Introduction
Biological nitrogen fixation (BNF) occurs in peanut (Arachis hypogaea L.) and other leguminous plants when nitrogen (N)-fixing Bradyrhizobia bacteria infect roots and convert atmospheric N into a plant useable form (Schiffman and Alper, 1968; Shimshi et al., 1967; Walker et al., 1976). Farmers rely on existing or native rhizobia in soil for this process or apply commercially available inoculant during planting to ensure adequate BNF. Commercial inoculants can be applied to the planter hopper box or in the seed furrow as granular or liquid products (Lanier et al., 2005). Although commercial inoculants are relatively inexpensive, ability to correct N deficient peanut is expensive if Bradyrhizobia is not viable, the formulated inoculant is not distributed uniformly in the hopper box with seed, formulated inoculant placement in the seed furrow is not adequate for infection, peanut is planted shallow and formulated inoculant does not remain viable due to extremes in soil temperature, the rate of formulated inoculant is too low, or inoculant is adversely affected in the spray solution. When inoculant fails to allow BNF, applying N directly to peanut to correct N deficiencies is expensive, and erratic response to N has been observed (Lanier et al., 2005).
In recent years, in addition to registrations later in the season, the fungicides azoxystrobin and tebuconazole have been marketed for use in the seed furrow to suppress or control Cylindrocladium black rot (CBR) (caused by Cylindrocladium parasiticum) (Phipps, 2003). Although currently not registered for application in the seed furrow in peanut, the fungicides boscalid, pyraclostrobin, propiconazole, and trifloxystrobin may affect seedling diseases but response of peanut to these fungicides has not been reported. A commercial fertilizer (Asset® RTU, Helena Chemical Co., Memphis, TN) containing several essential elements is applied by some farmers in the seed furrow at planting due to perceived benefits in early season root and shoot growth. Determining if these pesticides and fertilizer are compatible with in-furrow application of Bradyrhizobia is important in developing crop management strategies. Tomato spotted wilt virus, a Tospovirus, is a major concern in North Carolina peanut production, and while several strategies have been implemented to reduce disease in peanut, additional control options are being explored (Brandenburg, 2009). The insecticide imidacloprid, while not registered for use in peanut, is applied in greenhouse float trays to reduce incidence of tomato spotted wilt virus in tobacco (Nicotiana tabacum L.) (McPherson, 2006). Although Todd et al. (1994) reported increased tomato spotted wilt of peanut when imidacloprid was applied compared to no insecticide or other treatments, research to determine the effect of imidacloprid on tomato spotted wilt in peanut in North Carolina is limited (Brandenburg, R. L., North Carolina State University, personal communication). Determining if imidacloprid and fertilizer are compatible with in-furrow application of Bradyrhizobia is important if imidacloprid is shown to be effective against tomato spotted wilt.
Interactions among agrichemicals applied to peanut are numerous, complex, and can be unpredictable. Efficacy of commercial liquid inoculants and survival of Bradyrhizobia is of particular concern when considering co-application of inoculant with liquid fertilizer, fungicides, and insecticides. Previous research (Jordan et al., 2006) indicated that acephate and tebuconazole did not affect Bradyrhizobia when co-applied in the seed furrow at planting. However, possible interactions of Bradyrhizobia applied as a liquid in mixture with other agrichemicals have not been documented completely in the literature. Therefore, research was conducted to determine the influence of selected fungicides, imidacloprid, and fertilizer on peanut response to Bradyrhizobia when co-applied in the seed furrow at planting.
Materials and Methods
The experiment was conducted in North Carolina during 2002 at the Peanut Belt Research Station located near Lewiston on a Norfolk sandy loam (fine-loamy, siliceous, thermic, Typic Paleudults) and in two separate fields at the Upper Coastal Plain Research Station located near Rocky Mount on a Goldsboro loamy sand (fine-loamy, siliceous, thermic Aquic Paleudalts). A second experiment was conducted in commercial fields near Faison, NC on a Autryville fine sandy loam (fine-loamy, siliceous, Typic Paleudults). The experiment was conducted in two separate fields during 2004 and 2006 and in one field during 2005 and 2007. The experiment was also conducted during 2004, 2006, and 2007 at the Upper Coastal Plain Experiment Station near Rocky Mount, NC on the Goldsboro loamy sand soil described previously. During 2005, the experiment was also conducted at the Peanut Belt Research Station near Lewiston, NC and the Tidewater Agricultural Research and Extension Center near Suffolk, VA. Soil at these locations was a Norfolk sandy loam. Peanut cultivar NC-V 11 (Wynne et al., 1991) was seeded in conventionally-prepared raised seedbeds at a rate designed to establish an in-row population of 13–15 plants/m. Aldicarb at 1.1 kg ai/ha was applied at planting prior to seed drop and before slit closure.
Treatments during 2002 included a commercial liquid inoculant containing Bradyrhizobia (Lift, formerly Nitragin Corp. and now EMD Biosciences., Brookfield, WI) alone or with azoxystrobin (0.44 kg ai/ha), boscalid (0.45 kg ai/ha), propiconazole plus trifloxystrobin (0.02 + 0.02 kg ai/ha), pyraclostrobin (0.16 kg ai/ha), and tebuconazole (0.20 kg ai/ha). During 2004–2007, treatments consisted of a no-inoculant control, a commercial liquid inoculant (Lift, Nitragin Corp., during 2004 or Optimize Lift, EMD BioSciences, during 2005–2007), and liquid inoculant plus azoxystrobin, fertilizer solution (Asset® RTU, containing 6% N, 20% P2O5, 5% K2O, 0.02% B, 0.05% Cu, 0.10% Fe, 0.05% Mn, 0.0005% Mo, and 0.05% Zn) at 2.4 L/ha, and pyraclostrobin. Additional treatments during 2005, 2006, and 2007 included imidacloprid (0.21 kg ai/ha) plus inoculant or tebuconazole (0.20 kg/ha) plus inoculant during 2004, 2006, and 2007. Lift and Optimize Lift deliver approximately 2.2 × 1012 viable cells of bacteria/ha when applied at 1.2 L/ha. Treatments were applied in 47 L/ha distilled water. Spray solution was applied after seed drop and before slit closure and contacted seed.
Two non-peanut crops separated peanut plantings during 2002, fields at Lewiston, and Suffolk during 2005. Three non-peanut crops (2004 and 2006) and five non-peanut crops separated peanut plantings in fields at Rocky Mount during 2007. Non-peanut crops at all locations were either corn (Zea mays L.) or cotton (Gossypium hirsutum L.). Peanut had not been planted in fields at Faison. Fields were fumigated with metam sodium two weeks prior to planting at Lewiston and Suffolk to control CBR. Fields at Faison and Rocky Mount did not have a history of CBR. With the exception of in-furrow treatments, all other production and pest management strategies were held constant and were based on Cooperative Extension recommendations for the region (Brandenburg, 2009; Jordan, 2009a 2009b; Shew, 2009).
In experiments conducted during 2002, peanut seedling emergence was determined within 2 weeks after planting (WAP). Seedling emergence was not recorded in subsequent years. Visual estimates representing a plant condition rating (PCR) were recorded in 2002 within two weeks prior to harvest on a scale of 0 to 100% where 0 = no disease symptoms in the peanut canopy and 100 = the entire canopy expressing disease symptoms. The primary disease present in 2002 was tomato spotted wilt (Shew, 2009). In subsequent years, tomato spotted wilt was not observed in the experiments. Peanut in fields near Faison did not exhibit symptoms of CBR or other soil borne pathogens. Color of the peanut canopy was determined approximately 60 days after planting in all experiments using a scale of 0 to 5 where 0 = yellow foliage due to N deficiency and 5 = peanut expressing a dark green color. Determining canopy color at this stage of peanut development minimized possible confounding of canopy color due to development of disease. While determining N concentration in peanut leaves or recording chlorophyll content would have been more informative than assessing the canopy visually, practitioners most likely will use a visual assessment for this comparison.
Peanut was dug and vines inverted in late September or early October based on pod mesocarp color (Williams and Drexler, 1981) using a composite sample of peanut treated with inoculant alone. Pods and vines were allowed to air dry in the field for 4 to 7 days after digging prior to combining. Final pod yield was adjusted to 8% moisture.
The experimental design was a randomized complete block with treatments replicated four times. Data for peanut emergence within two weeks after planting (2002), PCR (2002), canopy color (2004, 2006, and 2007), and pod yield from all years were subjected to analyses of variance. The analyses from 2004–2008 included the no-inoculant control, inoculant alone, and inoculant plus azoxystrobin, pyraclostrobin, or fertilizer (11 experiments). Separate analyses including the no-inoculant control and inoculant alone compared with inoculant plus imidacloprid (7 experiments) or inoculant plus tebuconazole (8 experiments) were performed because imidacloprid and tebuconazole were not included in all 11 experiments. Means were separated using Fisher's Protected LSD test at p ≤ 0.05.
Results and Discussion
Experiments during 2002
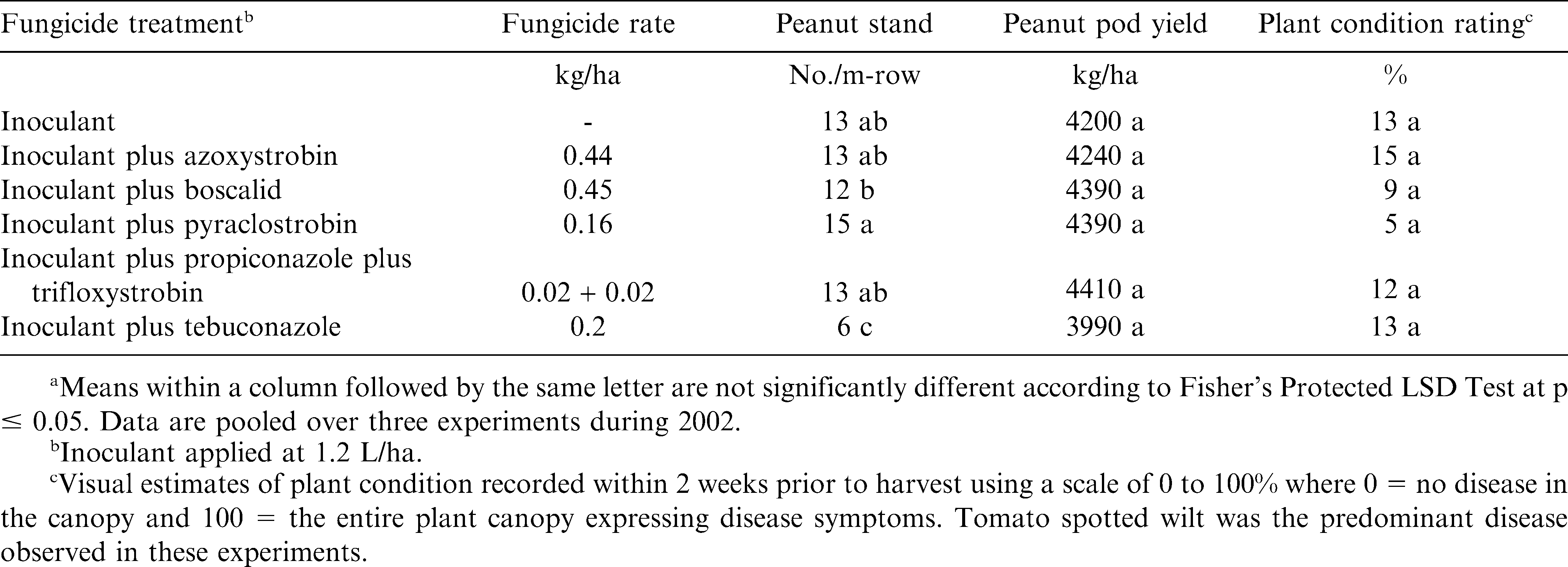
The interaction of experiment by treatment was not significant for peanut stand, pod yield, or PCR in the experiments conducted during 2002. When pooled over experiments, peanut stand 2 WAP was similar when inoculant was applied alone or with azoxystrobin, pyraclostrobin, or propiconazole plus trifloxystrobin (Table 1). Fewer peanut plants emerged 2 WAT when boscalid was applied with inoculant compared with inoculant plus pyraclostrobin (Table 1). However, when inoculant was applied with boscalid, peanut stand was similar to stand with inoculant only. Peanut stand was lower when tebuconazole was applied with inoculant compared with inoculant alone or when inoculant was co-applied with other fungicides (Table 1).
Pod yield and PCR, reflecting plants expressing visible symptoms of tomato spotted wilt virus, did not differ regardless of inoculant and fungicide treatment (Table 1). Delayed peanut emergence due to in-furrow application of tebuconazole does not always translate into a yield reduction (Jordan et al., 2006; Phipps, 2003). Previous research (Culbreath et al., 2003) indicated that fungicides do not affect incidence of tomato spotted wilt of peanut.
Experiments during 2004–2007
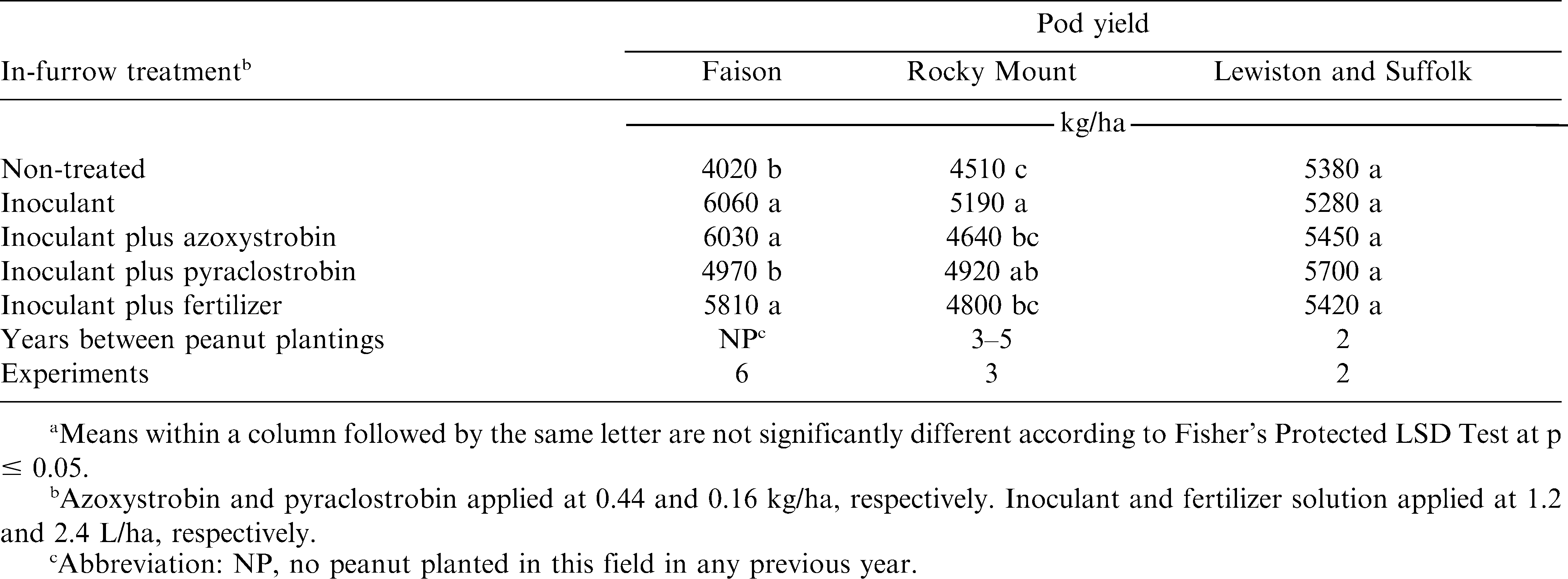
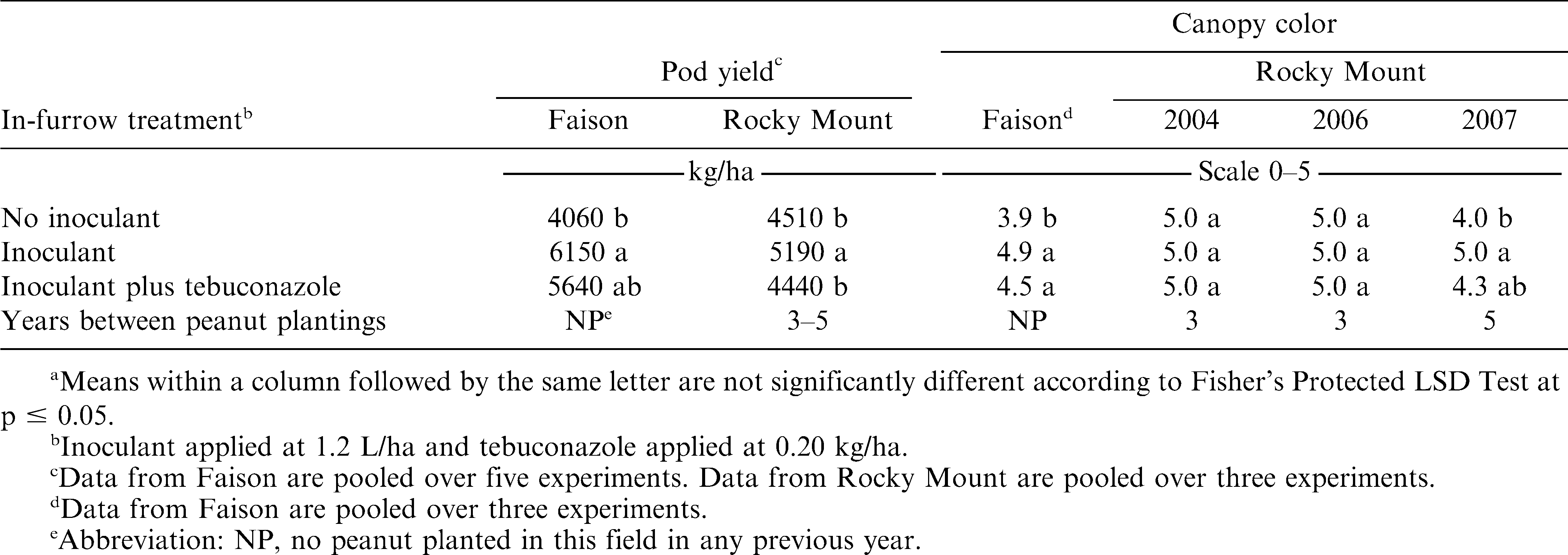
The interaction of experiment by in-furrow treatment was significant for pod yield when the eleven experiments were included in a single analysis. Separate analyses were performed for three groups (Faison, Rocky Mount, or Lewiston and Suffolk) due to previous cropping history and similar soil characteristics. Consequently, the interaction of experiment by in-furrow treatment was not significant for Faison, Rocky Mount, and Lewiston and Suffolk. However, the main effect of in-furrow treatment was significant at Faison and Rocky Mount but not for Lewiston and Suffolk. When pooled over six experiments at Faison, applying Bradyrhizobium inoculant alone or with azoxystrobin or fertilizer increased pod yield over the no-inoculant control (Table 2). Yield did not differ when comparing treatments including inoculant plus azoxystrobin or fertilizer and inoculant alone. In contrast, pod yield following application of inoculant plus pyraclostrobin was lower than inoculant plus azoxystrobin or fertilizer and was similar to the no-inoculant control. At Rocky Mount, applying inoculant alone increased pod yield over the no-inoculant control when data were pooled over the three years at this location (Table 2). In contrast to results at Faison, pod yield was lower when inoculant was applied with azoxystrobin or fertilizer compared with inoculant alone; yield following inoculant plus pyraclostrobin did not differ from inoculant alone. Pod yield following inoculant plus pyraclostrobin or fertilizer was similar to the no-inoculant control. No difference in pod yield was observed regardless of in-furrow treatment at Lewiston and Suffolk (Table 2).
Differences in yield when comparing the no-inoculant control with inoculant alone were expected at Faison and Rocky Mount, especially at Faison where peanut had not been planted in these fields in previous years. At Rocky Mount, three or five non-peanut crops separated peanut plantings, and response to inoculant was also anticipated. Lack of a response to inoculant was not unexpected at Lewiston and Suffolk due to the shorter rotations at these locations compared with Faison and Rocky Mount. Surprisingly, however, was the difference in peanut response to azoxystrobin, pyraclostrobin, and fertilizer when comparing results at Faison and Rocky Mount. No clear explanation exists for this response, and additional research is needed to elucidate the mechanism of this response.
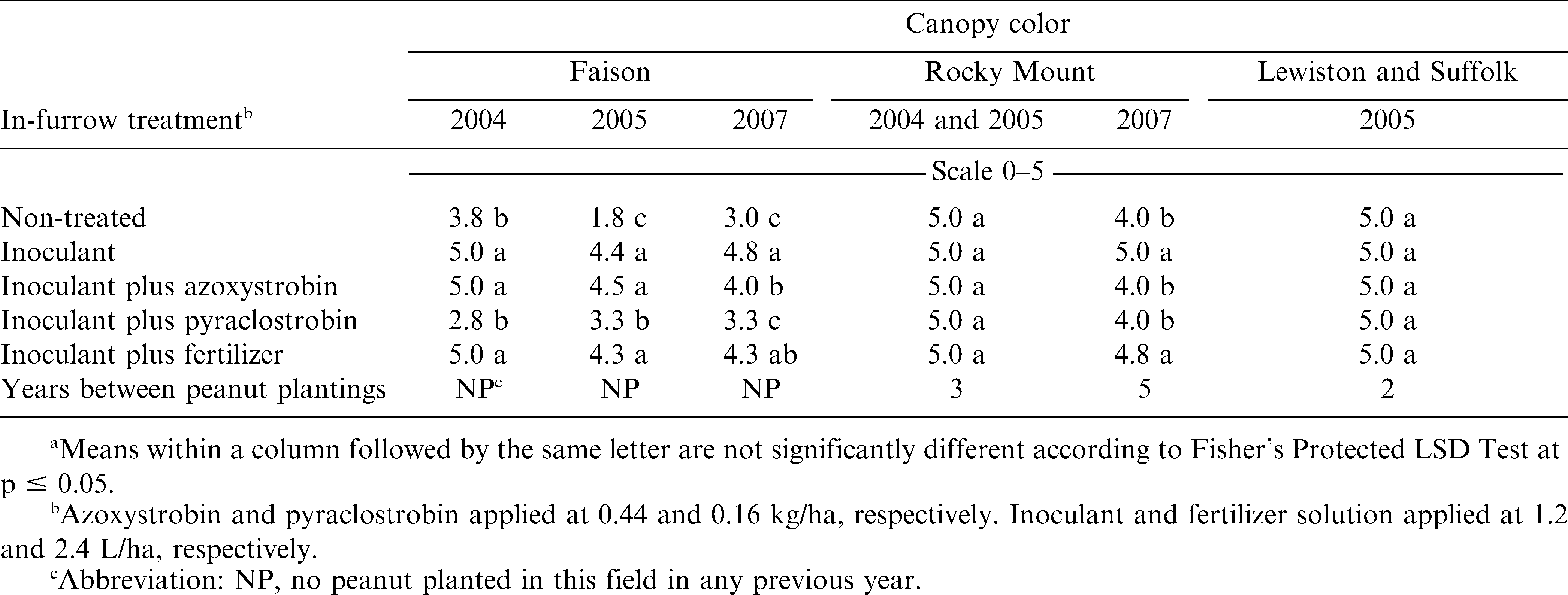
When comparing canopy color, the interaction of experiment by in-furrow treatment was significant at Faison and Rocky Mount. Peanut canopy presented a darker green color in all experiments when inoculant was applied alone or with fertilizer (Table 3). When inoculant alone was applied, peanut canopy color was darker green in two of three experiments at Faison and at Rocky Mount in 2007 compared with inoculant plus azoxystrobin. The peanut canopy also exhibited a lighter green canopy color when inoculant was applied with pyraclostrobin in these experiments compared with inoculant alone (Table 3). Peanut canopy color was not affected by combinations of inoculant and fungicide at Rocky Mount in 2004 or 2005 and at Lewiston and Suffolk. These data suggest that azoxystrobin and pyraclostrobin most likely affect inoculation of peanut by some mechanism as revealed by differences in visual estimates of peanut canopy color. Although the mechanism of this response is not currently defined and the benefits of fungicide in managing disease is not completely known, these data suggest that there is some risk to applying azoxystrobin and pyraclostrobin with Bradyrhizobia inoculant. Additional research is needed to elucidate the mechanism of reduced inoculation. Response to inoculant was not affected by fertilizer solution, although inconsistent results were noted for both pod yield and peanut canopy color (Tables 2 and 3).
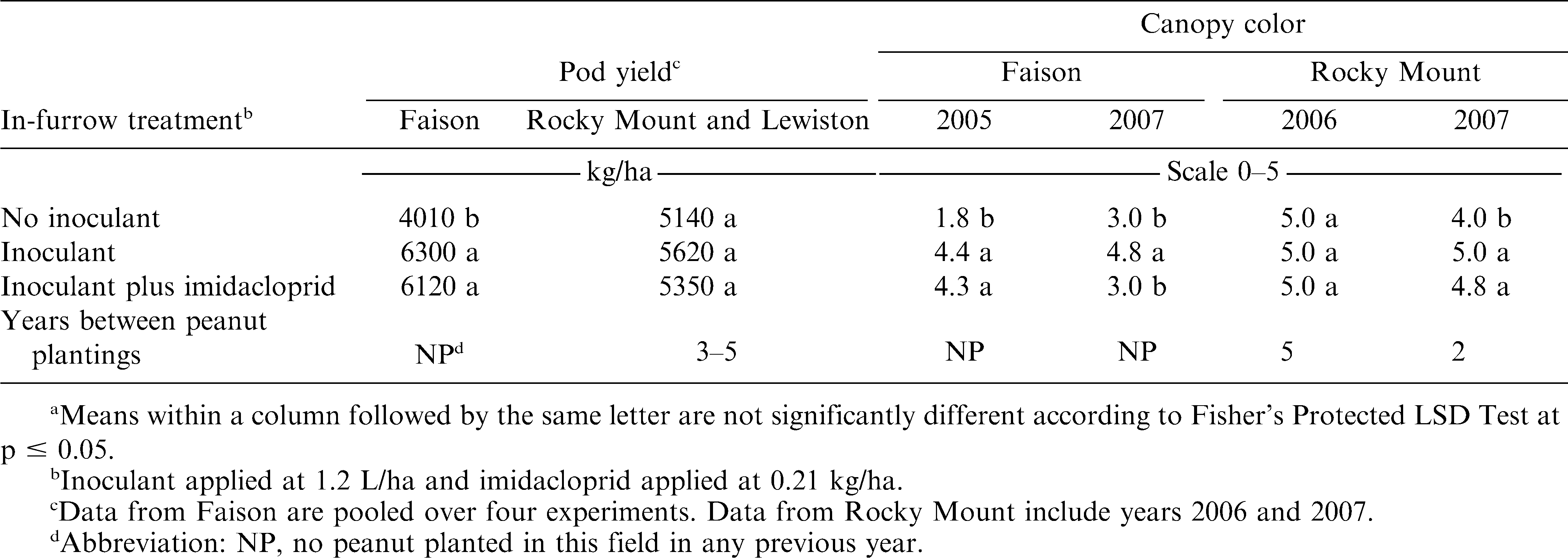
When comparing inoculant alone or with imidacloprid, pod yield in four experiments at Faison, in two experiments at Rocky Mount, and at Lewiston did not differ (Table 4). In 4 of these 7 experiments a difference was noted when comparing the no-inoculant control with inoculant alone (Table 4). In one of four experiments where peanut canopy color was visually estimated, including imidacloprid with inoculant resulted in a lighter green canopy color.
Pod yield did not differ when comparing the inoculated control with inoculant plus tebuconazole at Faison but was lower with tebuconazole when included at Rocky Mount (Table 5). However, unlike results with inoculant plus imidacloprid, pod yield of the no-inoculant control and inoculant plus tebuconazole did not differ. Canopy color did not differ when comparing inoculant alone with inoculant plus tebuconazole (Table 5).
Although the shade of green expressed by the peanut canopy is not the most consistent indicator of inoculum efficacy, it is a possible indicator of BNF. Consistent with effects noted for pod yield, pyraclostrobin appears to pose the greatest risk of decreased BNF and subsequent N deficiency in the peanut canopy. Imidacloprid, while not affecting yield, did affect canopy color indicating that BNF may be compromised when these products are co-applied. Previous research (Todd et al., 1994) suggests that imidacloprid plays a negative role in infection and manifestation of symptoms associated with tomato spotted wilt in peanut. Although the fertilizer solution in this experiment is used by some farmers in peanut, pod yield was lower when fertilizer was co-applied with inoculant compared with inoculant alone in 3 of 11 experiments.
Although data reflecting the degree of nodulation and a more complete set of data on stand establishment and peanut canopy would have been more revealing, results from these experiments do provide an indication of potential interactions of liquid in-furrow Bradyrhizobium inoculant with fungicides, insecticide, and fertilizer solution. Most of these experiments were conducted in fields with little to no expression of soil borne disease or tomato spotted wilt due to site selection or management. The goal of this research was to determine if these materials influence efficacy of Bradyrhizobia inoculant and not necessarily to determine efficacy of fungicides or imidacloprid. Additional research is needed to define interactions of inoculants and other agrichemicals in situations with greater potential for disease.
Acknowledgements
EMD Biosicences (formerly Nitragin Corp.), the National Peanut Board, the North Carolina Peanut Growers Association, Inc., and the Peanut CRSP project LAG-00-96-90013-00 provided partial funding for this research. Carl Murphey, Curtis Fountain, and staff at the Peanut Belt Research Station, the Tidewater Agricultural Research and Extension Center, and the Upper Coastal Plain Research Station provided technical assistance. Appreciation is also extended to Vic Swinson for allowing a portion of this research to be conducted on his farm.
Literature Cited
Brandenburg R. L. 2009 Peanut insect management. 77 – 94 In 2009 Peanut Information North Carolina Coop. Ext. Ser. AG-331 134 .
Culbreath A. K. , Todd J. W. , and Brown S. L. 2003 Epidemiology and management of tomato spotted wilt in peanut. Annu. Rev. Phytopathol 41 : 53 – 75. doi: 10.1146/annurev.phyto.41.052002.095522. Accessed 02-19-2009.
Jordan D. L. 2009a Peanut production practices. 27 – 49 In 2009 Peanut Information North Carolina Cooperative Extension Service Publication AG-331 134 .
Jordan D. L. 2009b Peanut weed management. 50 – 76 In 2009 Peanut Information North Carolina Cooperative Extension Service Publication AG-331 134 .
Jordan D. L. , Brandenburg R. L. , Bailey J. E. , Johnson P. D. , Royals B. M. , and Curtis V. L. 2006 Compatibility of in-furrow application of acephate, inoculant, and tebuconazole in peanut (Arachis hypogaea L.). Peanut Sci 33 : 112 – 117 .
Lanier J. E. , Jordan D. L. , Spears J. F. , Wells R. , and Johnson P. D. 2005 Peanut response to inoculation and nitrogen fertilizer. Agron. J 97 : 79 – 84 .
McPherson R. M. 2006 Incidence of thrips and tomato spotted wilt tospovirus in flue-cured tobacco protected from early season insect pest infestations. J. Econ. Ent 99 3 : 764 – 770 .
Phipps P. M. 2003 The response of CBR-susceptible and -resistant cultivars to preplant treatment with metam and in-furrow application of Folicur and Abound for disease management, 2002. F&N Reports 58 : FC020 .
Shew B. B. 2009 Peanut disease management. 94 – 120 In 2009 Peanut Information, North Carolina Cooperative Extension Service Publication AG-331 134 .
Schiffman J. and Alper Y. 1968 Effects of Rhizobium – inoculum placement on peanut inoculation. Exp. Agric 4 : 203 – 208 .
Shimshi D. , Schiffman J. , Kost Y. , Bielorai H. , and Alper Y. 1967 Effects of soil moisture regime on nodulation of inoculated peanuts. Agron. J 59 : 397 – 400 .
Todd J. W. , Culbreath A. K. , Rogers D. , and Demski J. W. 1994 Contraindications of insecticide use relative to vector control and tomato spotted wilt disease progress in peanut. Proc. Am. Peanut Res. and Ed. Soc 26 : 42 .
Walker M. E. , Minton N. A. , and Dowler C. C. 1976 Effects of herbicides, anematicide and Rhizobium inoculant on yield, chemical composition and nodulation of Starr peanuts (Arachis hypogaea L.). Peanut Sci 3 : 49 – 51 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Wynne J. C. , Coffelt T. A. , Mozingo R. W. , and Anderson W. F. 1991 Registration of “NC-V 11” peanut. Crop Sci 31 : 484 – 485 .
Notes
Author Affiliations
1 Professor and Research Specialist, Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695-7620, William Neal Reynolds Professor, Department of Entomology, North Carolina State University, Box 7613, Raleigh, NC 27695-7613, and former Assistant Professor, Tidewater Agricultural Research and Extension Center, 6421 Holland Rd., Suffolk, VA 23437.
*Corresponding author's e-mail: david_jordan@ncsu.edu.