Introduction
Legume crops such as soybean [Glycine max (L.) Merr.] and peanut (Arachis hypogaea L.) are capable of symbiotic nitrogen fixation with Brady rhizobium (Reid and Cox, 1973; Schiffman and Alper, 1968; Shimshi et al., 1967; Walker et al., 1976). Inoculants can be applied with the seed at planting or as granular or liquid products applied in the seed furrow. Research indicates that in-furrow applications are often more effective than application to the seed when peanut is planted in fields that have never been seeded to peanut (Lanier et al., 2004).
Minimizing damage from tobacco thrips (Frankliniella fusca Hinds) is important in optimizing early season peanut growth and maintaining yield (Brandenburg, 2006). Aldicarb is often applied in-furrow to control tobacco thrips in peanut (Brandenburg, 2006). More recently, phorate has become popular to control tobacco thrips because of greater suppression of tomato spotted wilt of peanut than aldicarb (Brown et al., 2003; Hurt et al., 2003). Acephate can be applied in-furrow or to peanut foliage to control tobacco thrips (Brandenburg, 2006). Growers applying liquid in-furrow inoculants can apply acephate with inoculant. Tebuconazole is routinely applied to control foliar and soil-borne diseases of peanut after the R3 stage of development (Shew, 2006). Additionally, tebuconazole can be applied in the seed furrow to suppress Cylindrocladium black rot (caused by Cylindrocladium parasiticum) (Phipps, 2003).
Growers applying acephate and/or inoculant could apply tebuconazole in the seed furrow to address the need to control tobacco thrips, provide sufficient Brady rhizobia for nitrogen fixation, and suppress seedling disease. However, research addressing interactions of acephate, inoculant, and tebuconazole co-applied in the seed furrow is limited. Therefore, research was conducted to compare tobacco thrips control, peanut emergence and early season growth, and pod yield following in-furrow application of acephate, inoculant, and tebuconazole.
Material and Methods
Experiments were conducted in North Carolina during 2000, 2001, and 2003 in grower fields located near Sunbury (2000 and 2001) and Faison (2003) and at the Peanut Belt Research Station located near Lewiston-Woodville (2001 and 2003). Soil at Sunbury was a Pantego fine sandy loam (fine-loamy, siliceous, thermic Umbric Paleaquults). Soil at Faison was a Wickham sandy loam (fine-loamy, mixed, semiactive, thermic Typic Hapludults). Soil at Lewiston-Woodville was a Norfolk sandy loam (fine-loamy, siliceous, thermic Aquic Paleudults). The cultivar NC-V 11 was planted in conventionally prepared seedbeds in early May to achieve a final in-row plant population of 13 plants/m. Plot size was 2 rows (91-cm spacing) by 9 m. Two other crops, either cotton (Gossypium hirsutum L.) or corn (Zea mays L.), were planted between the current and previous peanut crops at Lewiston-Woodville. Peanut had never been planted in fields at Sunbury or Faison, and the previous crop at both locations was cotton.
Treatments consisted of two rates of acephate (Orthene 97, Valent USA Corp., Walnut Creek, CA) (0 or 1.1 kg ai/ha), two rates of in-furrow inoculant (Lift®, Nitragin Corp., Brookfield, WI) (0 or 1.2 L/ha), and two rates of tebuconazole (Bayer Crop Science, Research Triangle Park, NC) (0 or 220 g ai/ha). Lift® delivers 2200 × 109 viable cells of bacteria/ha when applied at 1.2 L/ha. Treatments were applied in 47 L/ha distilled aqueous solution immediately after seed drop but prior to furrow closure using one 80015 regular flat fan nozzle per row (Spraying Systems Co., Wheaton, IL). Spray solution directly contacted seed. Aldicarb at 1.1 kg ai/ha was inadvertently applied in the seed furrow at Sunbury during 2000. All other production and pest management practices were held constant over the entire test area and were based on Cooperative Extension Service recommendations appropriate for the region.
The experimental design was a randomized complete block with treatments replicated four times. Tobacco thrips damage was recorded 4 wks after planting by evaluating the most recently emerged leaves for signs of tobacco thrips feeding on 25 plants per plot. Leaflets were scored as injured or non-injured based on the presence or absence of tobacco thrips feeding scars. The percentage of leaflets with feeding injury was then calculated and used to determine percent damage. Control of tobacco thrips was calculated by subtracting percent damage from 100%. Peanut stand was determined for the entire length of one of the two plot rows 3 wks after planting. Peanut diameter was determined by measuring the diameter of three plants per plot 4 wks after planting to the nearest cm. Peanut was dug and vines inverted in early October. No attempt was made to determine differences in pod maturity based on pod mesocarp color determination among treatments (Williams and Drexler, 1981). Digging was initiated when approximately 65% of pods were in the brown and black color category and were considered mature based on pods treated with the mixture of acephate and inoculant. Pods were harvested 4 to 7 days after digging and vine inversion. Data for tobacco thrips control, peanut stand and width, and pod yield were subjected to analysis of variance appropriate for the two (acephate rate) by two (inoculant rate) by two (tebuconazole rate) factorial arrangement of treatments. Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Results and Discussion
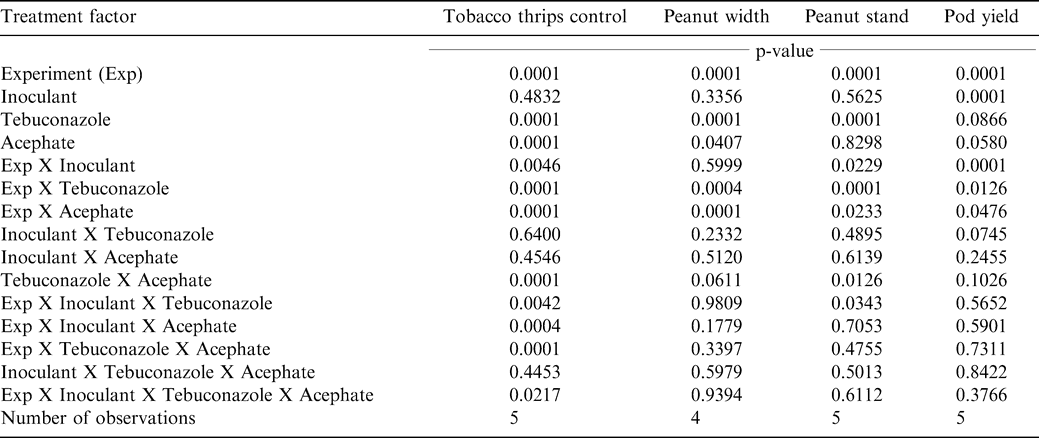
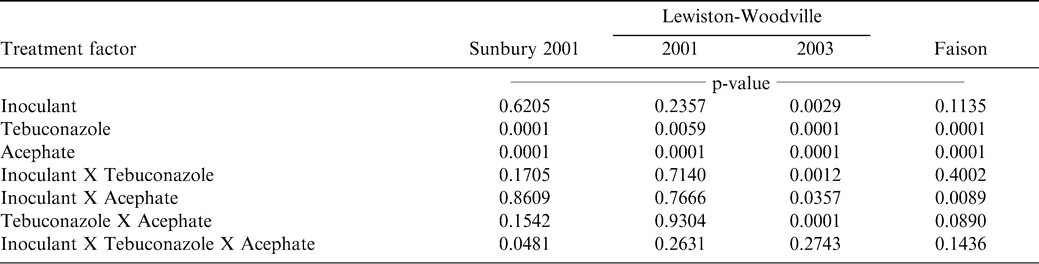
The interaction of experiment by inoculant X tebuconazole X acephate was significant for tobacco thrips control (Table 1). This analysis did not include Sunbury during 2000 because aldicarb was inadvertently applied in the seed furrow and no damage from tobacco thrips was observed. When analyzed by experiment, the interaction of inoculant rate X tebuconazole rate X acephate rate was significant at Sunbury in 2001 but not in the other experiments (Table 2). At Lewiston-Woodville in 2001, main effects of acephate rate and tebuconazole rate were significant while the main effect of inoculant rate and interactions of experiment X treatment factors were not significant. At Lewiston-Woodville in 2003 all two-way interactions were significant. At Faison, the interaction of inoculant rate X acephate rate was significant as were main effects of acephate rate and tebuconazole rate. Other main effects and interactions were not significant in this experiment.
At Sunbury during 2001, control of tobacco thrips was higher when tebuconazole was applied in the absence of acephate regardless of inoculant rate (Table 3). When acephate and inoculant were applied, tobacco thrips control was lower compared to acephate alone but increased when tebuconazole was applied with acephate plus inoculant compared with acephate plus tebuconazole.
Applying acephate increased tobacco thrips control at Lewiston-Woodville during 2001 and 2003 and at Faison (Table 4). Applying acephate increased control from 8% to 87% at Lewiston-Woodville during 2001. However, the interaction of inoculant rate X acephate rate was noted at Lewiston-Woodville during 2003 and at Faison. The highest level of control at Lewiston-Woodville was noted when acephate and inoculant were applied together. However, the opposite response was noted at Faison. The cause of this interaction cannot be explained.
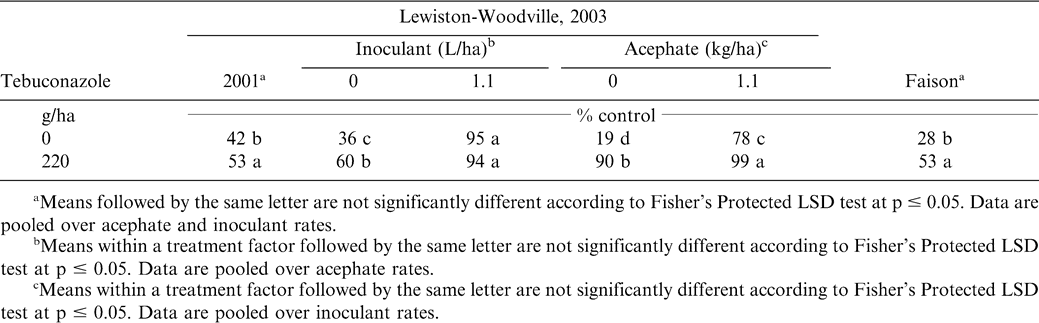
Tobacco thrips control was higher when tebuconazole was applied at Lewiston-Woodville during 2001 and at Faison (Table 5). During 2003 at Lewiston-Woodville, interactions of inoculant rate X tebuconazole rate and tebuconazole rate X acephate rate were noted. Tobacco thrips control was higher when tebucazole was applied without inoculant but not when inoculant and tebuconazole were co-applied. Tebuconazole increased control regardless of acephate rate.
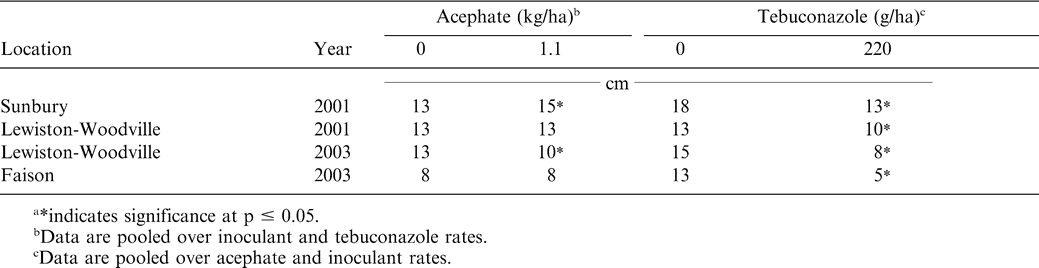
These data demonstrate the effectiveness in controlling tobacco thrips with in-furrow applications of acephate (Brandenburg, 2006). Results indicating that tebuconazole suppresses tobacco thrips are more difficult to explain because tebuconazole has no documented insecticidal properties. However, peanut emerged more slowly when tebuconazole was applied, and this was reflected in differences in peanut stand 3 wks after planting (Table 6) and subsequent peanut width 4 wks after planting (Table 7). Peanut emerging later were visually more compact and may have been less attractive to tobacco thrips. Hurt et al. (2005) reported less damage from tobacco thrips when peanut was planted in late May rather than early May. They postulated that fewer tobacco thrips entered fields during late May and early June than early or mid May. Tebuconazole-treated peanut emerged more slowly than peanut not treated with tebuconazole, and this may explain less damage. Similar findings have not been reported in the literature. Additionally, peanut width was greater when acephate was applied in two of four experiments and most likely resulted from less damage caused by tobacco thrips feeding that occurred earlier in the season (Table 7).
Interactions between inoculant rate and tebucanazole rate were also noted for plant stand (Table 6). Tebucanzole delayed emergence in three experiments. At Faison, applying inoculant and not including tebuconazole resulted in the highest plant population 3 wks after planting. Peanut stand was recorded only 3 wks after planting. Although qualitative observations indicated that peanut treated with tebuconazole eventually emerged, plant stand was not assessed later in the season. Lack of these data limit conclusions concerning overall peanut emergence differing between tebuconazole-treated peanut and non-treated peanut. Phipps (2003) reported lower stand counts early in the season when tebuconazole was applied in the seed furrow compared with non-treated peanut.
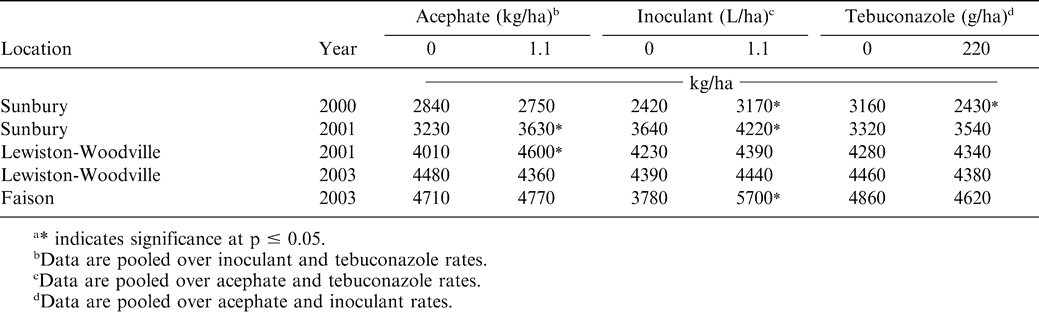
Peanut yield was affected by acephate, inoculant, and tebuconazole independently (Tables 1 and 8). Interactions of experiment with these treatment factors was significant. When pooled over incoculant and tebuconazole treatments, pod yield was lower when acephate was applied at Sunbury and Lewiston-Woodville during 2001. Acephate increased tobacco thrips control regardless of inoculant or tebucanazole rate in these experiments (Tables 3 and 4). At Lewiston-Woodville during 2003 and at Faison, yield was not affected by acephate. This was somewhat suprising because acephate increased tobacco thrips control at these locations (Table 4). However, increased tobacco thrips control does not always translate into increased yield (Hurt et al., 2005).
Peanut pod yield increases were noted when inoculant was applied at Sunbury during both years and at Faison regardless of acephate or tebuconazole treatment (Table 8). However, inoculant did not increase yield at Lewiston-Woodville during either year. These results were not surprising because peanut had never been planted in fields at Sunbury and Faison, and response to inoculant is often greatest under this scenario (Lanier et al., 2004). In contrast, peanut had been planted at Lewiston-Woodville in the fields for many years in a rotation with either two crops of cotton or corn separating peanut crops.
Tebuconazole reduced yield in only one of five experiments even though peanut emergence was delayed in most experiments and peanut diameter was less when tebuconazole was applied. Although interactions were noted among acephate, inoculant, and tebuconazole treatment factors with respect to tobacco thrips control, in several experiments tebuconazole-treated peanut had less damage from tobacco thrips. Partitioning the relative benefits of less thrips damage versus risks of slower emergence when tebuconazole was applied is difficult using these data. Tebuconazole controls soil-borne pathogens, and although very little disease was noted in these experiments, tebuconazole could have contributed to disease management that was not visually apparent and offset risks associated with delayed emergence.
Although a number of interactions were noted among treatment factors for tobacco thrips control and peanut emergence and peanut diameter, lack of interactions for pod yield indicated that yield response to acephate, inoculant, and tebuconazole is independent. These data indicate that acephate, inoculant, and tebuconazole are compatible with respect to peanut yield. These data also emphasize the importance of controlling tobacco thrips, and the value of in-furrow inoculation with Brady rhizobia in fields where peanut has not been planted in previous years. Additionally, applying tebuconazole reduced yield in one experiment and did not positively affect yield in the remaining four experiments when used in a situation with little disease occurrence.
Acknowledgements
This research was supported financially by the North Carolina Peanut Growers Association, Inc. and USAID Peanut CRSP (LAG-G-00-96-90013-00) Carl Murphy, Brenda Penny, Paul Smith, Bryant Spivey, and staff at the Peanut Belt Research Station provided technical support. Appreciation is expressed to Umphlett Brothers and Swinson Farms for allowing experiments to be conducted on their farms.
Literature Cited
Brandenburg R. L. 2006 Peanut insect management. 61 – 78 in 2006 Peanut Information North Carolina Coop. Ext. Ser. AG-331 116 .
Brown S. , Tood J. , Culbreath A. , Baldwin J. , Beasley J. , Kemerait B. , and Prostko E. 2003 Minimizing spotted wilt of peanut. Univ. of Georgia, Coop. Ext. Serv. Bull 1165 .
Hurt C. , Brandenburg R. , Jordan D. , Shew B. , Isleib T. , Linker M. , Herbert A. , Phipps P. , Swann C. , and Mozingo W. 2003 Managing tomato spotted wilt virus in peanuts in North Carolina and Virginia. North Carolina Coop. Ext. Serv. Publ. AG-638
Hurt C. A. , Brandenburg R. L. , Jordan D. L. , Kennedy G. G. , and Bailey J. E. 2005 Management of tomato spotted wilt virus vector Frankliniella fusca (Thysanoptera:Thripidae) in virginia market type peanut. J. Econ. Entomol 98 : 1435 – 1440 .
Jordan D. , Johnson D. , Spears J. , Penny B. , Shew B. , Brandenburg R. , Faircloth J. , Phipps P. , Herbert A. , Coker D. , and Chapin J. 2005 Determining peanut pod maturity and estimating the optimal digging date: using pod mesocarp color for digging Virginia market type peanut. North Carolina Cooperative Extension Service Publication AG-633
Lanier J. E. , Jordan D. L. , Spears J. F. , Wells R. , and Johnson P. D. 2005 Peanut response to inoculation and nitrogen fertilizer. Agron. J 97 : 79 – 84 .
Phipps P. M. 2003 The response of CBR-susceptible and –resistant cultivars to preplant treatment with metam and in-furrow application of Folicur and Abound for disease management, 2002. F&N Reports 58 : FC020 .
Reid P. H. and Cox F. R. 1973 Soil properties, mineral nutrition and fertilizer practices. in Wilson C. T. ed. Peanuts - culture and uses. A symposium Amer. Peanut Res. and Educ. Assoc., Inc Stillwater, Oklahoma .
Schiffman J. and Alper Y. 1968 Effects of Rhizobium – inoculum placement on peanut inoculation. Exp. Agric 4 : 203 – 208 .
Shew B. B. 2006 Peanut disease management. 79 – 104 in 2006 Peanut Information North Carolina Coop. Ext. Ser. AG-331 116 .
Shimshi D. , Schiffman J. , Kost Y. , Bielorai H. , and Alper Y. 1967 Effects of soil moisture regime on nodulation of inoculated peanuts. Agron. J 59 : 397 – 400 .
Walker M. E. , Minton N. A. , and Dowler C. C. 1976 Effects of herbicides, a nematicide and Rhizobium inoculant on yield, chemical composition and nodulation of Starr peanuts (Arachis hypogaea L.). Peanut Sci 3 : 49 – 51 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Notes
- Assoc. Prof., Prof., Dept. Crop Science, North Carolina State Univ., Box 7620, Raleigh, NC 27695-7620; Prof., Dept. Entomology, Box 7613, Raleigh, NC 27695-7613; Prof., Dept. Plant Pathology, Box 7616, North Carolina State Univ., Raleigh, NC 27695-7616; Technician, Dept. Crop Science, North Carolina State Univ., Raleigh, NC; Technician, Dept. Entomology, Raleigh, NC; and Technician, Dept. Plant Pathology, North Carolina State Univ. Raleigh, NC. [^] *Corresponding author's e-mail address: david_jordan@ncsu.edu.
Author Affiliations