Introduction
Over the past decade, the major emphasis of peanut breeding programs in the U.S has been on the development of cultivars resistant to tomato spotted wilt virus (TSWV) (Culbreath et al., 2003). Although these new varieties have outstanding resistance to TSWV and other diseases, some may have characteristics, such as reduced germination or poor seed vigor (Morton et al., 2008). In recent years, the cultivars DP-1 (Gorbet and Tillman, 2008) and Georgia-01R (Branch, 2002) had great potential due to their multiple disease resistances but were not widely planted by growers due to poor seed germination and vigor issues (Beasley, 2007).
Peanut seed quality and germination are influenced by many factors including water and calcium availability during pod fill, maturity at harvest, curing and storage at the recommended moisture and temperature levels, and shelling and conditioning with minimal mechanical damage (Guerke, 2005). Because of the recent problems with the vigor and germination of new cultivars, questions have been raised about the potential of peanut herbicides to have an influence on seed germination.
Numerous studies have been conducted on the effects of herbicides on soybean seed. Early research indicated that herbicides do not significantly alter oil content and cause only relatively small changes in fatty acid composition of soybean seed (Penner and Meggitt, 1970). Subsequent research confirmed that herbicides do not cause important alterations in soybean seed constituents (Stoller et al., 1973). More recently, Bradley et al. (2002) found that soybean herbicides had little or no effect on seed germination, protein, and oil content. However, herbicides used as preharvest desiccants, such as glyphosate, paraquat, and sodium chlorate, applied before soybean maturity (R7) have the potential to reduce soybean seed germination (Bennett and Shaw, 2000).
Twelve herbicide active ingredients are labeled for postemergence use in peanut, but only imazapic, imazethapyr, 2,4-DB, and chlorimuron are highly systemic in the peanut plant (Senseman, 2007) and thus have potential to influence seed characteristics. Of these four herbicides, only imazapic and 2,4-DB are used over significant acreages (NASS, 2009). The 2,4-DB molecule is not readily absorbed or phytotoxic, but the secondary metabolite (2,4-D) resulting from beta oxidation is both mobile and phytotoxic (Wathana et al., 1972; Wehtje et al., 1992) in many species. Much research can be found that documents peanut plant injury and/or yield response to 2,4-DB and imazapic (Brecke and Stephenson, 2006; Grey and Wehtje, 2005; Grichar, 2007; Hicks et al., 1998; Wehtje et al., 1993; York et al., 1993), however, little research has been reported on the effects that herbicides may have on seed quality. Lancaster et al. (2005) reported that 2,4-DB reduced seed germination of sickelpod (Senna obtusifolia L. Irwin and Barneby) 30 to 40% when applied just prior to flowering or during peak flowering. Ketchersid et al. (1978) studied the effects of 2,4-DB in peanut and noted enlarged pods with less total sound mature kernels (TSMK), but no yield reduction. Therefore, the objective of this research was to evaluate the effect of imazapic and 2,4-DB on peanut yield, grade, and subsequent seed properties (germination and vigor). TSWV incidence on peanut was also evaluated.
Materials and Methods
Field studies were conducted in 2005 and 2006 at the Ponder Farm near Tifton, GA and at the Bolton Research Farm near Dawson, GA. Soils were Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults) and Red Bay sand (fine-loamy, kaolinitic, thermic Rhodic Kandiudults) at Tifton and Dawson, respectively. Organic matter ranged from 1.0 to 1.1% with pH from 6.0 to 6.5 at Tifton and 1.0% organic matter and pH 6.2 at Dawson.
Peanut was sown in twin rows, spaced 90 cm apart at both locations on conventionally prepared seedbeds. Distance between paired twin rows was 23 cm. Experimental plot size in Tifton was two twin rows by 7.6 m in length, while Dawson plots were six twin rows by 9 m in length. Planting dates were May 9, 2005 at both Tifton and Dawson, and May 10 and May 16, 2006 for Tifton and Dawson, respectively. Rainfall was supplemented with overhead linear irrigation at Tifton, while surface drip irrigation was used at Dawson. Pendimethalin or ethalfluralin plus diclosulam was applied preemergence to the entire test area at both locations to suppress grass and broadleaf weeds. Weed-free conditions were maintained at both locations through mechanical cultivation and hand-weeding. Clethodim was applied for postemergence control of annual and perennial grasses that escaped preemergence herbicides and cultivation. Standard cultural practices for peanut production were followed using University of Georgia Extension recommendations.
The treatments were arranged in a split-plot design with four replications at each location. Peanut cultivars (main plots) were Georgia Green (Branch, 1996), Georgia-01R, and C-99R (Gorbet and Shokes, 2002). Herbicides (sub-plots) were imazapic at 71 g ai ha−1 applied at 30 or 45 d after planting (DAP); 2,4-DB at 270 g ai ha−1 applied at 75 or 90 DAP; and a non-treated control. All herbicides were applied with CO2-pressurized spray equipment calibrated to deliver 140 L ha−1 total spray volume and included crop oil concentrate at 1% v/v.
Prior to digging, peanut was visually rated for incidence of TSWV. The number of 31 cm sections (a single pair of twin rows) of row containing symptomatic plants was counted for each experimental plot according to Culbreath et al. (1997). Data were transformed into percentage based on total row length and reported as percent infection. Maturity was determined using the hull scrape method (Williams and Drexler, 1981) for each variety and plots were mechanically dug and inverted accordingly. The cultivar Georgia Green was dug approx. 2 wk prior to either Georgia-01R or C-99R at both locations in both years. Plots were mechanically harvested using a stationary plot thresher to ensure sample integrity. Yields were transformed to kg ha−1 at 10% moisture. A 500-g sample was collected randomly using a riffle divider then shelled to obtain medium-sized seed. Medium-sized seed were treated with commercial seed treatment fungicide3 and sent to the Georgia Department of Agriculture Seed Lab in Tifton, GA for germination analysis. Standard (25 C) and cold (15 C) germination tests were conducted. The cold germination test provides some indication of peanut seedling vigor (Guerke, 2005). A 1500-g sample was collected similarly and analyzed for grade, including total sound mature kernels (TSMK), sound splits, and damaged kernels4.
Yield, grade, TSWV incidence, standard germination and cold germination data were subjected to Mixed Models analysis of variance5. Year and year nested within location were used as random effects. When main effects or interactions were significant (P ≤ 0.05), Duncan's MRT test was used for mean separation. All percentage data were arcsine transformed prior to analysis. Data transformation did not improve homogeneity of variance, thus actual percentage data are presented.
Results and Discussion
Herbicide Effects
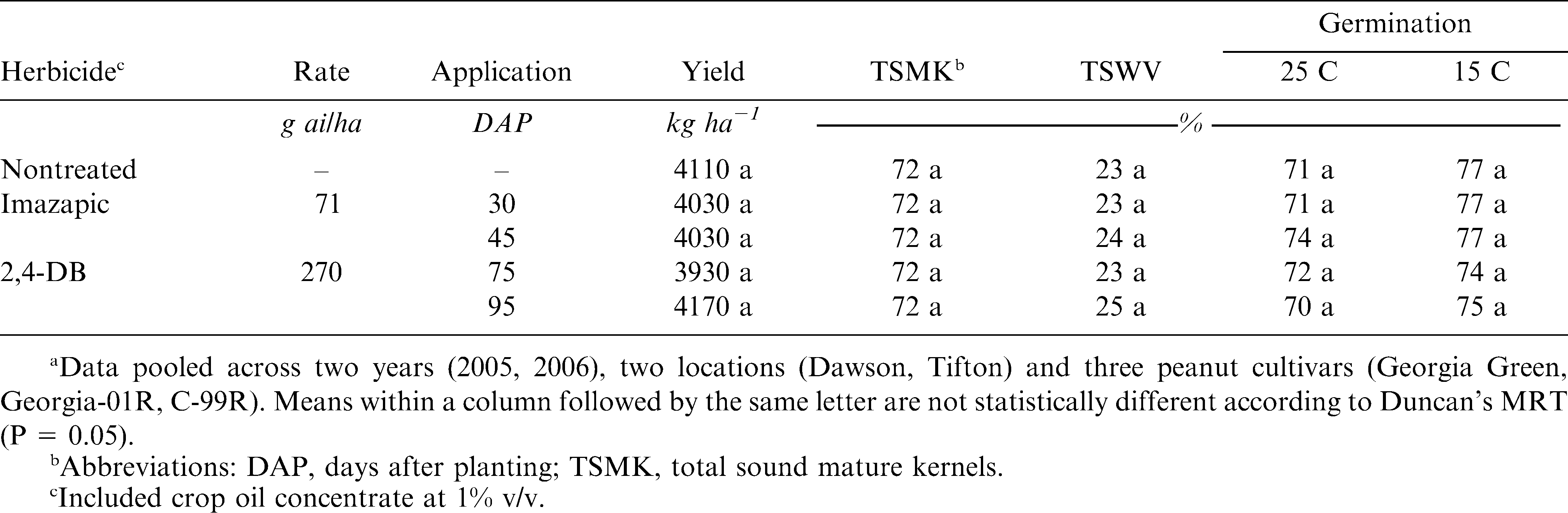
The interaction of herbicide, year, location, and cultivar was not significant. Therefore, data for herbicides are discussed pooled across year, location, and cultivar. Neither imazapic applied at 30 or 45 DAP nor 2,4-DB applied at 75 or 95 DAP effected peanut yield, seed quality, or TSWV incidence (Table 1). Peanut yield ranged from 3930 to 4170 kg ha−1, and light to moderate TSWV incidence (24%) was observed. Peanut seed quality was not affected by herbicide with an average grade of 72 and germination rates of 76% and 72% for standard and cold germination, respectively. These results are similar to Ketchersid et al. (1978) who reported that 2,4-DB applied at rates up to 0.45 kg ha−1 did not adversely affect Spanish-type peanut yield or seed quality. The rates used by Ketchersid et al. (1978) are approximately twice the current recommended rate for use in peanut. The current study utilized modern runner-type cultivars and also is distinctive in that 2,4-DB was applied much later in the growing season. Several studies have reported no effect of imazapic on peanut yield and grade despite visual observations of plant injury (Dotray et al., 2001; Grichar, 1997; Webster et al., 1997). However, these earlier studies did not consider seed germination, vigor, or interactions with TSWV.
The interactions of factors other than herbicide including cultivar × year, cultivar × location, and year × cultivar × location were significant for all five response variables (data not shown). Accordingly, data for cultivar effects were separated by location and/or year and will be presented by location.
Dawson
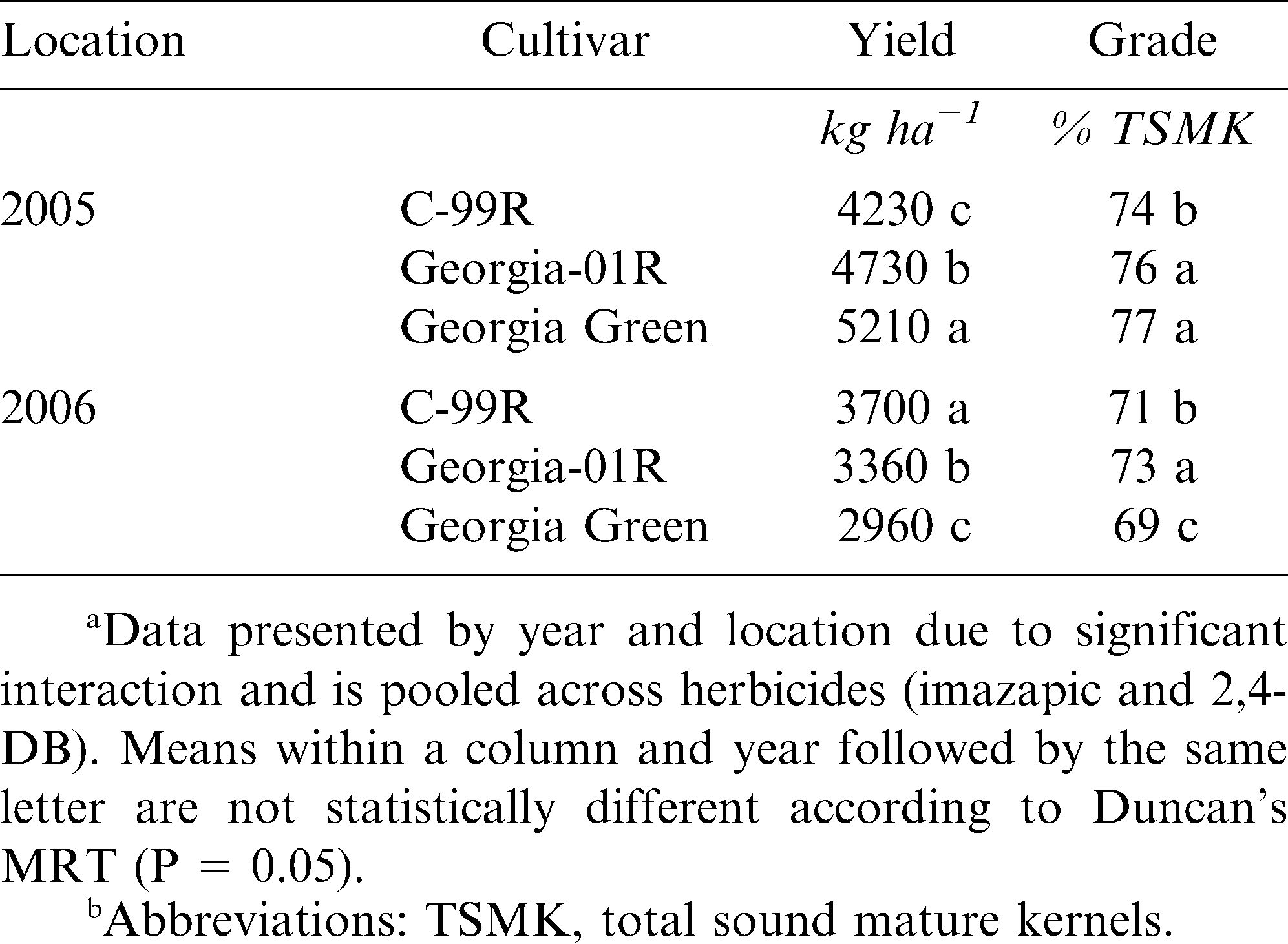
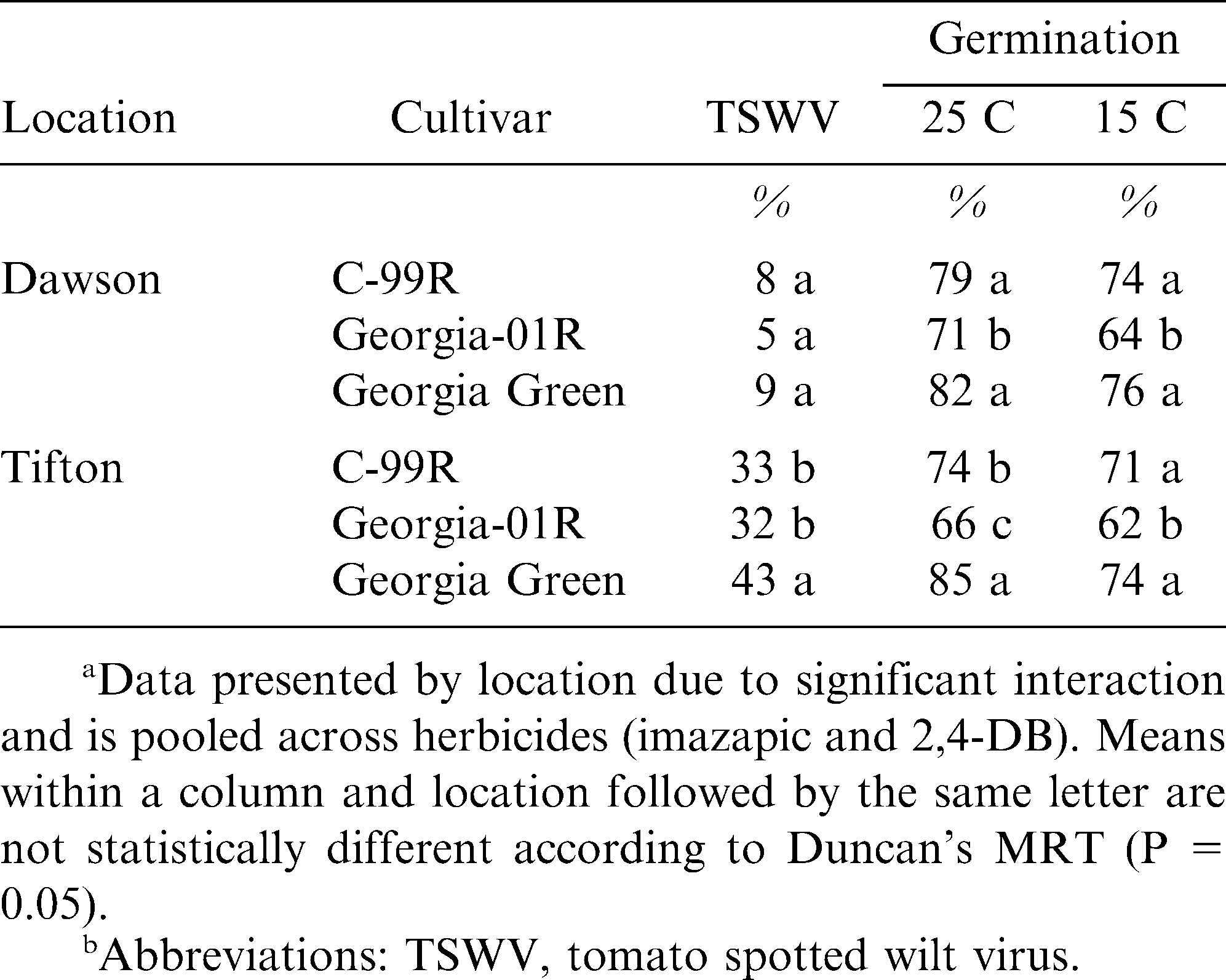
The interaction of year × location × cultivar was significant for both peanut yield and grade. Georgia Green was the highest yielding cultivar in 2005, but was the lowest yielding cultivar in 2006 (Table 2). Similarly, Georgia Green had the highest in grade in 2005, but the lowest grade in 2006. Yields in 2005 were 40% greater for all three cultivars versus 2006. The experimental field utilized in 2005 was previously in unimproved bahiagrass (Paspalum notatum Fluegge) pasture with no peanut grown in recent history. The portion of the field planted in 2006 had been in continuous peanut rotation since 2004. Thus, the high yields observed in 2005 were expected and likely due to decreased incidence of many common fungal diseases (Katsvairo et al., 2007; Norden et al., 1977). Georgia Green performed well in 2005 in the absence of disease pressure; however, in 2006 when incidence of leafspot (caused by both Cercospora spp. and Cercosporidium spp.) and white mold (caused by Sclerotium rolfsii Sacc.) increased, Georgia Green performance decreased. Conversely, Georgia-01R and C-99R produced high yields in 2005 and were superior to Georgia Green in 2006. Several studies have described the increased tolerance of Georgia-01R and C-99R cultivars to common diseases found in Southeastern U.S. peanut production (Cantonwine et al., 2006; Gorbet and Shokes, 2002). Although disease pressure increased in 2006 at this location, TSWV incidence was low with a maximum rating of 9% (Table 3). Accordingly, no significant response to TSWV was demonstrated.
Standard germination and cold germination were significant for cultivar × location, but not year. The cultivar Georgia-01R showed reduced germination at both temperatures compared to C-99R or Georgia Green (Table 3). Morton et al. (2008) have documented the reduced seed germination and vigor of several late-maturing peanut cultivars. Georgia-01R is considered a late-maturing peanut (Branch, 2002) and has shown tendencies towards reduced germination and vigor in on-farm demonstration. However, yields indicate that despite slow or inconsistent germination, the less determinant growth habit of this later-maturing peanut often compensates for less than optimal stand establishment (Tables 2 and 4).
Tifton
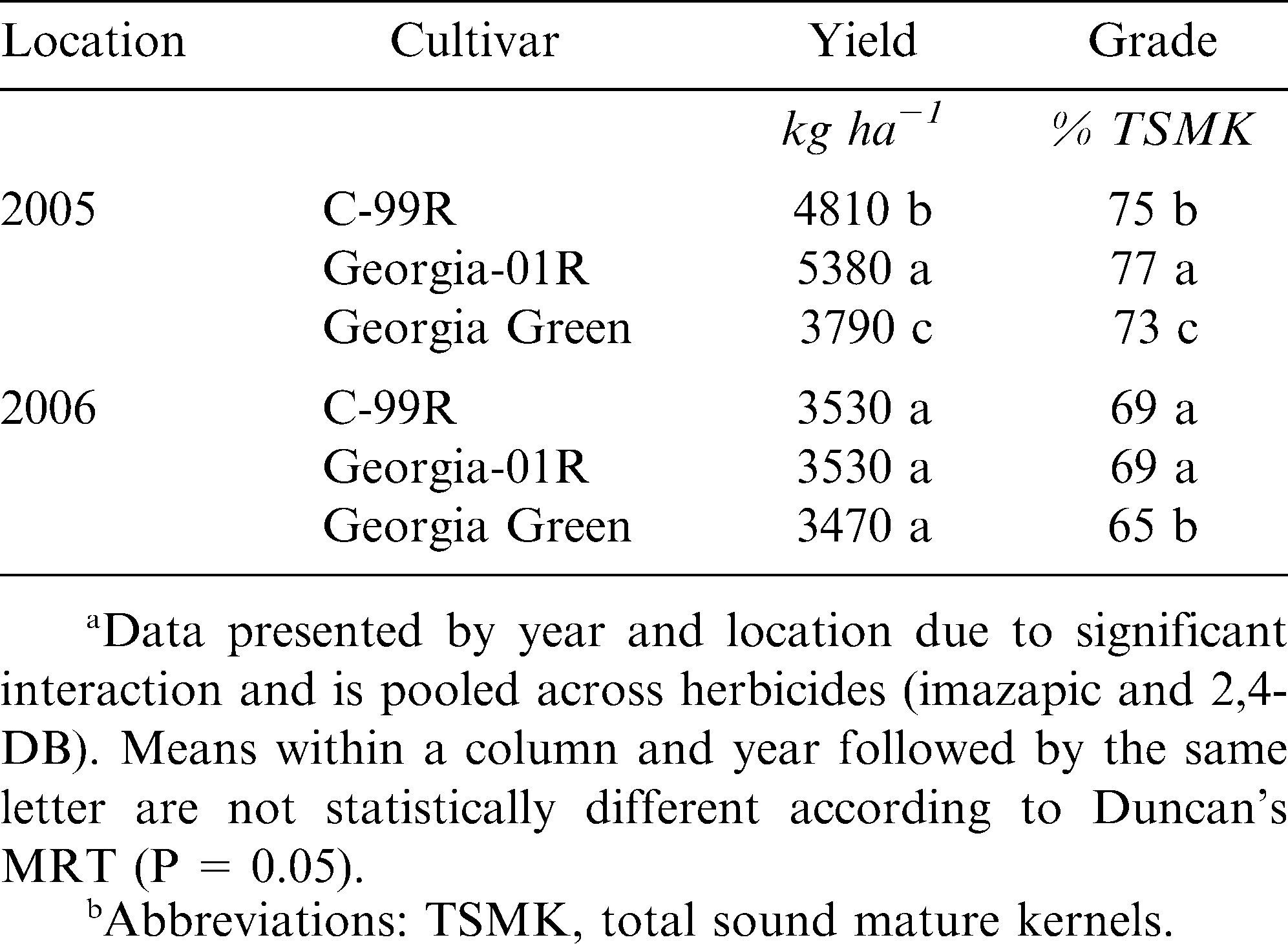
The interaction of year × location × cultivar was significant for both peanut yield and grade. Georgia-01R showed highest yield at Tifton for 2005, but no differences were detected between cultivars for 2006 (Table 4). All cultivars had high grades in 2005, with Georgia-01R exceeding both C-99R and Georgia Green (Table 4). Peanut grades were decreased overall during 2006, with both C-99R and Georgia-01R producing a higher percentage of sound mature kernels than Georgia Green. Generally, 2006 was a hotter and drier year when compared to 2005. This may have contributed to the reduced peanut yield and grades in 2006.
Incidence of TSWV and peanut germination data were significant for the interaction of location × cultivar, but not year. TSWV incidence was greater at Tifton both years when compared to Dawson, exhibiting a 3 to 4 fold increase in incidence (Table 3). As expected, the cultivar Georgia Green showed highest incidence of TSWV compared to either C-99R or Georgia-01R. Continuing a trend shown at the Dawson location, Georgia Green had highest germination response at both temperatures (25 and 15 C), while Georgia-01R consistently performed poorly in germination and vigor response.
Summary and Conclusions
Despite notable differences in cultivar response to growing conditions, neither imazapic nor 2,4-DB influenced peanut performance. Cultivar differences were the dominant effect in this series of studies as opposed to herbicides. As peanut germination and stand establishment concerns remain high, producers and researchers should focus on other production practices or post-harvest mechanisms that may be decreasing peanut performance rather than the negligible impacts of herbicides. Georgia-01R had reduced seed germination when compared to the other cultivars in this study. This would help explain the establishment problems that have been observed when this cultivar is planted.
Literature Cited
Beasley J. P.
2007
Peanut cultivars for 2007 in 2007 Peanut Update.
Univ. of Georgia, Dept. of Crop & Soil Sciences Publication CSS-07-0108,
7
–
8
.
Bennett A. C. and Shaw D. R. 2000 Effect of preharvest desiccants on Group IV Glycine max seed viability. Weed Sci 48 : 426 – 430 .
Bradley C. A. , Hartman G. L. , and Wax L. M. 2002 Quality of harvested seed associated with soybean cultivars and herbicides under weed-free conditions. Plant Dis 86 : 1036 – 1042 .
Branch W. D. 1996 Registration of ‘Georgia Green’ peanut. Crop Sci 36 : 806 .
Branch W. D. 2002 Registration of ‘Georgia-01R’ peanut. Crop Sci 42 : 1750 – 1751 .
Brecke B. J. and Stephenson D. O. 2006 Weed management in single- vs. twin-row peanut (Arachis hypogaea). Weed Technol 20 : 368 – 376 .
Culbreath A. K. , Todd J. W. , and Brown S. L. 2003 Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol 41 : 53 – 75 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Shokes F. M. , and Pappu H. R. 1997 Field response of new peanut cultivar UF 91108 to tomato spotted wilt virus. Plant Dis 81 : 1410 – 1415 .
Dotray P. A. , Baughman T. A. , Keeling J. W. , Grichar W. J. , and Lemon R. G. 2001 Effect of imazapic application timing on Texas peanut (Arachis hypogaea). Weed Technol 15 : 26 – 29.
Gorbet D. W. and Shokes F. M. 2002 Registration of ‘C-99R’ peanut. Crop Sci 42 : 2207 .
Gorbet D. W. and Tillman B. L. 2008 Registration of ‘DP-1’ peanut. J. Plant Registrations 2 : 200 – 204 .
Grey T. L. and Wehtje G. R. 2005 Residual herbicide weed control systems in peanut. Weed Technol 19 : 560 – 567 .
Grichar W. J. 1997 Control of Palmer amaranth (Amaranthus palmeri) in peanut (Arachis hypogaea) with postemergence herbicides. Weed Technol 11 : 739 – 743 .
Grichar W. J. 2007 Horse purslane (Trianthema portulacastrum), smellmelon (Cucumis melo), and Palmer amaranth (Amaranthus palmeri) control in peanut with postemergence herbicides. Weed Technol 21 : 688 – 691 .
Guerke W. R. 2005 Evaluating peanut (Arachis hypogaea L.) seed vigor. Seed Technol 27 1 : 121 – 126 .
Hicks T. V. , Wehtje G. R. , and Grey T. L. 1998 The interaction of pyridate and 2,4-DB in peanut (Arachis hypogaea), Florida beggarweed (Desmodium tortuosum), and sicklepod (Senna obtusifolia). Weed Sci 46 : 284 – 288 .
Katsvairo T. W. , Wright D. L. , Marois J. J. , Hartzog D. L. , Balkcom K. B. , Wiatrak P. P. , and Rich J. R. 2007 Performance of peanut and cotton in a bahiagrass cropping system. Agron. J 99 : 1245 – 1251 .
Ketchersid M. L. , Boswell T. E. , and Merkle M. G. 1978 Effects of 2,4-DB on yield and pod development in peanuts. Peanut Sci 5 : 35 – 39 .
Lancaster S. H. , Jordan D. L. , Spears J. F. , York A. C. , Wilcut J. W. , Monks D. W. , Batts R. B. , and Brandenburg R. L. 2005 Sicklepod (Senna obtusifolia) control and seed production after 2,4-DB applied alone and with fungicides or insecticides. Weed Technol 19 : 451 – 455.
[NASS] National Agricultural Statistics Service 2009 Agricultural Chemical Use Database. http://www.pestmanagement.info/nass/app_statcs3_crop.cfm. Accessed 16-Apr-2009.
Morton B. R. , Tillman B. L. , Gorbet D. W. , and Boote K. J. 2008 Impact of seed storage environment on field emergence of peanut (Arachis hypogaea L.) cultivars. Peanut Sci 35 : 108 – 115 .
Norden A. J. , Perry V. G. , Martin F. G. , and NeSmith J. 1977 Effect of age of bahiagrass sod on succeeding peanut crops. Peanut Sci 4 : 71 – 74 .
Penner D. and Meggitt W. F. 1970 Herbicide effects on soybean [Glycine max (L.L Merrill)] seed lipids. Crop Sci 10 : 553 – 555 .
Senseman S. A. 2007 Herbicide Handbook, 9th Ed Weed Science Society of America Lawrence, KS 458 .
Stoller E. W. , Weber E. J. , and Wax L. M. 1973 The effects of herbicides on soybean seed constituents. J. Environ. Qual 2 : 241 – 244 .
Wathana S. , Corbin F. T. , and Waldrep T. W. 1972 Absorption and translocation of 2,4-DB in soybean and cocklebur. Weed Sci 20 : 120 – 123 .
Webster T. M. , Wilcut J. W. , and Coble H. D. 1997 Influence of AC 263,222 rate and application method on weed management in peanut (Arachis hypogaea). Weed Technol 11 : 520 – 526 .
Wehtje G. , Wilcut J. W. , and McGuire J. A. 1992 Paraquat behavior as influenced by 2,4-DB in peanut and selected weeds. Peanut Sci 19 : 51 – 55 .
Wehtje G. , Wilcut J. W. , and McGuire J. A. 1993 Absorption, translocation, and phytotoxicity of chlorimuron and 2,4-DB mixtures in peanut (Arachis hypogaea) and selected weed species. Weed Sci 41 : 347 – 352 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
York A. C. , Wilcut J. W. , and Grichar W. J. 1993 Interaction of 2,4-DB with postemergence graminicides. Peanut Sci 20 : 57 – 61 .
Notes
- Research Agronomist, USDA-ARS National Peanut Research Laboratory, P.O. Box 509, Dawson, GA 39842. [^]
- Assoc. Professor, Dept. of Crop and Soil Sci., The University of Georgia, Tifton Campus, 104 Research Way, Tifton, GA 31793. [^] *Corresponding author email: wilson.faircloth@ars.usda.gov
- Vitavax PC, Bayer CropScience, P.O. Box 12014, Research Triangle Park, NC 27709. [^]
- Georgia Federal State Inspection Service, 2323 N. Slappey Blvd., Albany, GA 31701. [^]
- Statistical Analysis Software 9.1, SAS Institute Inc., Cary NC 27512. [^]
Author Affiliations