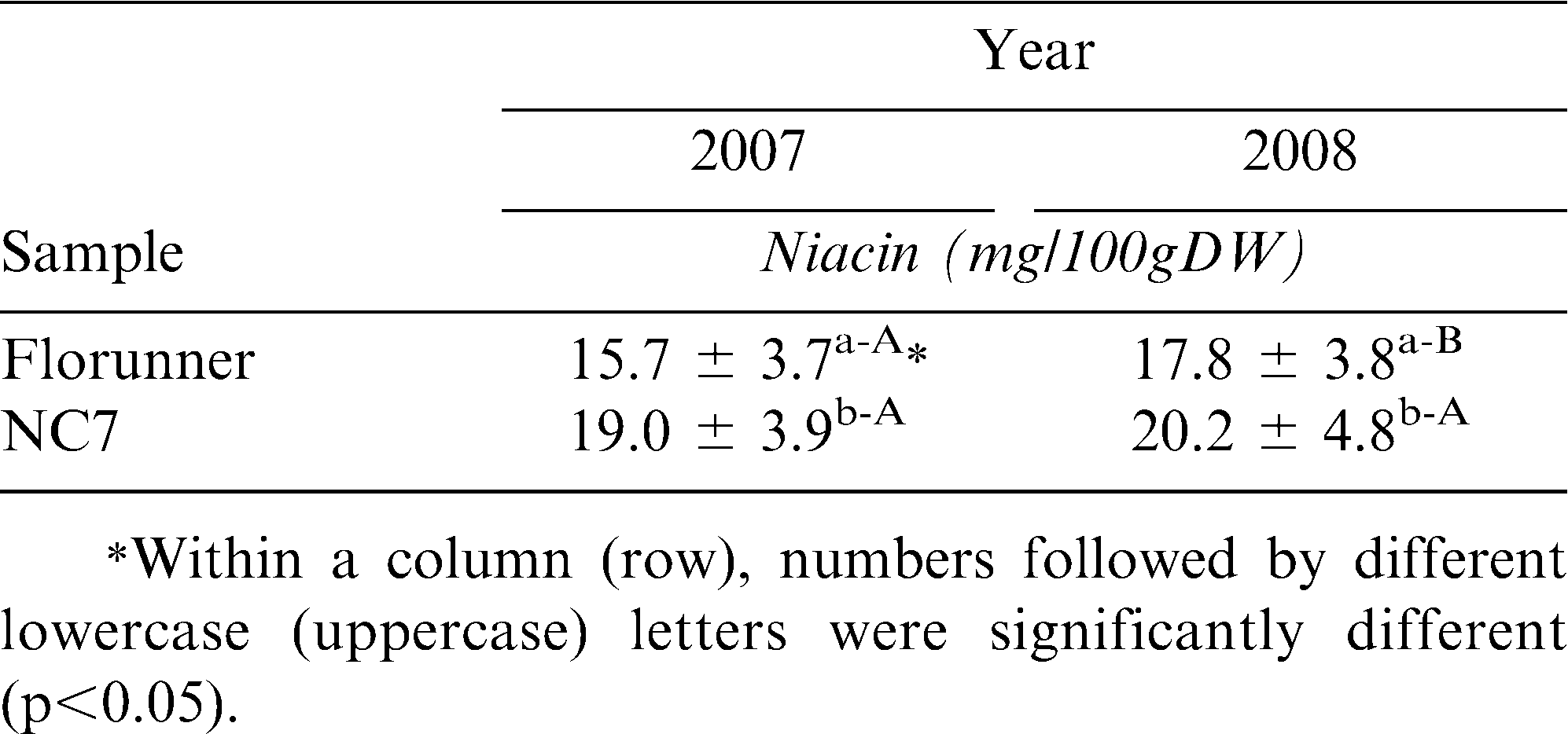Introduction
Niacin is synthesized in plants via the tryptophan degradation pathway, with tryptophan serving as the niacin precursor (Arditti and Tarr, 1979). Niacin, also known as nicotinic acid or Vitamin B3, and includes the derivative nicotinamide. It is used to form the coenzymes, nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP). As such, over 200 enzymes use niacin in the coenzyme form in plant and animal cells for reactions involving electron transfer, energy production and degradation of carbohydrates, proteins, fats and alcohol (Hunt and Groff, 1990). In humans, high doses of niacin have been found to contribution to good vascular health due to its involvement in maintaining proper blood lipid balance (Wu et al., 2010, Sorrentino et al., 2010, Lee et al., 2009). It has also been used therapeutically for blood cholesterol control (Costet, 2010, Shah et al., 2010, Silva and Ferreira, 2009).
Peanuts are considered a good source of tryptophan as are many high protein foods. In humans, niacin can be synthesized in the liver from dietary tryptophan in small amounts. It has been reported that 60 mg of tryptophan ingested will produce 1 mg of niacin (McCormick, 1988). An adequate intake of niacin would spare the need for tryptophan conversion. Currently, the USDA Nutrient Database lists peanuts at 12.07 mg/100g (as consumed) for raw and 13.50 mg/100g (as consumed) for dry roasted (USDA, ARS, 2009). This corresponds to one ounce of roasted peanuts providing about 20 percent of the daily recommended intake value (RDI).
The Uniform Peanut Performance Test (UPPT) provides an excellent set of samples for the analysis of various peanut components grown at 10 locations representative of most of the peanut-growing regions in the USA. The varieties, Florunner (runner type) and NC7 (virginia type) were grown as control samples at each location each year. The USDA Market Quality and Handling Unit has performed chemical characterization analysis on these samples since 2000. However; the only vitamin that has been measured in these trials is Vitamin E (tocopherols). This study used samples from the UPPT for 2 years from 10 locations to investigate the pattern of geographic and year-to-year variation in niacin content of two established commercial varieties.
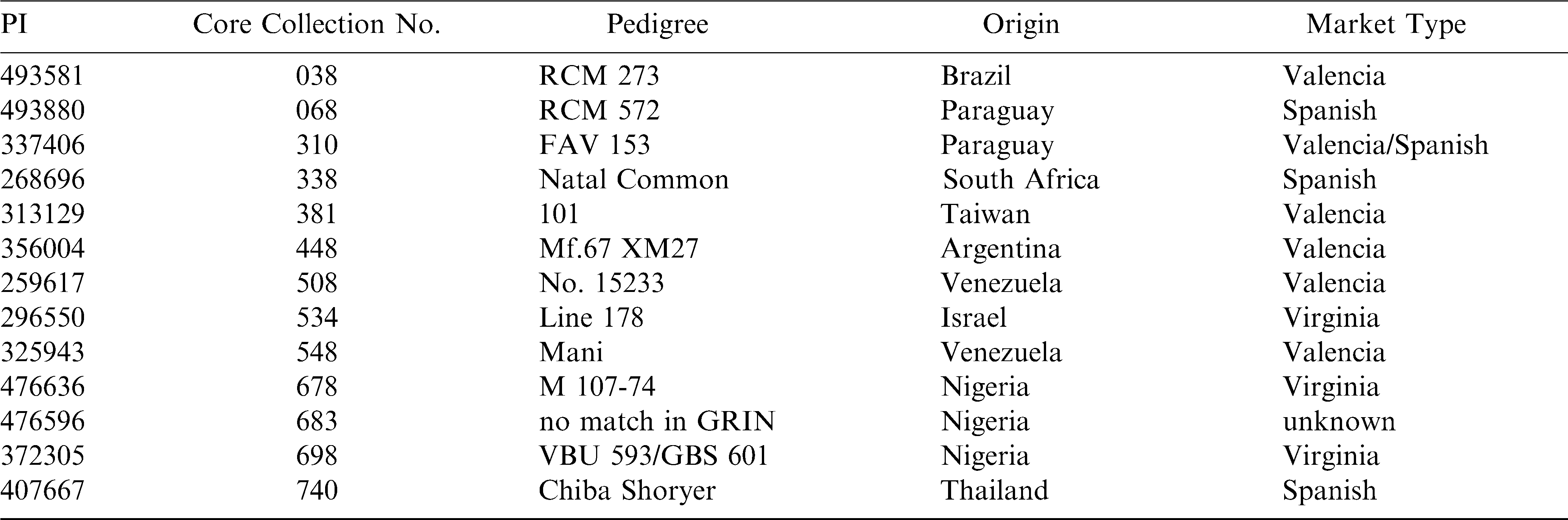
The U.S. peanut (Arachis hypogaea L.) germplasm collection contains 7432 accessions and is considered a wealth of genetic diversity (Holbrook et al., 1993). Through a series of studies, the collection has been reduced to 112 accessions, based on country of origin, available morphological data and cluster analysis for eight each of above and below ground characteristics and this group has been given the designation of “The Core of the Core” (Holbrook and Dong, 2005). A selection of these germplasm samples were chosen to be part of this study based on their availability in sufficient quantity to perform the analysis.
Materials and Methods
For the years, 2007 and 2008, UPPT samples were obtained from plots grown in each of ten test locations ( Suffolk, VA (VA), Lewiston, NC (NC) and Clemson, SC (SC) in the Virginia-Carolina production area; Tifton, GA (GA), Headland, AL (AL) and Marianna, FL (FL) in the Southeastern production area; and Stephenville, TX (C-TX), College Station, TX (S-TX), Lubbock, TX (W-TX) and Stillwater, OK (OK) in the Southwestern production area. Growing conditions are described in the annual reports of yield and grade of UPPT entries for the specific locations (Branch et al., 2008, 2009). From the 2007 UPPT, Florunner (medium grade size) and NC7 (Extra Large Kernel {ELK} grade size) were obtained from all locations except C-TX. In 2008, the same varieties and sizes were obtained from all growing locations. The 2007 samples were only analyzed as raw peanuts. The 2008 UPPT samples were analyzed as both raw and roasted peanuts, except for the GA samples which were in limited supply for roasting. Samples were roasted at 167°C for 17–20 minutes to a Hunter L value of 50 ∀ 1 (Sanders et al., 1989). In addition to the UPPT samples, 13 Core of the Core germplasm samples were selected from the 2008 crop year grown in Lewiston, NC. These 13 samples were a random sampling of the Core germplasm available for analysis and were compared Florunner and NC7 grown in the same location (NC) in the same year (2008). The samples analyzed for the Core of the Core are listed in Table 1.
R Reagents and Chemicals
All reagents were ACS grade or better and purchased from Fisher Chemical Corporation (Fairlawn, NJ) unless otherwise stated.
A Analysis Procedure
The samples were analyzed for niacin after conversion to nicotinic acid according to AOAC 961.14 (AOAC, 1990). In brief, peanuts were homogenized by grinding whole seed in a Krups coffee mill (Millville, NJ) until a fine meal was obtained. Five g of each homogenized sample was digested in 200 ml of 1 N H2SO4 using steam under reduced pressure in an autoclave (AMSCO Scientific Series 3021-S, STERIS Corp., Mentor, OH).
After pH adjustment, the samples were filtered and protein was precipitated out with the aid of (NH4)2SO4 and filtered again. Aliquots of the filtrate were added to tubes, buffered and sulfanilic acid and cyanogen bromide solutions were added to form a yellow color. The intensity of the color was read at 470 nm using a spectrophotometer (Genesys 20, Thermo Scientific, Waltham, MA) and compared to a standard curve prepared using nicotinic acid (Sigma Chemical Corp., St. Louis, MO) that had been subjected to the same derivatization reaction as the samples. Each sample was analyzed in triplicate.
Statistical Analysis
Statistics were performed using a mixed model with no interaction terms to test the significance of year (2007 versus 2008), location, variety (Florunner versus NC7 versus Core samples), and condition (raw versus roasted). No interaction terms were included because only one independent sample for each variety at each location for each year was available for analysis. Duncan's multiple range test was used for means comparisons. All statistics were performed using SAS (Slaughter and Delwiche, 2010).
Results
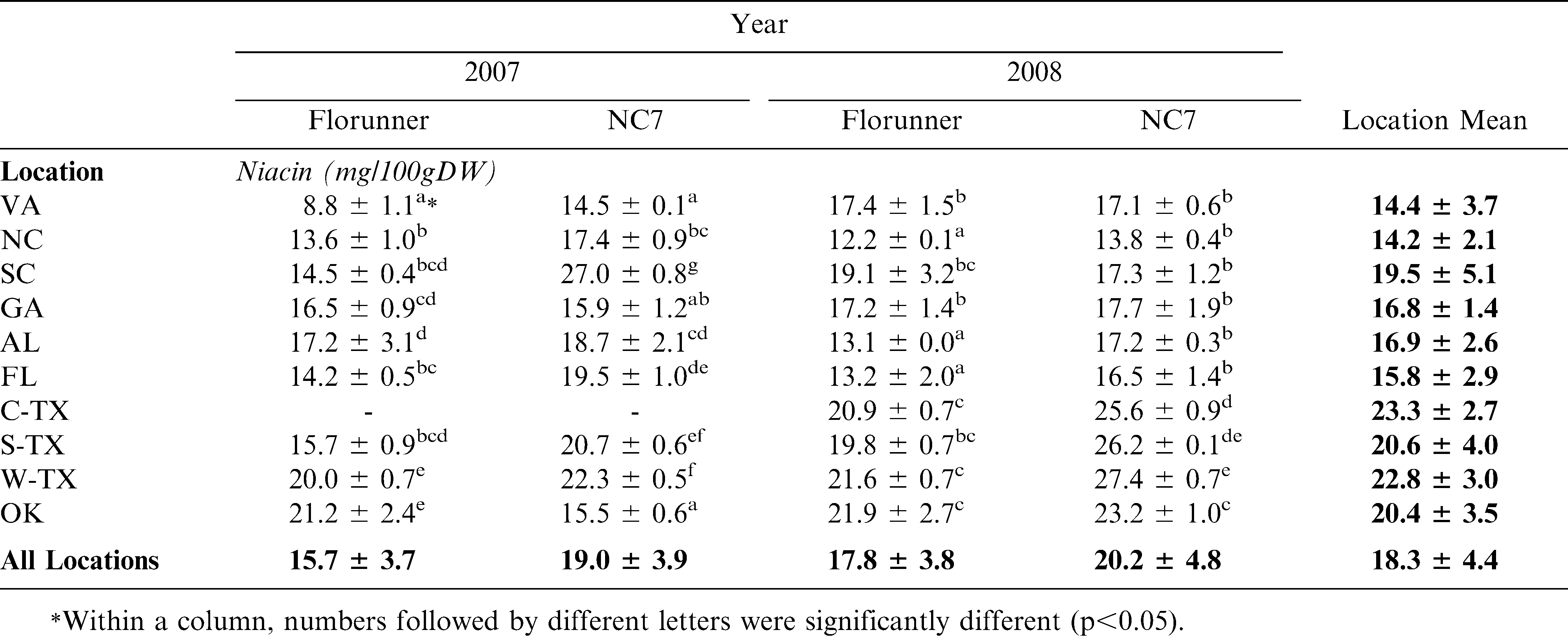
Average niacin contents were compared for 2 peanut varieties at 10 locations over 2 production years. Raw NC7 samples averaged 19.0 mg/100g dry weight basis (DW) in 2007 and 20.2 mg/100g DW in 2008 while Florunner averaged 15.7 and 17.8 mg/100g DW in 2007 and 2008 respectively (Table 2). NC7 was significantly higher than Florunner in both years (p<0.05). This was true for both varieties at every location, with the exception being NC7 in SC which had higher niacin in 2007 (Table 2). The average Florunner sample grown in 2007 had significantly lower niacin contents compared to those grown in 2008 (p<0.05) while NC 7 was not different between the two years (Table 2). Average niacin concentration for each variety at 10 locations ranged from 8.8 to 27.4 mag/100g DW (Table 3). For each variety in each year, there were significant differences among growing locations (p<0.05) with the exception of SC, the western locations, C-TX, S-TX and OK which were higher than the eastern locations for both Florunner and NC7 in both years (Table 3).
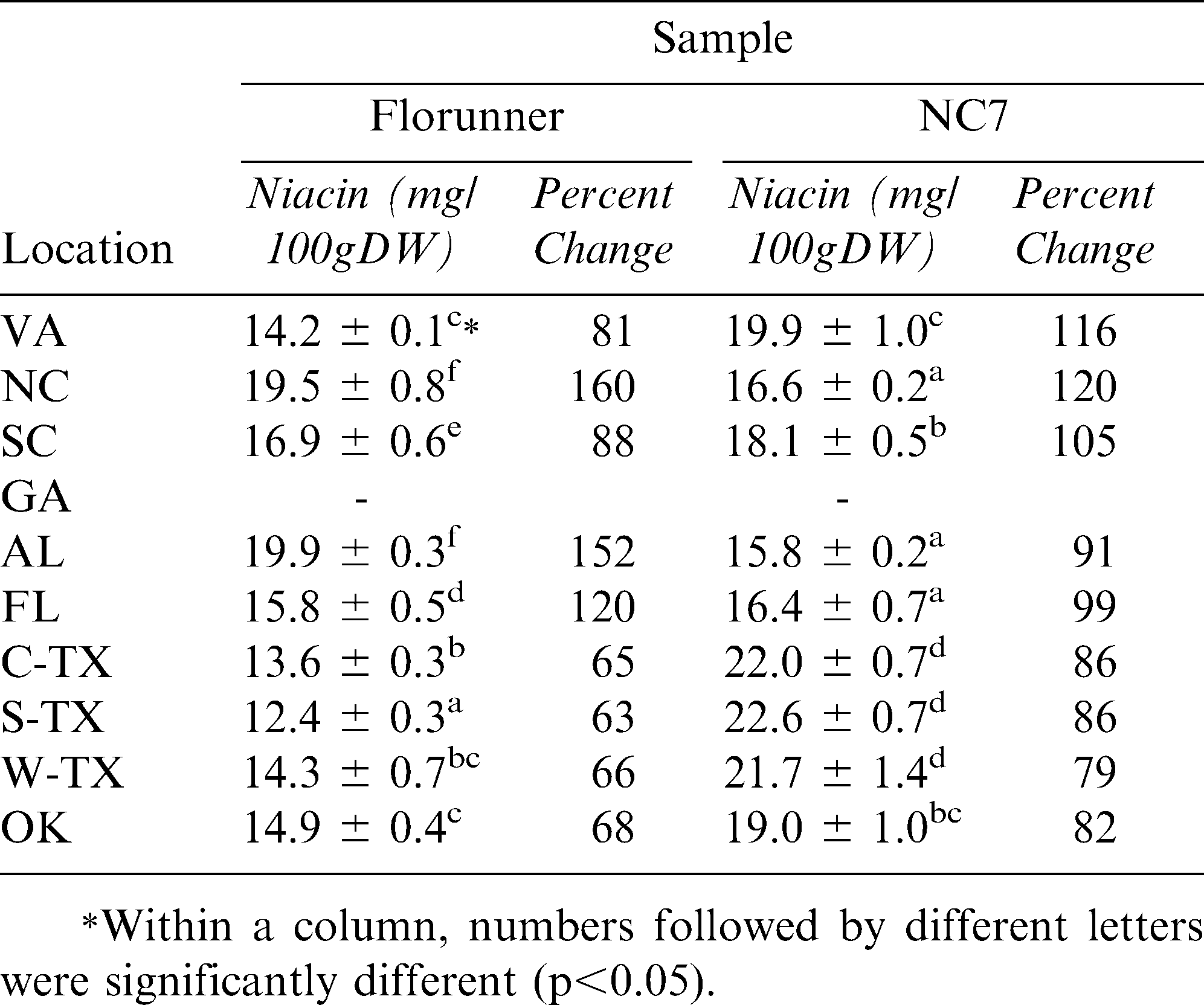
In order to determine the effect of roasting on niacin content, the 2008 UPPT samples were roasted to a predetermined Hunter L value of 50 ∀ 1 (Sanders, etal, 1989). Florunner and NC7 grown in GA were not included due to the lack of sample availability. Upon roasting, Florunner niacin levels dropped 9% from 17.8 to 15.7 mg/100g DW (Table 4). NC7 niacin values dropped from 20.2 to 19.0 mg/100g DW but the drop was not statistically significant (Table 4). These percentages were deceptive, however; as some locations dropped more than 9% and at some locations, niacin levels were elevated after roasting (Table 5). Niacin concentrations at the 10 locations ranged from 12.4 to 19.9 mg/100g DW for Florunner and 15.8 to 22.6 mg/100g DW for NC7 samples (Table 5). There was a statistically significant difference among growing locations (p<0.05) for both varieties but roasting response was difference for each. Like the raw samples of both raw varieties, the roast NC7 samples had higher niacin in western locations. Florunner had uniformly lower levels of niacin after roasting except for AL and NC which had significantly higher niacin concentrations than peanuts grown in other locations (p<0.05) (Table 5). For both varieties, the niacin loss was greater in the western regions (Table 5).
Select samples from the Core of the Core peanut germplasm were analyzed to obtain insight into the range of niacin for raw samples occurring within the germplasm collection. These Core of the Core samples were grown in 2008 at the NC UPPT location (Lewiston, NC) allowing for direct comparison to the Florunner and NC7 samples from that location and that data is included in Table 5. The raw Florunner and NC7 sample niacin levels at NC location were 12.2 and 13.8 mg/100g DW (Table 6). The 13 Core of the Core samples ranged from 13.9 to 20.7 mg/100g DW with an average of 18.0 mg/100g DW (Table 6). There were significant differences among the Core samples and the UPPT samples had less niacin than all the Core samples. (Table 6). It should also be noted that the niacin content in Florunner , but not NC7 was the lowest of all the UPPT locations.
Discussion
The UPPT data suggest a relationship between the amount of niacin present in a peanut sample and the environment in which it was grown. There was an increase at most locations from 2007 to 2008, and these types of changes are well documented for several peanut components Year-to-year variation was present but there was also a pattern in the niacin levels related to location, that is, the concentrations were generally consistently higher in the western locations when compared to the eastern locations (USDA-ARS, UPPT data, 2010). Temperature, water, and light are all important factors in vitamin synthesis (Gustafson, 1953). Temperature alters reaction rates and influences the kinetic properties of enzymes within the plant (Smirnoff, 1995). Optimum growth temperatures for peanut plants range between 27–30°C during daylight, which is characteristic among the 10 growing locations seen in this study (Ketring et al., 1982, Gustafson, 1955, Brumley and Sosebee, 1978). One difference between the eastern and the western locations is night temperature (Canty et al., 2010). The samples in this study grown in the western regions with lower night temperatures tended to have increased niacin contents in both years.
The higher levels of niacin content seen in peanuts grown in the western region exhibit a similar pattern to that of sugar content in peanuts (Casini et al., 2003, McMeans et al., 1990). The changes in temperature from day to night influence metabolic rate, resulting in differential effects on enzymes and pathways, such as carbohydrate metabolism and respiration (Smirnoff, 1995). During dark respiration, sugars, as well as other nutrients, are utilized for metabolic processes (Smirnoff, 1995). Respiration is slowed when temperatures are lower, resulting in less sugar being used at night (Burke, et al, 2009, Rolland, et al, 2002).
As with sugars, the significantly higher concentration of niacin in peanuts grown in TX and OK could be a result of these temperature fluctuations. NAD and NADP, two major components of niacin, are coenzymes for several dehydrogenase enzymes, which are essential for photosynthesis and respiration (Brumley, 1974, Orten and Neuhaus, 1970). These coenzymes accept electrons from substrates, in which NAD then oxidizes lipids and carbohydrates present in the plant, generating ATP (Brumley, 1974). NAD is essential in the mitochondrial electron transport chain and the tricarboxylic acid (TCA) cycle (Brumley, 1974). During dark respiration, when temperatures are lowest, less vegetative and reproductive growth occurs in plants (Ketring et al., 1982). Since the plant is not growing exponentially and does not need all the nutrients available for growth, metabolic processes such as the TCA cycle slow (Ketring et al., 1982, Smirnoff, 1995). The decrease in growth rate results in a decreased need to use and synthesize nutrients, including niacin. Therefore, nutrients present or slowly synthesized through the night are compartmentalized and stored, which would have otherwise been utilized for nutrient synthesis and metabolism (Ketring et al., 1982). This has been offered as an explanation for cases where plants grown in environments that experience these lower night temperatures are often higher in vitamins such as niacin compared to plants grown in warmer night temperatures (Gustafson, 1953).
In addition to temperature, light also plays a role in vitamin synthesis. Light supplies energy for photosynthesis and the variation of light throughout the day has an immediate effect on plant metabolism (Smirnoff, 1995). The duration of light increases niacin biosynthesis; therefore, during the night, vitamin synthesis slows (Mozafar, 1993). At lower temperatures, vitamin synthesis and requirements are reduced and at cooler night temperatures, respiration and general plant metabolism slows, with the result that any excess niacin will accumulate in the plant (Smirnoff, 1995).
Using the 2008 UPPT samples, the effects of roasting on the niacin content was evaluated. Niacin is an extremely stable vitamin that can withstand exposure to heat and light (Morris et al., 2004) and that fact was verified by this study. Across all locations, significant differences were found only between 2008 raw and roasted Florunner (p<0.05). Average roasting losses varied from 0 to 9 percent, which would be an advantage circumstance since the majority of peanuts consumed in the US are roasted (American Peanut Council, 2010).
The Core of the Core samples showed a two-fold range in niacin concentration and all were higher in niacin than the Florunner and the NC7 samples grown at the same location that year. Although the Core samples in this study all came from one location (NC), if they follow the same pattern as the Florunner and the NC7, it may be expected that this set of Core of the Core samples would have higher niacin levels at other locations, particularly in the western region.
The change in niacin with geographic location represents an obvious environmental effect. The significant difference between Florunner and NC7 in niacin content at all 10 UPPT locations and the similar pattern of variation with respect to location indicated that genotypic variation for niacin expression may exist between the two lines. Analysis of 2008 samples of the Core of the Core grown in NC with the Florunner and NC7 samples grown there that year supports the idea that genetic variation may exist. If the higher niacin levels in Core of the Core samples in under sufficient genetic control, then useful germplasm may be found and exploited by breeders.
Conclusion
Environment was found to influence the niacin concentration in raw peanuts. Peanuts grown in regions with lower night temperatures tended to have increased niacin concentrations. The differences may be attributable to a slowing of respiration and other metabolic processes at cooler temperatures. Slower metabolism does not require the normal amount of specific vitamins to be involved in energy transport, and thus, less niacin is used and is accumulated in the plant. Genetic variation in niacin expression was suggested by the consistent differences in niacin concentration between Florunner and NC7 at each of 10 growing locations in 2008. Significant differences among the Core of the Core germplasm, Florunner and NC7 grown in NC in 2008 also support the theory that useful genetic variation may exist for the production of niacin in the peanut seed.
Acknowledgements
The authors thank all the breeders involved in the UPPT for their sample contributions.
Literature Cited
American Peanut Council 2010 About the Peanut Industry. www.peanutusa.com/USA/Index.cfm. (accessed 26 Jan 2010).
AOAC 1990 Official Methods of Analysis of the Association of Official Analytical Chemists, 15th ed Helrich K. (ed.) Association of Official Analytical Chemists, Inc Arlington, VA .
Arditti J. and Tarr J. B. 1979 Niacin biosynthesis in plants. Amer. J. Bot 66 : 1105 – 1113 .
Branch W. D. 2008 Uniform Peanut Performance Tests. 2007. Coastal Plain Exp. Sta. Res. Prog. Rep. No. 4-08, 25 pp.
Branch W. D. 2009 Uniform Peanut Performance Tests. 2008. Coastal Plain Exp. Sta. Res. Prog. Rep. No. 4-09, 25 pp.
Brumley C. S. and Sosebee R. E. 1978 Influence of soil temperature on niacin and thiamine in honey mesquite. Plant Soil 50 : 13 – 23 .
Burke J. J. , Chen J. , Rowland D. L. , Sanders T. H. , and Dean L. L. 2009 Temperature effects on carbohydrates of hydroponically-grown peanuts. Peanut Sci 36 : 150 – 156 .
Canty J. L. , Canty W. S. , Breitbart S. , and Frischling D. A. B. 2010 Weatherbase. Canty and Associates LLC. [Accessed on 2010 February 11]. Available from: http://www.weatherbase.com/ .
Casini C. , Dardanelli J. L. , Martinez M. J. , Calzarini M. , Corgogno C. S. , and Nassetta M. 2003 Oil quality and sugar content in peanuts (Arachis hypogaea) grown in Argentina: Their relationship with climatic variables and seed yield. J. Agric. Food Chem 51 : 6309 – 6313 .
Costet P. 2010 Molecular pathways and agents for lowering LDL-cholesterol in addition to statins. Pharm. Therap 126 : 263 – 278 .
Gustafson F. G. 1953 Influence of photoperiod on thiamine, riboflavin and niacin content in green plants. Amer. J. Bot 40 : 256 – 259 .
Hunt S. M. and Groff J. L. 1990 The Vitamins. pp. 170 – 263 In Advanced Nutrition and Human Metabolism West Publishing Co Boca Raton, FL .
Holbrook C. C. , Anderson W. F. , and Pittman R. N. 1993 Selection of a core collection from the U. S. germplasm collection of peanut. Crop Sci 33 : 859 – 861 .
Holbrook C. C. and Dong W. 2005 Development and evaluation of a mini core collection for the U. S. peanut germplasm collection. Crop Sci 45 : 1540 – 1544 .
Ketring D. , Brown R. , Sullivan G. , and Johnson B. 1982 Growth physiology. pp. 411 – 457 In Pattee H. and Young C. (eds.) Peanut Science and Technology American Peanut Research and Education Society, Inc Yoakum, TX .
Lee J. M. S. , Robson M. D. , Yu L. M. , Shirondania C. C. , Cunnington C. , Kylintiveas I. , Digby V. E. , Bannister T. , Handa A. , Niesmann F. , Durrington P. N. , Channnon K. M. , Neubauer S. , and Choudhury R. P. 2009 Effects of high dose modified-release nicotinic acid on atheroschlerosis and vascular function. A randomized, placebo-controlled, magnetic resonance imaging study. J. Amer. Coll. Cardio 54 : 1787 – 1794 .
McCormick D. B. 1988 Niacin. pp. 370 – 375 In: Shils M. E. and Young V. R. (eds.) Modern Nutrition in Health and Disease Lea and Febiger Philadelphia .
McMeans J. L. , Sanders T. H. , Wood B. W. , and Blankenship P. D. 1990 Soil temperature effects on free carbohydrate concentrations in peanut (Arachis hypogaea L.). Seed. Peanut Sci 17 : 31 – 35 .
Morris A. , Barnett A. , and Burrows O. J. 2004 Effect of processing on nutrient content of foods. J. Pub. Health 37 : 160 – 164 .
Mozafar A. 1993 Climate and plant vitamins. pp. 89 – 155 In Plant Vitamins: Agronomic, Physiology, and Nutritional Aspects CRC Press, Inc Boca Raton, FL .
Orten J. M. and Neuhaus O. 1970 Water-soluble vitamins (Nicotinamide). pp. 808 – 811 In Orten J. M. and Neuhaus O. (eds.) Biochemistry. 8th edition C.V. Mosby Co St. Louis, MO .
Rolland F. , Moore B. , and Sheen J. 2002 Sugar sensing and signaling in plants. Plant Cell. Suppl S185 – S205 .
Sanders T. H. , Vercellotti J. R. , Crippen K. L. , and Civille G. V. 1989 Effects of maturity on roast color and descriptive flavor of peanuts. J. Food. Sci 54 : 475 – 477 .
Shah S. , Ceska R. , Gil-Extremera B. , Paolini J. F. , Giezek H. , Vandormael K. , Mao A. , Sisk C. M. , and Maccubbin D. 2010 Efficacy and safety of extended-release niacin/laropiprant plus statin vs. doubling the dose of statin in patients with primary percholesterolaemia or mixed dyslipidoemia. Int. J. Clin. Prac 64 : 727 – 738 .
Silva R. S. and Ferreira F. H. 2009 The effects of extended-release niacin for increasing high-density lipoprotein cholesterol levels: effectiveness and tolerability. Ather. Suppl 10 : 5 – 6 .
Slaughter S. and Delwiche L. 2010 SAS 9.1.3 Help and Documentation. pp. 1 – 388 In The Little SAS Book for Enterprise, 4.2 SAS Press Cary, NC .
Smirnoff N. 1995 Metabolism Flexibility in Relation to the Environment. pp. 1 – 13 In Smirnoff N. (ed.) Environmental and Plant Metabolism: Flexibility and Acclimation BIOS Scientific Publishers Limited Oxford .
Sorrentino S. A. , Besler C. , Rohrer L. , Meyer M. , Heinrich K. , Bahlmann F. H. , Mueller M. , Horrath T. , Doerries E. , Heinemann M. , Flemmer S. , Markowski A. , Manes C. , Bahr M. J. , Haller H. , von Eckardstein A. , Drexler H. , and Landmesser U. 2010 Endothelial-vasoprotective effects of high-density lipoprotein are impaired in patients with type 2 diabetes mellitus but are improved after extended-release niacin therapy. Circ 121 : 110 – 122 .
USDA, U.S. Department of Agriculture, Agricultural Research Service 2009 USDA National Nutrient Database for Standard Reference, Release 22. Nutrient Data Laboratory Home Page, http://www.ars.usda.gov/ba/bhnrc/ndl. (accessed June 24, 2010).
USDA-ARS 2009 Uniform Peanut Performance Tests Chemical, Sensory and Shelf-life Properties. Data Presented by Location. USDA-ARS-SAA Market Quality and Handling Research Unit Raleigh, NC http://152.1.118.27/downloads.htm. (accessed July 06, 2010).
Wu B. J. , Yan L. , Charlton F. , Witting P. , Barter P. J. , and Rye K. A. 2010 Evidence that niacin inhibits acute vascular inflammation and improves endothelial dysfunction independent of changes in plasma lipids. Arter. Throm. Vas. Biol 30 : 968 – 975 .
Notes
- Department of Food, Bioproc. Nutr. Sci., Box 7624, N. C. State Univ., Raleigh, NC 27695-7624. [^]
- Department of Crop Sci.. Box 7629, N.C. State Univ., Raleigh, NC 27696-7629. [^]
- USDA-ARS Market Quality and Handling Res. Unit, Raleigh, NC 27695-7624. [^] *Corresponding author: E-mail: lisa.dean@ars.usda.gov.
Author Affiliations