 2006; Kemerait, 2007; Kemerait, 2008; Kemerait et al., 2010). Although stem rot often occurs in rainfed peanut (Watkins, 1961), irrigation can exacerbate the problem (Davis et al., 1996). High soil moisture promotes infection and fungal mycelial spread between and within plants, especially in dense stands resulting from use of high seeding rates (Punja, 1985; Sconyers et al., 2005; Augusto et al., 2010). Infection of S. rolfsii is limited to basal stems, roots, pegs and pods, and colonization of the tissues coincides with beginning peg and beginning pod [R2 and R3 as defined by Boote (1982)] when peanut branches spread rapidly across the soil (Augusto et al., 2010a).
Most of the irrigated peanut in Georgia is treated with fungicides approximately seven times during the season to control leaf spot and soilborne diseases, but rainfed peanut usually receives fewer applications (Kemerait, 2008). Fungicide applications are initiated approximately 30 DAP and subsequent applications follow a 14-day interval. Chlorothalonil is typically sprayed at applications 1, 2, and 7 in the seven-application regime to control leaf spots (Culbreath et al., 2002). Since chlorothalonil is a broad spectrum protectant fungicide with no curative activity, it is most effective when applied prior to infection (Culbreath et al., 2001). Chlorothalonil may also be applied in alternating applications, alternating blocks of applications, or in application regime mixtures with other fungicides to prevent late-season or secondary infections, and to reduce the risk of developing resistance in C. arachidicola or C. personatum populations to systemic fungicides (Kemerait et al., 2006). Because chlorothalonil has no activity against stem rot (Brenneman et al., 1991), fungicides with some systemic activity such as quinone outside inhibitors (QoI) (azoxystrobin, fluoxastrobin, and pyraclostrobin), sterol biosynthesis inhibitors (SBI) (tebuconazole and prothioconazole), or benzamides (flutolanil) are applied to control the disease (Kemerait et al., 2010). Prothioconazole is registered in a triazole mixture with tebuconazole as a foliar fungicide to control leaf spots and stem rot (Kemerait et al., 2010). Although tebuconazole remains effective against stem rot, leaf spot isolates with resistance to this fungicide have been reported (Culbreath et al., 2005; Stevenson and Culbreath, 2006; Stevenson et al., 1999). Flutolanil is not active against leaf spots (Culbreath et al., 1992), but a formulation mixture of flutolanil and propiconazole provides control of stem rot and leaf spots (Kemerait et al., 2010). High rates of pyraclostobin may be needed for effective control of stem rot, but it is the most effective currently labeled fungicide for leaf spot control even at lower rates (Kemerait et al., 2010). Azoxystrobin and fluoxastrobin are applied at beginning pod (50–60 DAP) and at beginning seed (80–90 DAP) corresponding to applications 3 and 5 in the seven-application regime to control both stem rot and leaf spots (Kemerait et al., 2010). Flutolanil + propiconazole, tebuconazole, prothioconazole + tebuconazole, and pyraclostrobin are usually applied in a four-block application program corresponding to applications 3 to 6 (Kemerait et al., 2010).
The objective of this study was to determine the interactive effects of fungicide application timing and post-spray sprinkler irrigation on disease control and peanut yield with commercial use patterns of the most commonly used peanut fungicides.
Materials and Methods
Location, experimental design, and treatments
Field experiments were conducted at the Blackshank Farm, University of Georgia Tifton Campus during the 2008 and 2009 growing seasons to evaluate commonly used peanut fungicides applied at night versus day under post-spray irrigated or nonpost-spray irrigated conditions for stem rot and leaf spot control and benefits on yield. The soil type was a Tifton loamy sand, 2 to 5% slope (Calhoun, 1983). The experiments were established in an area with history of continuous peanut cultivation and leaf spot and stem rot prevalence. The field was plowed to a depth of 20 to 25 cm, disk harrowed, and marked off in beds 1.83 m wide during April each year. In 2008, the field was fumigated with nematicide 1,3-dichloropropene (Telone II; Dow AgroSciences, Indianapolis, IN) at 91 kg a.i./ha before planting on 21 April. The preemergence herbicides ethalfluralin (Sonalan; Dow AgroSciences) at 0.72 kg a.i./ha and s-metolachlor (Dual Magnum; Syngenta Crop Protection, Inc., Greensboro, NC) at 1.5 kg a.i./ha were applied and incorporated within the upper 5 cm of soil with a rototiller on 21 April 2008 and 30 April 2009.
The cv. Georgia Green (Branch, 1996) was planted with a two-row Monosem planter at 23 seeds/m on 6 May 2008 and 8 May 2009. Aldicarb (Temik 15G; Bayer CropScience, Research Triangle Park, NC) was applied in furrow (0.67 kg a.i./ha) at planting and on a 0.3-m band (1.68 kg a.i./ha) approximately 70 DAP to control thrips and nematodes, respectively. The postemergence herbicide imazapic (Cadre 70 DF; BASF Corp., Research Triangle Park, NC) at 0.07 kg a.i./ha was applied with herbicide surfactant paraffinic petroleum oil at 1.94 kg a.i./ha + surfactant blend at 0.40 kg a.i./ha (Crop oil; Van Diest Supply Co., Webster City, IA) approximately 60 DAP.
The experimental design was a split-split-plot and treatments were replicated five times. Each sub-sub-plot was a two-row bed, 7.62 m long, and row spacing was 0.91 m. The whole-plot factor was post-spray irrigation and plots were either not irrigated, or sprinkler irrigated with 2.54 cm of water every week regardless of rainfall (unless it was equivalent to or in excess of 2.54 cm). An overhead sprinkler irrigation system designed to divide the field in blocks of irrigated and nonirrigated plots had been installed in previous years. The blocks of post-spray irrigated and nonpost-spray irrigated plots were separated by 2.4-m alleyways. Irrigation water was applied one day after a fungicide treatment. All plots were sprinkler irrigated weekly with 2.54 cm of water prior to fungicide treatment initiation to insure uniform plant development. The sub-plot factor was fungicide application timing in which plots were sprayed either at night (3 a.m.–5 a.m., when peanut leaves were folded), or during the day (10 a.m.–12 p.m., when peanut leaves were unfolded). The sub-sub-plot factor was fungicide program, and each plot was sprayed with either a protectant, chlorothalonil (1.26 kg a.i./ha, 4 applications, Bravo WeatherStik; Syngenta Crop Protection Inc.), or a systemic, prothioconazole + tebuconazole (0.23 kg a.i./ha, 4 applications, Provost 433 SC; Bayer CroScience, Research Triangle Park, NC), tebuconazole (0.21 kg a.i./ha, 4 applications, Folicur 3.6 F; Bayer CropScience), fluoxastrobin (0.17 kg a.i./ha, 2 applications, Evito 4F; Arysta LifeScience, Cary, NC), azoxystrobin (0.31 kg a.i./ha, 2 applications, Abound 2.08 F; Syngenta Crop Protection, Inc.), flutolanil + propiconazole (0.45 kg a.i./ha, 4 applications, Artisan; Nichino America, Inc., Wilmington, DE), or pyraclostrobin (0.21 kg a.i./ha, 4 applications, Headline 2.09 EC; BASF Corp.). Fungicide treatments were initiated 30 to 40 DAP and subsequent applications followed a 14-day interval in a seven-application regime. All plots were day-sprayed with chlorothalonil (1.26 kg a.i./ha) on applications 1, 2 and 7 by a conventional tractor-mounted sprayer at about 345 kPa using three D2–13 nozzles per row. Fungicides with two applications were at applications 3 and 5 and the remaining applications in the regime were completed with chlorothalonil (1.26 kg a.i./ha) sprayed during the day. Fungicides with four applications were at applications 3 to 6 in this seven-application regime. All fungicide treatments were applied with a CO2-pressurized beltpack sprayer using a 2-liter bottle and a broadcast boom set up to apply 187 liters/ha at 276 kPa. The sprayer was equipped with three Conejet TX-SS6 hollow cone nozzles (Spraying Systems Co., Wheaton, IL) per row.
Environmental assessment
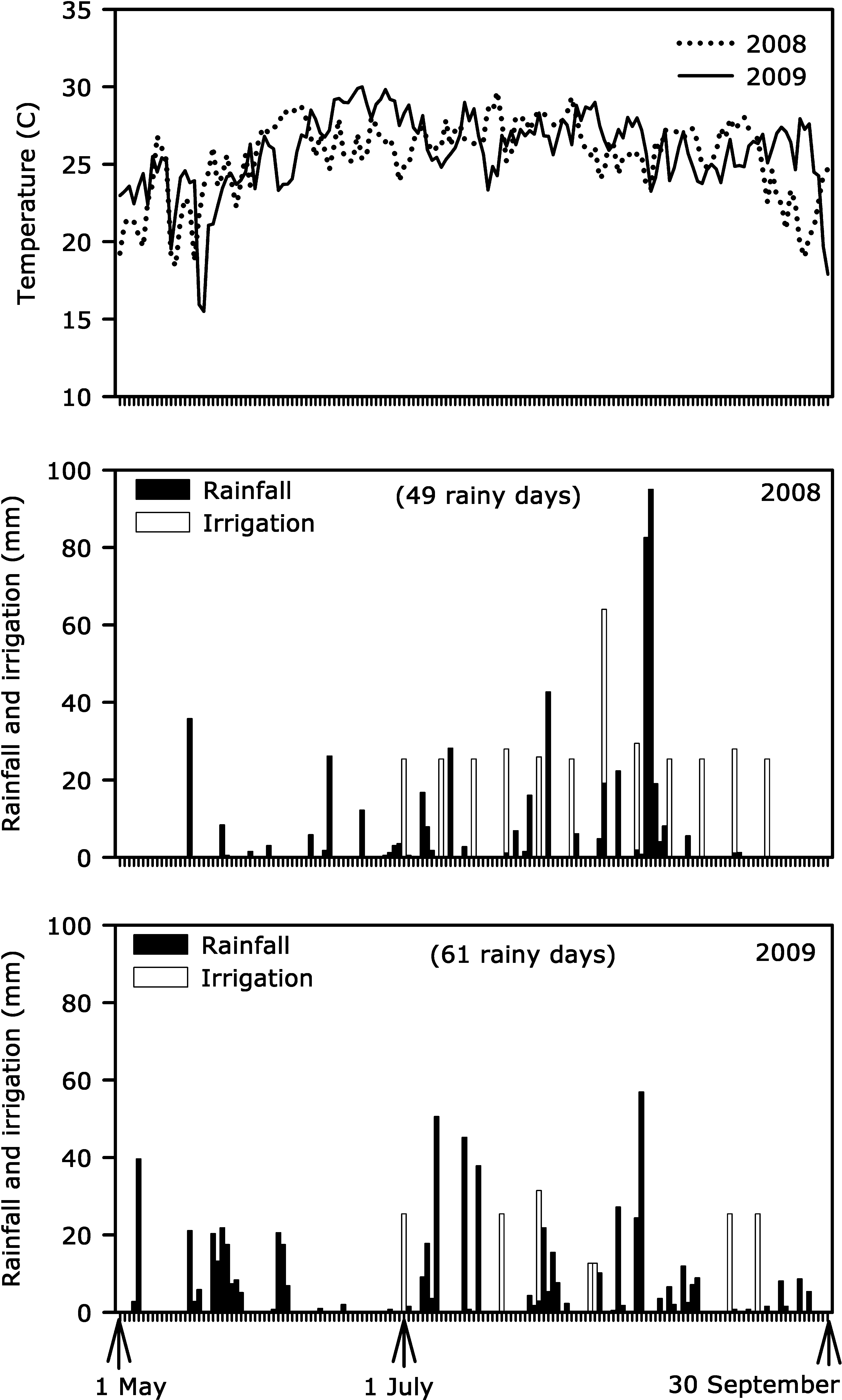
Temperature and rainfall in 2008 and 2009 were measured at the University of Georgia Tifton Coastal Plain weather station within 2000 m of the experimental field. The station collects the weather data through standard sensors (Campbell Scientific, Inc., Logan, UT) at one-second intervals. The data is further processed to provide average daily temperature and daily total rainfall. Average daily temperature and total daily rainfall data were plotted against the time duration of the experiment for each year.
Leaf spot assessment
Leaf spot, primarily early leaf spot, was evaluated two days before digging using the Florida 1-to-10 intensity scale, where 1 = no disease (0% defoliation); 2 = very few lesions and none on upper canopy (0% defoliation); 3 = few lesions and very few on upper canopy (0% defoliation); 4 = some lesions with more on upper canopy and noticeable defoliation (5%); 5 = lesions noticeable even on upper canopy and noticeable defoliation (20%); 6 = lesions numerous and very evident on upper canopy with significant defoliation (50%); 7 = lesions numerous on upper canopy with much defoliation (75%); 8 = upper canopy covered with lesions with high defoliation (90%); 9 = very few leaves remaining and those covered with lesions, some plants completely defoliated (98%); and 10 = plants completely defoliated or dead (Chiteka et al., 1988).
All plants in a plot were visually assessed, but leaf spot estimate was an arithmetic mean of the individual evaluations. The leaf spot ratings in this Florida 1-to-10 intensity scale are discrete variables at the level of the individual plant; nevertheless, as a result of the averaging process of all plants, they are (pseudo-)continuous variables at the plot level.
Stem rot assessment
Stem rot was evaluated within a day after digging by counting the number of disease foci in a plot. A stem rot focus was one or more consecutive affected plants in a 30-cm section, or less, of row. The symptoms and signs of infection were wilted and dead stems or entire plants, accompanied with white fungal mycelium and light to dark brown sclerotia on the plant tissues near or at the soil surface. Stem rot incidence was determined as: % stem rot = [(# 30-cm sections affected) ÷ (# 30-cm total sections in the plot)] × 100
Peanut pod yield assessment
The hull-scrape method of estimating pod maturity (Williams and Drexler, 1981) was used to determine the optimum digging date. All plots were mechanically dug at 139 and 133 DAP in 2008 and 2009, respectively. The windrows were harvested with a two-row combine approximately five days after digging and air-drying. Pods were then dried in a commercial drying trailer to approximately 8% moisture content and weighed after separation of soil and foreign material.
Statistical analysis
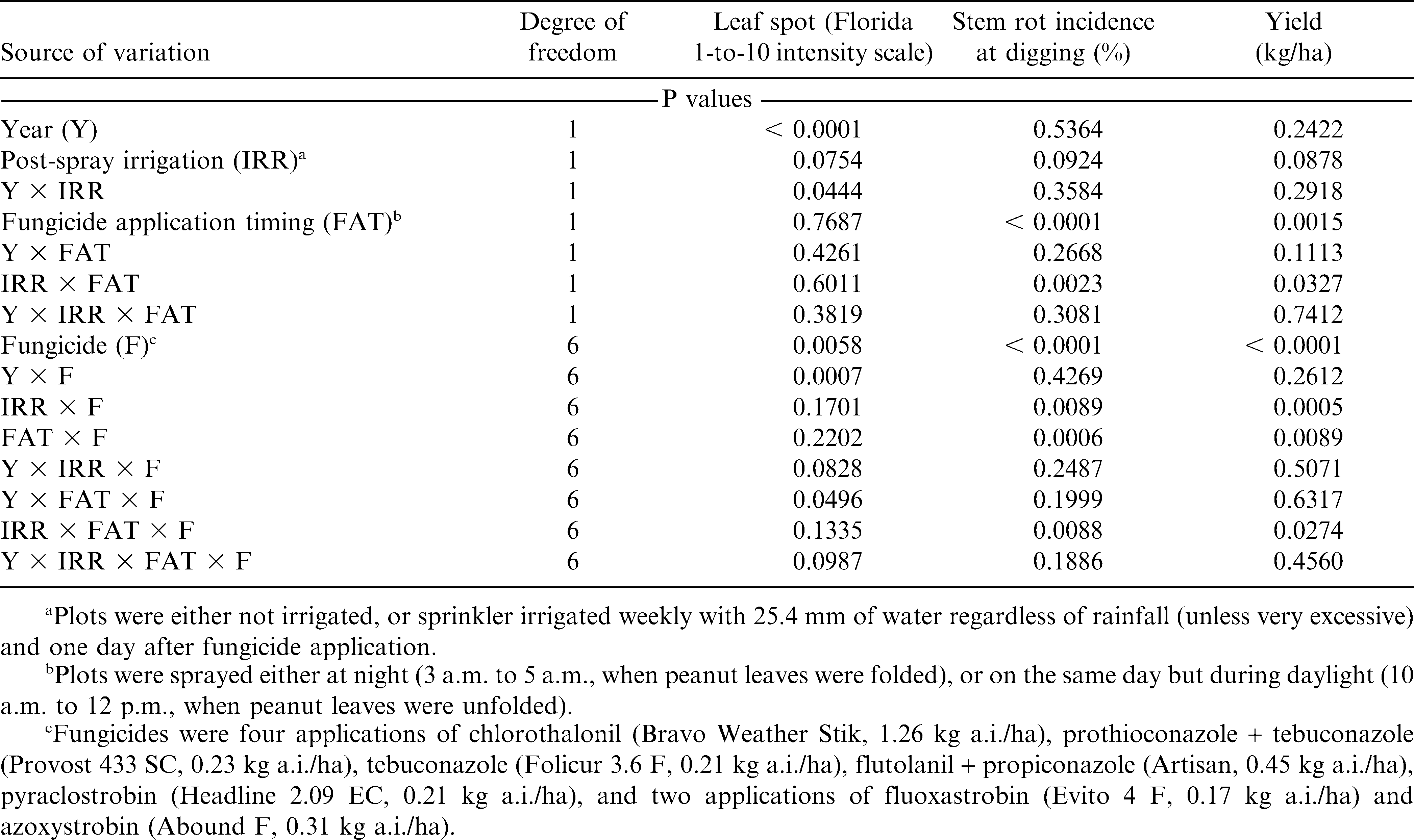
Leaf spot intensity, stem rot incidence and pod yield in response to post-spray irrigation, fungicide, and fungicide application timing were subjected to analysis of variance using the mixed procedure of Statistical Analysis Systems (SAS Institute, version 9.1, Cary, NC). The mixed procedure was used to provide the correct error terms for the partial and interactive effects of post-spray irrigation, fungicide, and fungicide application timing of the split-split-plot design. The ddfm = satterth option in the model statement performed Satterthwaite approximation of the degree of freedom. The P values of the interaction year × post-spray irrigation × fungicide application timing × fungicide, and of all derived interactions, for leaf spots, stem rot and yield were used to assess whether data could be combined across years and treatments (post-spray irrigation, fungicide application timing and fungicide). The least significant difference (P ≤ 0.05) was then calculated for mean separations for significant treatments.
Results
The need for irrigation was less in 2009 than in 2008 (Fig. 1). Although temperature patterns did not differ markedly, the 2009 growing season received more and well-distributed rainfall than 2008. As a result, the irrigation water applied prior to fungicide applications for general crop development in 2009 was 50% of that applied in 2008.
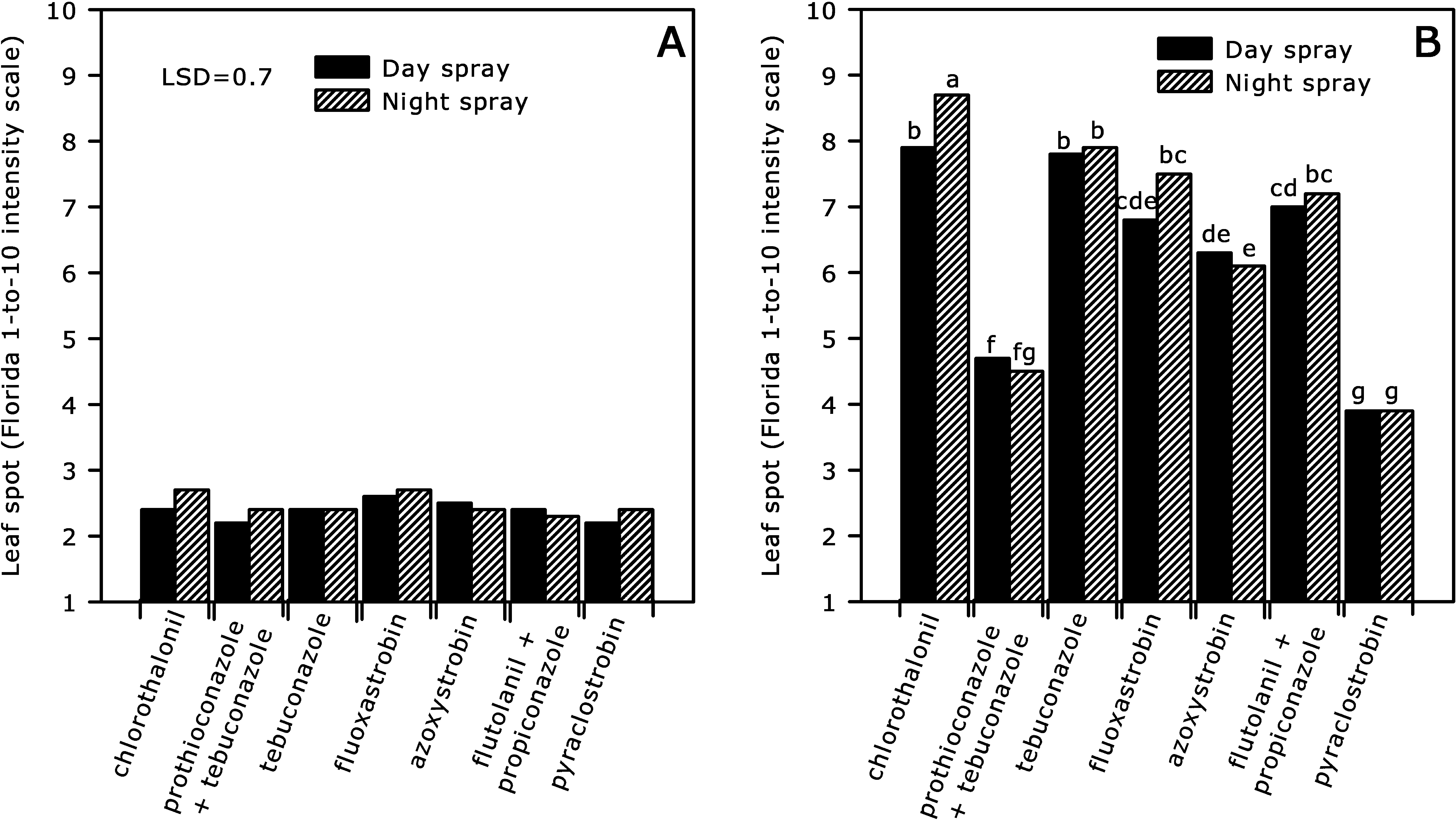
Interaction of year × fungicide application timing × fungicide was significant for leaf spots, and post-spray irrigation had no impact in this interaction (Table 1). Leaf spot intensity was low in 2008 and disease control was similar regardless of fungicide and fungicide application timing (Fig. 2, A). In 2009, the level of leaf spot intensity was high in the field. Night and day applications of the systemic fungicides had similar intensities of leaf spots, but night applications of chlorothalonil were not as effective on leaf spot compared with day applications. Leaf spot control with night applications of fluoxastrobin, flutolanil + propiconazole and night and day applications of tebuconazole were not greater than that of day applications with chlorothalonil. Pyraclostrobin and prothioconazole + tebuconazole had the lowest leaf spot ratings irrespective of fungicide application timing (Fig. 2, B).
Effect of fungicide and fungicide application timing on leaf spot (Florida 1-to-10 intensity scale) during 2008 (A) and 2009 (B) pooled across post-spray irrigation at Blackshank Farm, University of Georgia Tifton Coastal Plain Experiment Station. Bar heights are means for interaction of fungicide × fungicide application timing, and bars with the same letter(s) are not significantly different according to P ≤ 0.05.
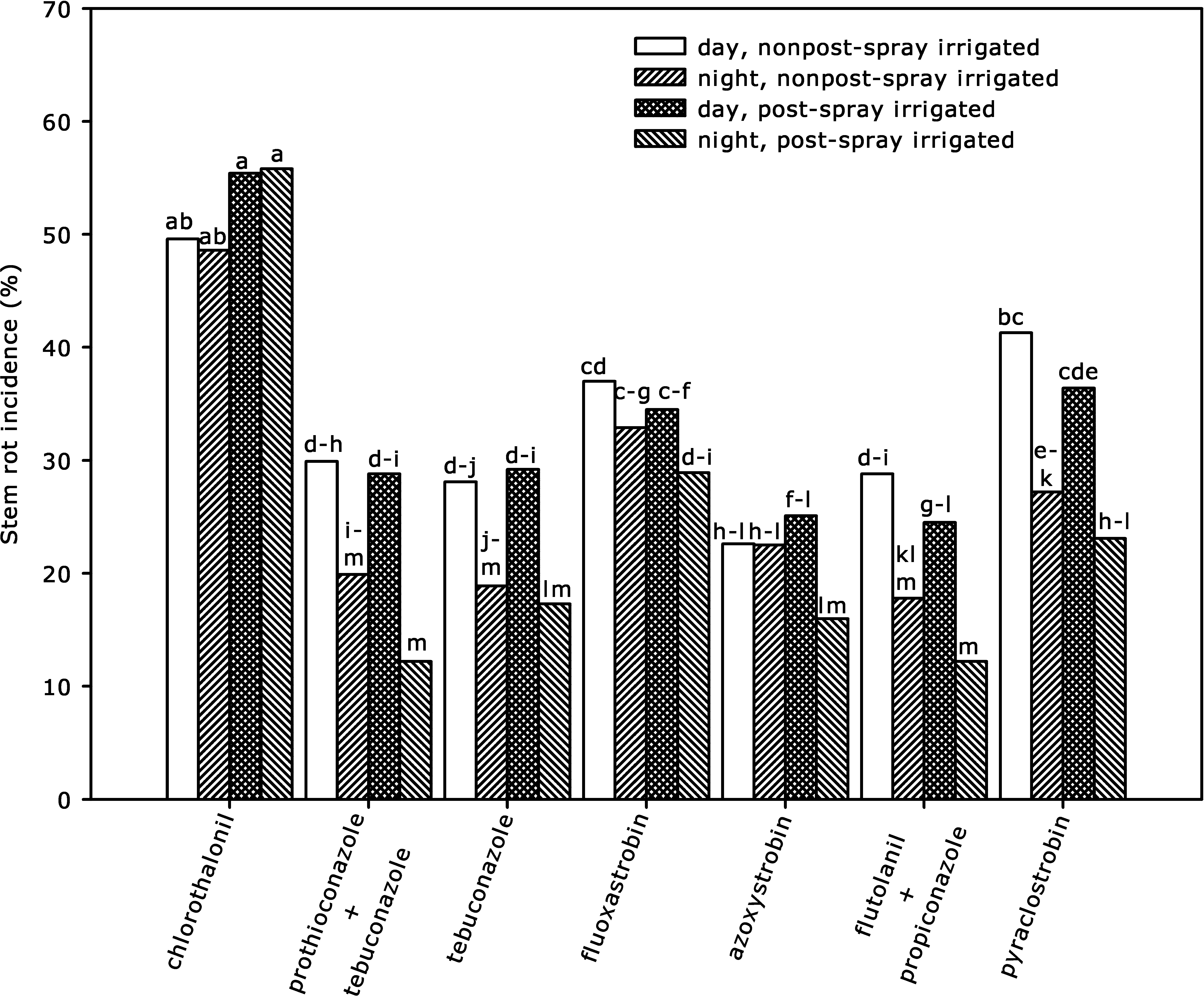
Interaction of post-spray irrigation × fungicide application timing × fungicide was significant for stem rot incidence and yield (Table 1). Data on stem rot incidence (Fig. 3) and yield (Fig. 4) in response to post-spray irrigation, fungicide, and fungicide application timing were pooled across years. Day application of pyraclostrobin in nonpost-spray irrigated plots did not reduce stem rot incidence compared with the chlorothalonil-control treatments in nonpost-spray irrigated plots (Fig. 3). However, night applications of pyraclostrobin, prothioconazole + tebuconazole, or flutolanil + propiconazole all decreased stem rot incidence compared with day applications in both nonpost-spray irrigated and post-spray irrigated plots, while night application of tebuconazole decreased stem rot incidence compared with day application only in post-spray irrigated plots. Also, post-spray irrigation did not affect stem rot control as greatly as application timing did, but plots with post-spray irrigation tended to have less stem rot. For instance, night application of tebuconazole with post-spray irrigation had significantly less stem rot than the day application without post-spray irrigation, whereas night and day applications of the same fungicide without post-spray irrigation were not different. The control of stem rot with fluoxastrobin and azoxystrobin was not affected by application timing or post-spray irrigation (Fig. 3).
Effect of post-spray sprinkler irrigation, fungicide, and fungicide application timing on stem rot incidence across 2008 and 2009 seasons at Blackshank Farm, University of Georgia Tifton Coastal Plain Experiment Station. Bar heights are means for interaction of post-spray sprinkler irrigation × fungicide × fungicide application timing, and bars with the same letter(s) are not significantly different according to P ≤ 0.05.
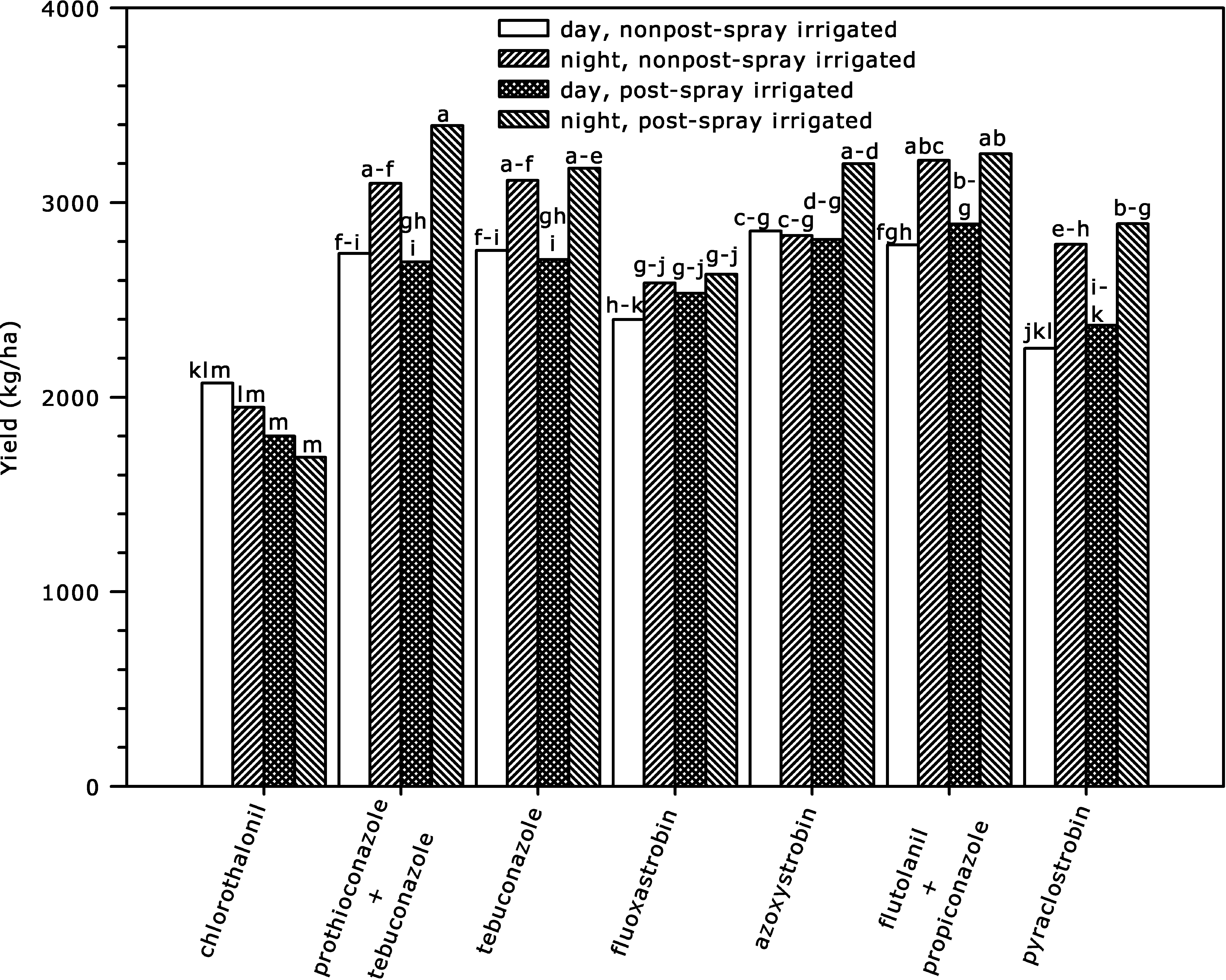
Effect of post-spray sprinkler irrigation, fungicide, and fungicide application timing on yield across 2008 and 2009 seasons at Blackshank Farm, University of Georgia Tifton Coastal Plain Experiment Station. Bar heights are means for interaction of post-spray sprinkler irrigation × fungicide × fungicide application timing, and bars with different letter(s) are significantly different according to P ≤ 0.05.
There were some differences in yield with individual fungicide due to post-spray irrigation treatments (Fig. 4). Night applications of tebuconazole and prothioconazole + tebuconazole with post-spray irrigation had significantly higher yields than the day applications of the same fungicides without post-spray irrigation, while night and day applications of these fungicides without post-spray irrigation were not significantly different. Also, some fungicides increased yield when plots were post-spray irrigated compared to chlorothalonil standard, while the day applications of the same fungicides and chlorothalonil were not different in plots without post-spray irrigation (ex. fluoxastrobin). In contrast, night applications increased yield over day applications in either the post-spray irrigated or nonpost-spray irrigated plots with all treatments except fluoxastrobin and azoxystrobin. The differences in yield between night and day fungicide applications occurred in plots either post-spray irrigated (prothioconazole + tebuconazole, and tebuconazole alone), nonpost-spray irrigated (flutolanil + propiconazole), or in both post-spray irrigated and nonpost-spray irrigated (pyraclostrobin) (Fig. 4).
Discussion
Leaf spot intensity was low in 2008 and high in 2009. The levels of recorded leaf spot may reflect differences in the rainfall intensity and pattern in those years. However, rainfall and temperature were conducive for development of stem rot during both seasons. The research findings reported here corroborate our previous studies (Augusto et al., 2010a; Augusto et al., 2010b) in which applying systemic fungicides at night when peanut leaves were folded increased stem rot control, augmented yield, and provided similar level of control of leaf spots compared with standard day applications.
In 2009 with high leaf spot intensity, night applications of chlorothalonil provided less disease control compared with day applications. Chlorothalonil is a protectant fungicide with no systemic movement; therefore, thorough coverage is essential for effective control of leaf spots. Most of the chlorothalonil applied at night, when leaves are folded, is deposited on the lower leaf surface (Augusto et al., 2010b), whereas infection and sporulation by C. arachidicola occurs on the upper leaf surface (Smith and Littrell, 1980). Therefore, night application of chlorothalonil or other nonsystemic fungicides can be expected to provide limited control of leaf spots compared with day application in which most of the fungicide is deposited on the upper leaf surface (Augusto et al., 2010b). At this time of the morning, leaves are usually wet from dew also which would further reduce the retention of chlorothalonil on leaves where it is needed for foliar diseases. Systemic tebuconazole applied at night or during the day did not improve control of leaf spots compared with day application of standard chlorothalonil when disease pressure was high. However, tebuconazole has had limited effectiveness against leaf spots in this area for several years due to the presence of triazole-resistant isolates of C. arachidicola (Culbreath et al., 2005; Stevenson and Culbreath, 2006; Stevenson et al., 1999). Pyraclostrobin and prothioconazole + tebuconazole were the most effective leaf spot fungicides, regardless of application timing. Pyraclostrobin, in particular, has been the most effective labeled peanut fungicide against leaf spots in Georgia (Kemerait et al., 2010).
The effect of irrigating peanut a day after fungicide application to redistribute fungicide residuals within the canopy for a balanced control of leaf spots and stem rot was less clear. Woodward (2006) showed that control of leaf spots and stem rot could be maximized with timely irrigation after applying a systemic fungicide. In this study, the interaction of post-spray irrigation and fungicide application timing with commonly used peanut fungicides only occurred for stem rot and yield.
The interaction of post-spray irrigation and application timing (day and night) was the most significant among the evaluated fungicides with pyraclostrobin for stem rot (P = 0.0299) and yield (P = 0.0371). Although pyraclostrobin is the standard fungicide for leaf spot control, it has been erratic against stem rot and high rates (0.21–0.27 kg a.i./ha) are recommended for effective disease control (Kemerait et al., 2010). The reason for inconsistent stem rot control is thought to be due, at least partially, to high affinity of pyraclostrobin to leaf surface waxes and quick binding when applied to dry foliage (Martin, 2003), resulting in decreased redistribution on the lower canopy. This study is the first to report on the potential redistribution of pyraclostrobin with a post-spray irrigation, although the effect was minimal compared to the difference in day versus night applications. In our previous study (Augusto et al., 2010b), stem rot control and yield were greatly improved when pyraclostrobin was applied at night when peanut leaves were folded and wet compared with day application when leaves were unfolded and dry.
As discussed previously, the impact of timely peanut irrigation after application of systemic fungicides to manage stem rot or increase yield was marginal, but it did have a positive effect in some cases. All plots, even the nonpost-spray irrigated treatments, received weekly irrigation and often some rainfall which helped to redistribute fungicide residues. The potential of irrigating peanut soon after application of systemic fungicides to improve control of diseases caused by soilborne pathogens may vary with fungicide, disease pressure and rainfall intensity and pattern. More than half of the peanut fields in Georgia receive some irrigation, primarily from overhead center-pivot irrigation, so this is an option for many growers. Also, scheduling a timely irrigation to reallocate fungicide residues within the peanut canopy will not add apparent production costs. Benefits of night fungicide applications have been discussed extensively (Augusto et al., 2010a; Augusto et al., 2010b). While combining these two disease management strategies, e.i., timely post-spray irrigation of peanut and night fungicide applications, can maximize the stem rot control and yield, it appears that application timing has much more of an effect than does post-spray irrigation. It is also important to not compromise control of leaf spots by removing significant residual of contact fungicides from the top foliage when combining night applications and washing off fungicide residuals with post-spray irrigation.
Acknowledgements
The authors would like to thank P. Hilton, L. Mullis and R. Griffin for their field assistance.
Literature Cited
Alderman S. C. and Beute M. K. 1987 Influence of temperature, lesion water potential, and cyclic wet-dry periods on sporulation of Cercospora arachidicola on peanut. Phytopathology 77 : 960 – 963 .
Augusto J. , Brenneman T. B. , Baldwin J. A. , and Smith N. B. 2010 Maximizing economic returns and minimizing stem rot incidence with optimum plant stands of peanut in Nicaragua. Peanut Sci 37 : 137 – 143 .
Augusto J. , Brenneman T. B. , Culbreath A. K. , and Sumner P. 2010a Night spraying peanut fungicides. I. Extended fungicide residual and integrated disease management. Plant Dis 94 : 676 – 682 .
Augusto J. , Brenneman T. B. , Culbreath A. K. , and Sumner P. 2010b Night spraying peanut fungicides. II. Application timings and spray deposition in the lower canopy. Plant Dis 94 : 683 – 689 .
Beasley J. P. 2007 Irrigation strategies. pp. 7 – 8 in. 2007 peanut update Prostko E. P. (ed.) Coop. Ext. Ser. College of Agric. Environ. Sci. University of Georgia .
Beasley J. P. 2010 Non-irrigated peanut production. pp. 30 – 32 in: 2010 peanut update Beasley J. P. (ed.) Coop. Ext. Ser. College of Agric. Environ. Sci. University of Georgia .
Boote K. J. 1982 Growth stages of peanut (Arachis hypogaea L.). Peanut Sci 9 : 35 – 40 .
Branch W. D. 1996 Registration of ‘Georgia Green’ peanut. Crop Sci 36 : 806 .
Brenneman T. B. , Murphy A. P. , and Csinos A. S. 1991 Activity of tebuconazole on Sclerotium rolfsii and Rhizoctonia solani, two soilborne pathogens of peanut. Plant Dis 75 : 744 – 747 .
Calhoun J. W. 1983 Soil survey of Tift County, Georgia USDA-SCS Washington, DC .
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , Kucharek T. A. , and Knauft D. A. 1988 Components of resistance to late leaf spots in peanut. I. Levels of variability – implications for selection. Peanut Sci 15 : 25 – 30 .
Culbreath A. K. , Brenneman T. B. , and Kemerait R. C. 2001 Applications of mixture of copper fungicides and chlorothalonil for management of peanut leaf spots diseases. Plant Health Progress doi:10.1094/PHP-2001-1116-01-RS.
Culbreath A. K. , Brenneman T. B. , Kemerait R. C. , and Stevenson K. L. 2005 Relative performance of tebuconazole and chlorothalonil for control of peanut leaf spot from 1994 through 2004. (Abstr.) Proc. Am. Peanut Res. Educ. Soc 37 : 54 – 55 .
Culbreath A. K. , Minton N. A. , Brenneman T. B. , and Mullinix B. G. 1992 Response of Florunner and Southern Runner peanut cultivars to chemical treatments for management of late leaf spot, southern stem rot, and nematodes. Plant Dis 76 : 1199 – 1203 .
Culbreath A. K. , Stevenson K. L. , and Brenneman T. B. 2002 Management of late leaf spot of peanut with benomyl and chlotothalonil: A study in preserving fungicide utility. Plant Dis 86 : 349 – 355 .
Davis R. F. , Smith F. D. , Brenneman T. B. , and McLean H. 1996 Effect of irrigation on expression of stem rot of peanut and comparison of aboveground and belowground disease ratings. Plant Dis 80 : 1155 – 1159 .
Harrison K. 2009 Irrigation scheduling methods. Univ. Georgia Coop. Ext. Bull. B-974.
Kemerait R. 2006 Georgia plant disease loss estimates. Coop. Ext. Ser. Bull. 41-09, University of Georgia, Athens.
Kemerait R. 2007 Georgia plant disease loss estimates. Coop. Ext. Ser. Bull. 41-10, University of Georgia, Athens.
Kemerait R. 2008 Georgia plant disease loss estimates. Coop. Ext. Ser. Ann. Pub. 102-1, University of Georgia, Athens.
Kemerait B. , Brenneman T. , and Culbreath A. 2006 Peanut disease control. pp. 126 – 127 in: 2006 Georgia Pest Management Handbook. Special Bull. 28 Guillebeau P. (ed.) University of Georgia Coop. Ext. College of Agric. Environ. Sci. University of Georgia .
Kemerait B. , Brenneman T. , and Culbreath A. 2010 2010 peanut disease update. pp. 57 – 80 in: 2010 peanut update Beasley J. P. (ed.) Coop. Ext. Ser. College of Agric. Environ. Sci. University of Georgia .
Lyle J. A. 1964 Development of Cercospora leaf spot of peanut. J. Ala. Acad. Sci 35 : 9 .
Martin B. 2003 A new strobilurin fungicide for turfgrass disease control. Golf Course Manage 71 : 188 – 191 .
Punja Z. K. 1985 The biology, ecology, and control of Sclerotium rolfsii. Annu. Rev. Phytopathol 23 : 97 – 127 .
Sconyers L. E. , Brenneman T. B. , Stevenson K. L. , and Mullinix B. G. 2005 Effect of plant spacing, inoculation date, and peanut cultivar on epidemics of peanut stem rot and tomato spotted wilt. Plant Dis 89 : 969 – 974 .
Smith D. H. and Crosby F. L. 1973 Aerobiology of two peanut leaf spot fungi. Phytopathology 63 : 703 – 707 .
Smith D. H. and Littrell R. H. 1980 Management of peanut foliar diseases with fungicides. Plant Dis 64 : 356 – 361 .
Stansell J. R. , Shepherd J. L. , Pallas J. E. , Bruce R. R. , Minton N. A. , Bell D. K. , and Morgan L. W. 1976 Peanut responses to soil water variables in the Southeast. Peanut Sci 3 : 44 – 48 .
Stevenson K. L. and Culbreath A. K. 2006 Evidence of reduced sensitivity to tebuconazole in leaf spot pathogens. (Abstr.) Proc. Am. Peanut Res. Educ. Soc 38 : 62.
Stevenson K. L. , Padgett G. B. , and Culbreath A. K. 1999 Sensitivity of early and late peanut leaf spot pathogens to DMI fungicides. (Abstr.) Proc. Am. Peanut Res. Educ. Soc 31 : 23 .
Truman C. C. , Strickland T. C. , Potter T. L. , Franklin D. H. , Bosch D. D. , and Bednarz C. W. 2007 Variable rainfall intensity and tillage effects on runoff, sediment, and carbon losses from a loamy sand under simulated rainfall. J. Environ. Qual 36 : 1495 – 1502 .
Watkins G. M. 1961 Physiology of Sclerotium rolfsii, with emphasis on parasitism. Phytopathology 51 : 110 – 113 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Woodward J. E. 2006 Optimizing efficacy and economic benefits of fungicides for peanut disease control via pre-plant analysis of disease risk and irrigation timing Ph.D. Dissertation, University of Georgia Athens 247 pp.
Notes
- Postdoctoral Research Associate and Professor, respectively, Department of Plant Pathology, The University of Georgia, Coastal Plain Experiment Station, P. O. Box 748, Tifton, Georgia 31793-0748. [^] *Corresponding author email: jaugusto@uga.edu
Author Affiliations