Introduction
Arachis L., the genus that includes the cultivated peanut, has about 69 species occurring in South America east of the Andes, south of the Amazon, north of La Plata, and from northwest Argentina to northeast Brazil. The species have been assigned to nine sections on the basis of cross-incompatability and exo-morphologic character clustering (Krapovickas and Gregory, 1994). Section Rhizomatosae contains one diploid species (A. burkartii Handro) and two tetraploid species [A. glabrata Benth. and A. pseudovillosa (Chodat & Hassl.) Krapov. & W.C. Gregory]. The largest section, Arachis, contains many diploid species and two tetraploids (A. monticola Krapov. & Rigoni and A. hypogaea L., the cultivated peanut) (Fernandez and Krapovickas, 1994; Krapovickas and Gregory, 1994). Evidence from meiotic chromosome associations (Seetharam et al., 1973; Stalker, 1985), karyotypes (Stalker and Dalmacio, 1986), isozymes (Lu and Pickersgill, 1993; Stalker et al., 1994), and DNA restriction fragment length polymorphisms (RFLP) (Halward et al., 1991, 1992; Kochert et al., 1991, 1996) indicate that the cultivated peanut is an allotetraploid. Evidence from nuclear cDNA and chloroplast DNA RFLP data (Kochert et al., 1996) as well as sequence data (Jung et al., 2003) support A. duranensis Krapov. & W.C. Gregory and A. ipaensis Krapov. & W.C. Gregory as the species most similar to progenitors that hybridized producing A. hypogaea. RFLP data (Kochert et al., 1996) suggests that A. monticola is likely the existing taxon that most resembles the original allotetraploid that resulted from hybridization between the aforementioned species. However, sequence data used to generate phylogenetic trees (Jung et al., 2003) and cytogenetic evidence (Stalker and Dalmacio, 1986) suggest that A. monticola is not a progenitor of the cultivated peanut. While a great deal has been elucidated in regard to the domesticated peanut and its wild relatives, little is known about the origins of the other tetraploids in the genus, other than that they likely evolved independently of the domesticated peanut and its wild relatives (Smartt and Stalker, 1982; Halward et al., 1991, 1992).
RAPD methods have previously been used in genetic studies of peanuts and were used to examine several wild tetraploid species in the genus. Halward et al. (1992) examined two peanut cultivars; 25 unadapted germplasm lines of A. hypogaea, the putative, allotetraploid progenitor of the cultivated peanut (A. monticola); a tetraploid from section Rhizomatosae (A. glabrata); and 29 diploid species using 10 random primers. No variation was found in A. hypogaea cultivars or germplasm, but variation was identified among the Arachis species. They concluded that the tetraploids in section Arachis and Rhizomatosae were the result of polyploidy evolving twice within the genus (Halward et al., 1992). Intraspecific variation was lowest in A. hypogaea and A. monticola and the two species clustered together in dendograms based on RAPD data (Hilu and Stalker, 1995). Arachis glabrata was genetically distinct when compared to the A. hypogaea-A.monticola complex (Halward et al., 1992). In addition, a diploid Rhizomatosae species (A. burkartii) was shown to be genetically different from tetraploid taxa of the section (Gimenes et al., 2002).
Because hypotheses regarding the origin of the domesticated peanut have been established (Kochert et al., 1996) and taxonomic boundaries have been defined for sections and species within the genus (Krapovickas and Gregory, 1994), RAPD markers would be useful in examining these hypotheses as well as other tetraploids in the group in regard to their genetic diversity and sectional affinities. Specifically, the objectives of this investigation were to examine the two sections that contain tetraploid species (Arachis and Rhizomatosae), assess the levels of genetic diversity in A. hypogaea, A. monticola, and A. glabrata, as well as evaluate the position of A. pseudovillosa within the genus.
Materials and Methods
Plant Materials
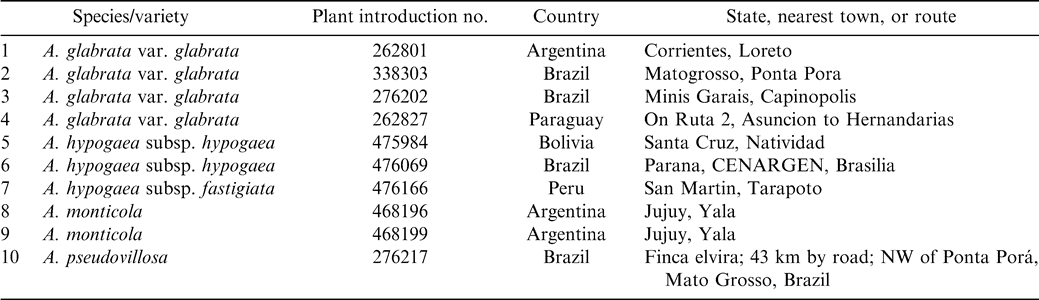
Seeds from 10 collections (Table 1) were obtained from Dr. Charles Simpson at the Texas Agric. Exp. Sta., Stephenville, TX. Plants were grown in the Dept. of Biological Sciences Greenhouse at Tarleton State Univ., Stephenville,TX. Young leaf material from these plants was used for DNA isolation.
Dna Isolation
Total genomic DNA used as templates was isolated using the DNeasy Plant Maxi Kit (Qiagen, Valencia, CA). Nucleic acid purity and approximate concentration was determined using an ultraviolet spectrophotometer.
Rapd Protocols
The RAPD procedure followed that of Williams et al. (1990) with some modifications (Hilu and Stalker, 1995). Ten samples of tetraploid peanut species (Table 1) were run with 40 10-base oligonucleotide primers (kits A and B from Operon Technologies, Inc., Almeda, CA). Only clear, well-separated amplification products were scored, and each of these was tested several times for repeatability. Of the 40 10-base oligonucleotide primers initially evaluated, 16 were polymorphic and utilized for analyses (primers OPA 1, 3, 4, 7, 8–11, 13, 14, 18–20 and OPB 10, 12, 17).
Each reaction mixture for PCR amplification totaled 25 µL and contained 2.5 µL of 20× reaction buffer, 2 mM MgCl2; 1 mM each of dATP, dCTP, dGTP, and dTTP; 5 to 6 pM of single PCR primer; 0.9 unit of Taq polymerase (Qiagen); and approximately 25 ng of genomic DNA template. All reaction mixtures were prepared as master mixes for each primer to minimize measurement errors. Amplification was carried out in a Techne Genius thermal cycler using one initial denaturation cycle at 95 C for 5 min followed by 75 cycles at 95 C for 10 sec, 36 C for 10 sec, and 72 C for 2 min. On completion of the cycles, samples were refrigerated at 4 C before electrophoresis. Amplification products were separated by electrophoresis in a 1.5% agarose gel. About 20 µL of sample was loaded in the gel and electrophoresed in Tris-acetate-EDTA (TAE) buffer at 100 millivolts for 4 to 5 hr. A 1 kb DNA marker (Fisher Scientific) was used as a standard. DNA in gels was stained with ethidium bromide and photographed under ultraviolet light.
Data Analysis
Polymorphic bands were scored as either present or absent and analyzed using Numerical Taxonomy and Systematics Personal Computer (NTSYS-pc) software (Rohlf, 1990) using the unweighted pair group method using arithmetic averages (UPGMA) analysis based on Dice's similarity between populations, given by 2a/(2a + b), where a is the number of positive matches (shared bands) and b is the number of mismatches. Similarity values obtained from the Dice algorithm were utilized to group accessions via the UPGMA method to produce a single unrooted phenogram. The cophenetic coefficients for the clusters and the correlation between these coefficients and the similarity matrix were computed (Sneath and Sokal, 1973). The Dice coefficient was used to calculate similarities on the basis of shared presence of a DNA band and excluded shared absence of fragments from the analysis (Sneath and Sokal, 1973).
Results
A total of 16 reliable RAPD primers were scored from the 40 primers that were used in the investigation, and 156 loci were identified for a mean of 9.75 loci per primer. Ninety-two percent of the loci were polymorphic. Forty-three RAPD markers were observed exclusively in A. glabrata, none in A. hypogaea, one in A. monticola, and 15 in A. pseudovillosa.
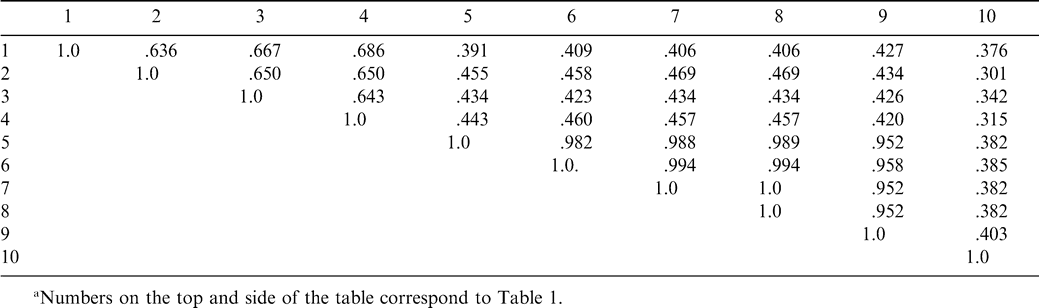
The five accessions of A. hypogaea and A. monticola had genetic similarity values that ranged from 0.952 to 1.000 (Table 2), whereas four accessions of A. glabrata had values that ranged from 0.683 to 0.686 (Table 2). Arachis pseudovillosa had a genetic similarity value that was far from either of the aforementioned species with a similarity value of 0.382 to 0.403 to the A. hypogaea and A. monticola group and 0.301 to 0.376 to the A. glabrata group (Table 2).
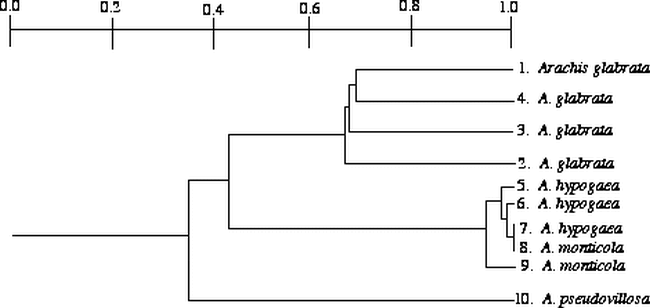
Clusters formed from UPGMA analysis indicate that accessions of A. glabrata are more genetically diverse than accession in the A. hypogaea-A. monticola complex (Fig. 1). Within this complex, A. hypogaea subsp. fastigiata (population 7) is identical to an accession of A. monticola (accession 8 in Fig. 1). Arachis pseudovillosa is distinct from the other groups, but clusters slightly closer to the A. hypogaea-A. monticola complex (Fig. 1).
Discussion
The tetraploids in section Arachis cluster together and are genetically very similar (Table 1; Fig. 1). This is concordant with previous investigations using isozymes (Lu and Pickersgill, 1993; Stalker et al., 1994), RAPD data (Hilu and Stalker, 1995), and RFLP data (Halward et al., 1991, 1992; Kochert et al., 1991). The two species included in this investigation from the section Rhizomatosae do not cluster together (Fig. 1) and are genetically diverse (Table 1). The section Rhizomatosae has been described as problematic and needs further investigation (Smartt and Stalker, 1982). Our data supports the hypothesis that the tetraploid species in section Rhizomatosae arose from a polyploidization event that was independent of that which formed the A. hypogaea-A. monticola complex (Smartt and Stalker, 1982; Halward et al., 1991). Because A. glabrata and A. pseudovillosa are genetically divergent, this investigation also provides support for multiple origins of the tetraploids A. pseudovillosa and A. glabrata in the section Rhizomatosae. Perenniality and asexual reproduction by rhizomes could allow for more recovered examples of chromosome doubling (Grant, 1971) in different geographic regions and involving more progenitor populations. Multiple origins of taxa in the section Rhizomatosae, as opposed to the probable single origin of tetraploids in the section Arachis (Kochert et al., 1996), could account for the differences in genetic diversity observed in tetraploids within the two sections. The diversity of four populations of A. glabrata indicates that this tetraploid species may have evolved several times at different locations, or more likely, because of its very broad distribution in diverse habitats, selection resulted in genetic diversity within the species.
Our limited RAPD data supports the hypothesis that A. monticola and A. hypogaea are genetically similar, with A. monticola possibly being the direct, ancestral species of the cultivated peanut (Hilu and Stalker, 1995). This hypothesis has also been supported by isozyme (Lu and Pickersgill, 1993; Stalker et al., 1994), RAPD (Hilu and Stalker, 1995), and RFLP data (Halward et al., 1991, 1992; Kochert et al., 1991). Genetic analysis of the RAPD data in this study indicates that A. monticola is genetically more similar to A. hypogaea subsp. fastigiata than A. hypogaea subsp. hypogaea. Previous researchers using isozymes (Lu and Pickersgill, 1993; Stalker et al., 1994) supported this pattern, but RFLP variation indicated that A. monticola is more closely related to A. hypogaea subsp. hypogaea (Paik-Ro et al., 1992). Even though the relationship of A. monticola to a particular subspecies of A. hypogaea remains unresolved, our limited data and that from isozyme (Lu and Pickersgill, 1993; Stalker et al., 1994), RAPD (Hilu and Stalker, 1995), and RFLP data (Halward et al., 1991, 1992; Kochert et al., 1991) indicate that there is very little interspecific genetic diversity within the A. hypogaea-A. monticola complex or intraspecific genetic diversity in A. hypogaea. However, He and Prakash (1997) detected low levels of DNA polymorphism using DNA amplification fingerprinting and amplified fragment length polymorphism approaches; but they also concluded that the level of genetic variation in cultivated peanut is still low compared to most other cultivated crops.
Acknowledgements
We wish to thank the V. H. McNutt Memorial Foundation and Organized Faculty Research at Tarleton State Univ. for providing funding for the project and Dr. Charles Simpson at the Texas AM Univ. Agric. Ext. Res. Sta. for providing plant materials for DNA extraction.
Literature Cited
Fernandez A. and Krapovickas A. 1994 Cromosomas y evolución en Arachis (Leguminosae). Bonplandia 8 : 187 – 220 .
Gimenes M. A. , Lopes C. R. , and Valls J. F. M. 2002 Genetic relationships among Arachis species based on AFLP. Genet. and Mol. Biol 25 : 349 – 353 .
Grant V. 1971 Plant Speciation Columbia Univ. Press New York .
Halward T. M. , Stalker H. T. , LaRue E. A. , and Kochert G. 1991 Genetic variation detectable with molecular markers among unadapted germ-plasm resources of cultivated peanut and related wild species. Genome 34 : 1013 – 1020 .
Halward T. , Stalker T. , LaRue E. , and Kochert G. 1992 Use of single-primer DNA amplifications in genetic studies of peanut (Arachis hypogaea L.). Plant Mol. Biol 18 : 315 – 325 .
He G. and Prakash C. S. 1997 Identification of polymorphic DNA markers in cultivated peanut (Arachis hypogaea L.). Euphytica 97 : 143 – 149 .
Hilu K. W. and Stalker H. T. 1995 Genetic relationships between peanut and wild species of Arachis sect. Arachis (Fabaceae): Evidence from RAPDs. Plant Syst. Evol 198 : 167 – 178 .
Jung S. , Tate P. L. , Horn R. , Kochert G. , Moore K. , and Abbott A. G. 2003 The phylogenetic relationship of possible progenitors of the cultivated peanut. J. Hered 94 : 334 – 340 .
Kochert G. , Halward T. , Branch W. D. , and Simpson C. E. 1991 RFLP variability in peanut (Arachis hypogaea L.) cultivars and wild species. Theor. Appl. Genet 81 : 565 – 570 .
Kochert G. , Stalker H. T. , Gimenes M. , Galgaro L. , Lopes C. R. , and Moore K. 1996 RFLP and cytogenetic evidence on the origin and evolution of allotetraploid domesticated peanut, Arachis hypogaea (Leguminosae). Amer. J. Bot 83 : 1282 – 1291 .
Krapovickas A. and Gregory W. C. 1994 Taxonomia del genero Arachis (Leguminosae). Bonplandia 8 : 1 – 186 .
Lu J. and Pickersgill B. 1993 Isozyme variation and species relationships in peanut and its wild relatives (Arachis L. Leguminosae). Theor. Appl. Genet 85 : 550 – 560 .
Paik-Ro O. G. , Smith R. L. , and Knauft D. A. 1992 Restriction fragment length polymorphism evaluation of six peanut species within the Arachis section. Theor. Appl. Genet 84 : 201 – 208 .
Rohlf F. J. 1990 Numerical Taxonomy and Multivariate Analysis System, Ver. 1.80 Exeter Software New York .
Seetharam A. , Nayar K. M. D. , Sreekantaradhya R. , and Achar D. K. T. 1973 Cytological studies on the interspecific hybrid of Arachis hypogaea × Arachis duranensis. Cytologia 38 : 277 – 280 .
Simpson C. E. and Higgins D. L. 1984 Catalog of Arachis germplasm collections in South America, 1976–1983 Texas Agric. Exp. Sta., The Texas AM Univ. System College Station, TX .
Smartt J. and Stalker H. T. 1982 Speciation and cytogenetics in Arachis. In Pattee H. E. and Young C. T. eds. Peanut Science and Technology Amer. Peanut Res. Educ. Soc Yoakum, TX 21- 49.
Sneath P. H. and Sokal R. R. 1973 Numerical Taxonomy W. H. Freeman San Francisco .
Stalker H. T. 1985 Cytotaxonomy of Arachis. In Moss J. P. ed. Proc. Intl. Workshop on the Cytogenetics of Arachis, Hyderabad, India. 31 Oct.–2 Nov ICRISAT Patancheru, India 65 – 79 .
Stalker H. T. and Dalmacio R. D. 1986 Karyotype analysis and relationships among varieties of Arachis hypogaea. Caryologia 51 : 617 – 629 .
Stalker H. T. , Phillips T. D. , Murphy J. P. , and Jones T. M. 1994 Variation of isozyme patterns among Arachis species. Theor. Appl. Genet 87 : 746 – 755 .
Williams J. G. K. , Kubelik A. R. , Livak K. J. , Rafaliski J. A. , and Tingey S. V. 1990 DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl. Acids Res 18 : 6531 – 6535 .
Notes
- 1 Assoc. Prof. of Biological Sciences, Grad. Stud., Grad. Stud., Grad. Stud., and Grad. Stud., respectively, Dept. of Biological Sci., Tarleton State Univ., Box T-0100, Stephenville, TX 76402 [^] *Corresponding author: (email: nelson@tarleton.edu).
Author Affiliations