Introduction
Foliar and soilborne diseases can severely affect yield and quality of peanut. Backman and Crawford (1984) found that yield losses in Alabama from early leaf spot ranged from 10 to 15% but exceeded 50% in growing seasons where conditions were conducive to disease development. Early leaf spot (Cercospora arachidicola Hori.) has caused 50 to 70% pod yield loss in the absence of fungicides (Shokes and Culbreath, 1997). In Georgia and Florida, yield reductions exceeded 1100 kg/ha when early leaf spot was not controlled (Jacobi and Backman, 1991). Early leaf spot can attack healthy peanut plants and reduces tissue development (Nutter and Shokes, 1995). Grichar et al. (2000) reported that early leaf spot is more prevalent in south Texas due to high humidities and longevity of leaf moisture. Under ideal environmental conditions, disease symptoms occur within 10 to 14 d after inoculation and develop throughout the growing season. The pathogen thrives when temperatures are between 24 and 28 C and relative humidity exceeds 90% for 2 d or more (Smith and Littrell, 1980). Early leaf spot and late leaf spot caused by Cercosporidium personatum (Berk. and M. A. Curtis) Deighton have potential to develop in south Texas where these environmental conditions prevail.
Control of early leaf spot includes the use of fungicides and cultural practices. Until recently, south Texas growers relied on the use of chlorathalonil for control of early leaf spot. Chlorothalonil does not control soilborne pathogens, resulting in the need for additional fungicides to control southern stem rot (caused by Sclerotium rolfsii Sacc.). Most growers in south Texas initiate fungicide applications 45 to 60 d after planting (DAP) to control both foliar and soilborne diseases and apply no more than two to five applications throughout the growing season, depending on temperature and relative humidity. Therefore, only one to two applications of a chlorothalonil product may be used. A 2- to 3-yr crop rotation without peanut has proven to suppress leaf spot (Nutter and Shokes, 1995).
Much like early leaf spot, rust (caused by Puccinia arachidis Speg.) can be devastating to peanut production. Subrahmanyan et al. (1989) reported pod yield reductions as high as 50 to 60% under severe rust pressure. Rust epidemics develop faster than leaf spot and may cause pod maturation 2 to 3 wk early, resulting in smaller seed and pod loss at digging (Nutter and Shokes, 1995). In south Texas, rust is prevalent due to tropical spore deposition (Van Arsdel and Harrison, 1972). Control of rust can be achieved through the use of chlorothalonil, although a spray schedule of every 7 to 10 d may be needed.
Southern stem rot is a soilborne disease that south Texas growers have to manage every growing season. Southern stem rot is greatly influenced by moisture (Boyle, 1961). Therefore, in irrigated fields with short rotations between peanut crops, southern stem rot incidence can increase significantly within a season. Yield reductions of up to 10% have been reported as a result of southern stem rot in the Southeast and up to 5% in Southwest peanut regions (Melouk and Backman, 1995). Smith and Lee (1986) reported that southern stem rot damage costs growers in Texas and Oklahoma approximately $15 million annually. Because this disease can significantly reduce peanut yields, growers include fungicide and cultural control measures in their management practice.
The development of peanut fungicides over the last 5 to 10 yr has given growers additional options in controlling early leaf spot, rust, and southern stem rot. In Georgia, tebuconazole controlled southern stem rot when applied multiple times as a foliar spray (Brenneman et al., 1991). Hagan et al. (1991) concluded that tebuconazole reduced southern stem rot and enhanced yields as compared to the untreated check. Besler et al. (2001) found that tebuconazole reduced southern stem rot on several peanut cultivars when applied as few as two times. Also, when applied on 14-, 21-, and 28-d schedules, tebuconazole reduced leaf spot severity and increased yield compared with an untreated check. Jaks et al. (1998) reported significant rust control with tebuconazole when used in an advisory schedule program.
Azoxystrobin controls leaf spot and southern stem rot similar to tebuconazole (Grichar et al., 2000). Lunsford et al. (1998) found that azoxystrobin reduced early leaf spot defoliation when applied at the rate of 0.17 kg/ha and was comparable to chlorothalonil.
Little research documents the effects of a twin-row planting pattern on the development of foliar and soilborne diseases in conjunction with fungicides. Growers in south Texas have been slow to adopt twin-rows because of concerns that a quicker canopy closure may increase the risk of damage by soil and foliar diseases from favorable microclimate conditions. Harrison (1970) found no apparent differences in soilborne disease development with a twin- and narrow-row pattern when compared to the single row pattern. Sconyers et al. (2002) concluded that southern stem rot disease incidence was greater in 10-cm twin-rows than a wider single row pattern. They also found that southern stem rot disease incidence was higher along linear rows in a 10-cm twin-row pattern than in a 30-cm twin-row or single row pattern.
The objective of this study was to evaluate the response of four runner-type cultivars to disease development (southern stem rot, early leaf spot, and rust), pod yield, and quality when sprayed with azoxystrobin and tebuconazole and planted in single and twin-row planting patterns. To exclusively determine season-long disease control with these two fungicides, chlorothalonil was not applied to test areas. However, alternate use of fungicides with different modes of action should be used commercially to minimize risk of disease tolerance to azoxystrobin or tebuconazole.
Material and Methods
Field studies were conducted in Texas during 2001 near Pearsall and 2002 at the Texas Agric. Exp. Sta. near Yoakum. Soil at the Pearsall location was a Duval very fine sand (fine-loamy, mixed, hyperthermic Aridic Haplustalfs) with less than 1% organic matter. Soil at Yoakum was a Straber loamy sand (fine mixed thermic Aquic Palenstalfs) with less than 1% organic matter. The test site in Pearsall was in a 2-yr crop rotation with corn and grain sorghum while the Yoakum site was in continuous peanut for more than 5 yr. Both sites had a known history of leaf spot and southern stem rot disease incidence. The runner-type cultivars AT 1-1, Flavor Runner 458, Georgia Green, and Tamrun 96 were planted on 30 May 2001 and 3 Jun 2002. Seed of each cultivar was planted with a Monosem precision vacuum planter (Monosem ATI, Inc., Lenoxa, KS). Number of seeds per meter on a bed were held constant for both the single and twin-row patterns to achieve approximately 87 and 120,000 plants/ha, respectively. The experimental design was a split-split plot arrangement with three replications. Whole plots consisted of cultivars, fungicide treatments represented the split-plot, and row patterns represented split-split plots. Plot size was two rows × 7.6 m long.
Azoxystrobin (Syngenta, Greensboro, NC) at 0.34 kg/ha was applied 62 and 90 DAP in 2001 and 59 and 87 DAP in 2002. Tebuconazole (Bayer Crop Sciences, Research Triangle Park, NC) at 0.23 kg/ha was applied 62, 76, 90, and 105 DAP in 2001 and 59, 73, 87, and 102 DAP in 2002. Fungicides were applied with a CO2-pressurized backpack sprayer with D3-13 hollowcone nozzles delivering 187 L/ha. Rust only developed in 2001 at Pearsall and was evaluated using the Intl. Crops Res. Inst. for the Semi-Arid Tropics (ICRISAT) scale of 1 to 9 (where 1 = no disease and 9 = plants severely affected) and 50 to 100% of the leaves are withering (Subrahmanyan et al., 1982). One rating was taken late season on 9 Oct. Early leaf spot occurred only in 2002 at Yoakum and was evaluated using the Florida scale of 1 to10 (where 1 = no leaf spot and 10 = plants completely defoliated and killed by leaf spot) was used to assess leaf spot severity (Chiteka et al., 1988). Early leaf spot was rated on 9 Aug., 1 Oct., and 15 Oct. However, only the final rating is reported. Southern stem rot developed both years at each location. Southern stem rot was assessed immediately after inversion on 19 Oct. in 2001. In 2002, due to high early leaf spot severity, southern stem rot was assessed above ground on Oct. 1. A disease locus was defined as ≤ 30 cm of consecutive southern stem rot damage of plants in a plot bed (Rodriquez-Kabana et al., 1975). Identification of southern stem rot was determined by dead or wilted branches with visible mycelial growth and the presence of sclerotia.
Peanuts were dug on 19 Oct. 2001 and 15 Oct. 2002, allowed to field dry 5 to 7 d, and then harvested using a power-take-off driven combine. Plot weights were taken after all foreign matter was removed. Analysis of variance (ANOVA) was performed on southern stem rot disease incidence and peanut yield to test the effect of year, row pattern, fungicide treatments and all possible interactions. All main effects and possible interactions were analyzed for rust and early leaf spot for the year when these diseases occurred. Mean separation was then calculated using Fisher's Protected LSD test (P ≤ 0.05).
Results and Discussion
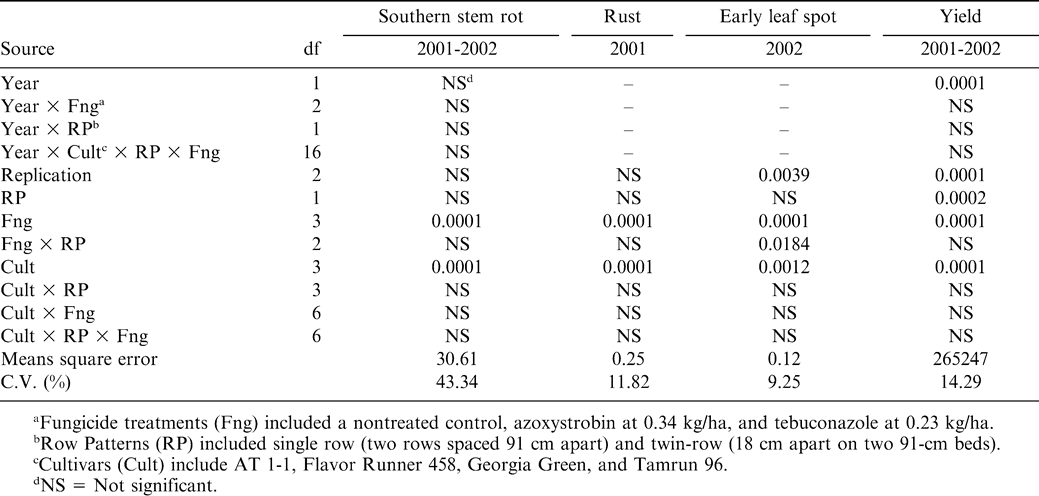
Data for southern stem rot and yield were combined over years due to lack of year × treatment interaction (Table 1). A significant cultivar and fungicide treatment effect occurred for southern stem rot, while a significant cultivar, fungicide treatment, and row pattern effect occurred for yield. In 2001, a significant fungicide treatment and cultivar effect resulted for rust (Table 1). Significant fungicide treatment, cultivar, and fungicide treatment × row pattern effects occurred for early leaf spot.
Southern Stem Rot
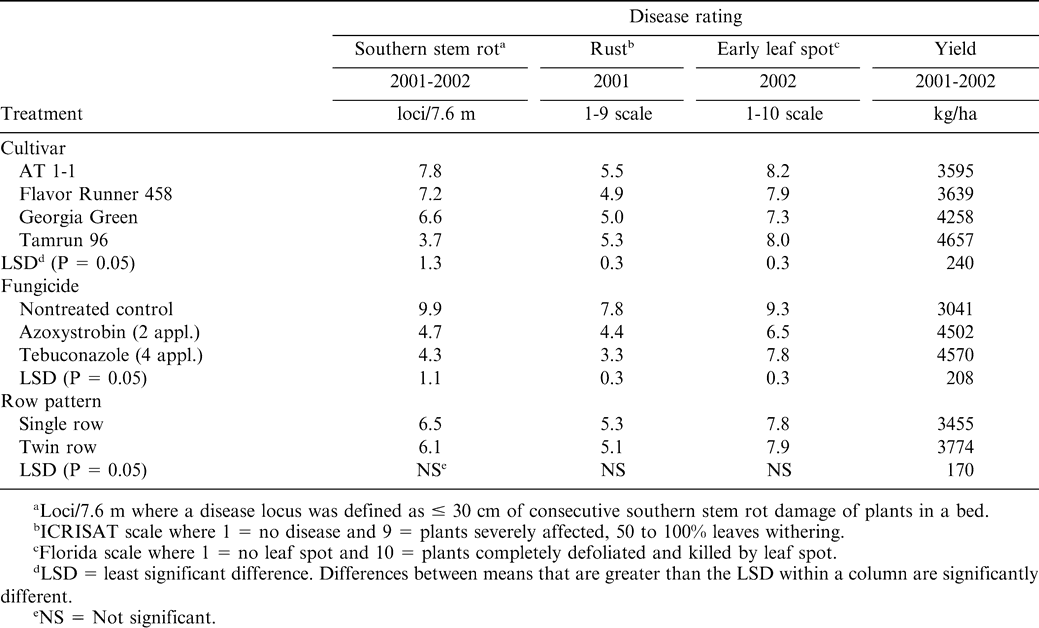
Southern stem rot incidence was lower when azoxystrobin and tebuconazole were applied regardless of row pattern or cultivar compared with the nontreated control (Table 2). Besler et al. (1996) found that tebuconazole reduced southern stem rot incidence by 53%. No difference in southern stem rot control was noted between azoxystrobin and tebuconazole. These results also agree with Grichar et al. (2000) who concluded that azoxystrobin was comparable with tebuconazole for control of southern stem rot. The cultivar Tamrun 96 had lower southern stem rot disease incidence than all other cultivars when averaged across fungicide treatments and row pattern (Table 2). Besler et al. (1997) concluded that Tamrun 96 was low in southern stem rot disease incidence over a 3-yr period when compared to other cultivars. While AT 1-1 had the highest southern stem rot disease incidence, the reactions of Flavor Runner 458 and Georgia Green were intermediate.
Rust
Azoxystrobin and tebuconazole reduced rust when compared to the nontreated control (Table 2). Tebuconazole controlled rust more effectively than azoxystrobin. Because rust was more evident later in the season, improved control could be attributed to the late-season application of tebuconazole. Most cultivars had similar rust pressure when averaged across fungicide treatments and row pattern. Flavor Runner 458 had less rust than AT 1–1 and Tamrun 96 but not Georgia Green (Table 2).
Early Leaf Spot
Early leaf spot severity was extremely high in 2002 when fungicides were not applied (Table 2). This resulted in some plants being completely defoliated. Although azoxystrobin and tebuconazole reduced early leaf spot incidence when compared with the nontreated control, early leaf spot was still considered high in those treatments (Table 2). Heavy rainfall mid- to late-season coupled with favorable temperatures and humidity caused severe early leaf spot pressure, which overwhelmed plots that received fungicide applications. When averaged across cultivars and row pattern, azoxystrobin and tebuconazole reduced leaf spot compared to the nontreated control. Grichar et al. (2000) reported that azoxystrobin applied at rates that ranged from 0.22 to 0.45 kg/ha controlled early leaf spot comparably to tebuconazole.
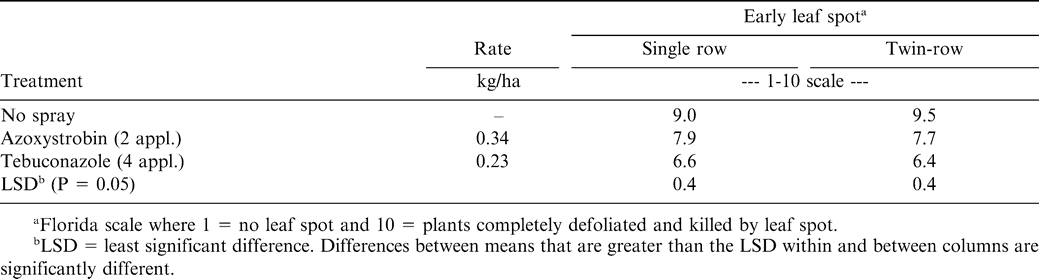
Each cultivar displayed a high level of early leaf spot incidence (Table 2). However, when averaged across fungicide treatments and row pattern, Georgia Green had less early leaf spot than all other cultivars. Georgia Green was reported to have intermediate leaf spot tolerance between Southern Runner and Florunner (Branch and Culbreath, 1995). There was a significant fungicide treatment × row pattern with more early leaf spot in the nontreated control plots where cultivars were planted in a twin-row configuration (Table 3). Azoxystrobin and tebuconazole lowered early leaf spot severity in both row pattterns when compared to the nontreated control. Tebuconazole lowered early leaf spot severity compared to azoxystrobin in both the single and twin-row pattern.
Pod Yield
When averaged across row patterns and cultivar, pod yields were higher when azoxystrobin and tebuconazole were applied when compared with the nontreated peanut (Table 2). However, no difference in yield was noted when comparing peanut treated with azoxystrobin and tebuconazole. Tamrun 96, when averaged across fungicide treatments and row pattern, yielded more than the other cultivars (Table 2). Georgia Green was higher in yield than AT 1–1 or Flavor Runner 458. Moderate southern stem rot disease incidence both years, high rust in 2001, and high early leaf spot severity in 2002, respectively, contributed to lower yields with cultivars AT 1–1 and Flavor Runner 458. However, due to excessive late-season rainfall in 2002, these two cultivars were not inverted at the optimum time, which may have contributed to lower yields.
Row pattern effects on yield revealed that the twin-row pattern was significantly higher in yield then the single row pattern (Table 2). Baldwin et al. (1998) found that when averaged across four runner cultivars and locations in Georgia, twin rows significantly increased yield by 381 kg/ha in the absence of disease. Another study conducted in Georgia in 1997 and 1998 found 716 kg/ha and 375 kg/ha increases in yield, respectively, in twin-rows compared to the single row pattern (McGriff et al., 1999). In the aforementioned studies, plants/m on a bed was held constant for both row patterns.
This study revealed that azoxystrobin and tebuconazole effectively controlled both southern stem rot and rust but was not as effective for control of early leaf spot. Azoxystrobin and tebuconazole were applied two and four times, respectively, throughout the growing season. Because leaf spot severity was high, additional fungicide applications would be required for better control. This study also revealed that Tamrun 96 was lower in southern stem rot incidence than AT 1–1, Flavor Runner 458, and Georgia Green. Therefore, growers who have fields with a history of southern stem rot may benefit by planting this cultivar. Peanuts planted in a twin-row pattern had higher yields than peanuts planted in a single row pattern when averaged across cultivar and fungicides. Based on these findings, there may be an advantage to planting twin-rows to increase yield.
Acknowledgements
The authors would like to express their appreciation to Kevin Brewer, Bill Klesel, and Dwayne Drozd for their assistant in data collection and plot harvesting. We would also like to thank the Texas Peanut Producers Board for their partial support of this research.
Literature Cited
Backman P. A. and Crawford M. A. 1984 Relationship between yield loss and severity of early and late leaf spot diseases of peanut. Phytopathol 74 : 1101 – 1103 .
Baldwin J. A. , Beasley J. P. , Brown S. L. , Todd J. W. , and Culbreath A. K. 1998 Yield, grade, and tomato spotted wilt virus incidence of four peanut cultivars in response to twin versus single row planting patterns. Proc. Amer. Peanut Res. Educ. Soc 30 : 51 (abstr.) .
Besler B. A. , Grichar W. J. , and Jaks A. J. 1996 Southern blight and leaf spot control in peanut using selected fungicides. Texas J. Agric. Nat. Resour 9 : 105 – 114 .
Besler B. A. , Grichar W. J. , and Smith O. D. 1997 Reaction of selected peanut cultivars and breeding lines to southern stem rot. Peanut Sci 24 : 6 – 9 .
Besler B. A. , Grichar W. J. , Smith O. D. , and Jaks A. J. 2001 Response of peanut cultivars to full and reduced spray programs of tebuconazole for control of southern stem rot. Peanut Sci 28 : 5 – 8 .
Boyle L. W. 1961 The ecology of Sclerotium rolfsii with emphasis on the role of saprophytic media. Phytopathol 51 : 117 – 119 .
Branch W. D. and Culbreath A. K. 1995 Combination of early maturity and leaf spot tolerance within advanced Georgia peanut breeding lines. Peanut Sci 22 : 106 – 108 .
Brenneman T. B. , Murphy A. D. , and Csinos A. S. 1991 Activity of tebuconazole on Sclerotium rolfsii and Rhizoctonia solani, two soilborne pathogens of peanut. Plant Dis 75 : 744 – 747 .
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , Kucharek T. A. , and Knauft D. A. 1988 Components of resistance to late leaf spot in peanut. I. Levels and variability - Implications for selection. Peanut Sci 15 : 25 – 30 .
Grichar W. J. , Besler B. A. , and Jaks A. J. 2000 Use of azoxystrobin for disease control in Texas peanut. Peanut Sci 27 : 83 – 87 .
Hagan A. K. , Weeks J. R. , and Bowen K. 1991 Effects of application timing and method on control of southern stem rot of peanut with foliar-applied fungicides. Peanut Sci 18 : 47 – 50 .
Harrison A. L. 1970 The effects of seeding rates and multiple rows per bed on peanut production under irrigation. J. Amer. Peanut Res. Educ. Assoc 2 : 47 – 50 .
Jacobi J. C. and Backman P. A. 1991 Effects of tebuconazole on seed quality and fungal colonization on two cultivars of peanut. Phytopathol 81 : 812 .
Jaks A. J. , Grichar W. J. , and Besler B. A. 1998 Evaluation of advisory and calendar spray programs on peanut disease control and yield in Texas. Proc. Amer. Peanut Res. Educ. Soc 30 : 43 (abstr.) .
Lunsford J. N. , Black D. , and Royal S. 1998 Early leaf spot control in peanuts with azoxystrobin formulations. Proc. Amer. Peanut Res. Educ. Soc 30 : 35 (abstr.) .
McGriff D. E. , Baldwin J. A. , and Hudgins J. E. 1999 Yield response of several peanut cultivars when planted in single and twin row patterns during 1997–98 in Decatur County. Proc. Amer. Peanut Res. Educ. Soc 31 : 54 (abstr.) .
Melouk H. A. and Backman P. A. 1995 Management of soilborne fungal pathogens. 75 – 82 In Melouk H. A. and Shokes F. M. eds. Peanut Health Management APS Press St. Paul, MN.
Nutter F. W. and Shokes F. M. 1995 Management of foliar diseases caused by fungi. 43- 49 In Melouk H. A. and Shokes F. M. eds. Peanut Health Management APS Press St. Paul, MN.
Rodriquez-Kabana R. , Backman P. A. , and Williams J. C. 1975 Determination of yield losses to Sclerotium rolfsii in peanut fields. Plant Dis. Rep 59 : 855 – 858 .
Sconyers L. E. , Brenneman T. B. , and Stevenson K. L. 2002 Effect of twin row spacing on epidemiology of peanut stem rot. Proc. Amer. Peanut Res. Educ. Soc 34 : 46 (abstr.) .
Shokes F. M. and Culbreath A. K. 1997 Early and late leaf spots. 17 – 20 In Burelle N. K. , Porter D. M. , Kabana R. R. , Smith D. H. , and Subrahmanyan P. eds. Compendium of Peanut Diseases APS Press St. Paul, MN .
Smith D. H. and Littrell R. H. 1980 Management of peanut foliar diseases with fungicides. Plant Dis 64 : 356 – 361 .
Smith H. R. and Lee T. A. 1986 Effects of propiconazole, PCNB, and metalaxyl + PCNB on Sclerotium rolfsii of peanuts. Proc. Amer. Peanut Res. Educ. Soc 18 : 71 (abstr.) .
Subrahmanyam P. , McDonald D. , Gibbons R. W. , Nigam S. N. , and Nevill D. J. 1982 Resistance to rust and late leaf spot diseases in some genotypes of Arachis hypogaea. Peanut Sci 9 : 6 – 10 .
Subrahmanyam P. , Vidyasagar K. R. , McDonald D. , Moss J. P. , and Gibbons R. W. 1989 Origins of resistance to rust and late leaf spot in peanut (Arachis hypogaea Fabaceae). Econ. Bot 43 : 444 – 455 .
Van Arsdel E. P. and Harrison A. L. 1972 Possible origin of peanut rust epidemics in Texas. Phytopathol 62 : 794 (abstr.) .