Introduction
Weed flora associated with peanut are characterized by populations of many plant species that have different competitive abilities (Buchanan et al., 1982). Florida beggarweed consistently ranks as the most troublesome weed in peanut surveys for Alabama, Florida, and Georgia (Buchanan et al., 1982; Elmore, 1989; Dowler, 1992, 1998; Prostko, 2001; Webster and MacDonald, 2001). Florida beggarweed is a vigorous competitor when established within the first 8 wks of peanut emergence (Buchanan et al., 1976; Hauser et al., 1982; 1975; Cardina and Brecke, 1991). Lack of early season Florida beggarweed control can lead to season-long interference that reduces yield (Hauser et al., 1975; 1982; Cardina and Brecke, 1991; Wehtje et al., 2000a; 2000b). Poor control hinders crop harvest by interfering with digging and threshing (Buchanan et al., 1982; Wehtje et al., 1999).
Florida beggarweed morphology can vary with location and this contributes to its adaptability to changing environments, cultural practices, and control measures (Cardina and Brecke, 1989). Light interception by Florida beggarweed reaches a maximum at 80 to 90 d after peanut emergence (Cardina and Brecke, 1991). Florida beggarweed that emerges with peanut can overtop the crop canopy at 52 d after planting; at 80 d after planting the distance of light interference to the crop canopy can extend to 160 cm around the weed. At densities of 1.0 plant/m2, crop yield can be reduced 20 to 40% (Barbour and Bridges, 1995).
Postemergence (POST) weed control often results in variable Florida beggarweed control. Paraquat provides only early postemergence (EPOST) contact control (Grey et al., 2003), while chlorimuron provides contact and residual activity (Wehtje et al., 2000a). Peanut tolerance to paraquat was first noted in the late 1970s (Wehtje et al., 1986). However, lack of residual activity and the narrow window of application for control of Florida beggarweed reduced the effectiveness of this treatment (Wilcut et al., 1995). Chlorimuron effectively controls Florida beggarweed, but registration states that application can begin only after 60 d have passed since peanut emergence (Johnson et al., 1992b). This frequently becomes a mid-season salvage treatment (Johnson et al., 1992a). By 60 d after peanut emergence, Florida beggarweed can be taller than the 25 cm in height, which is the maximum specified on the chlorimuron registration (Webster et al., 1997; Wehtje et al., 1999). Chlorimuron is still applied to approximately 18% of the Georgia crop (NASS, 2003). Producers have complained that they cannot consistently control Florida beggarweed with chlorimuron. Alternative methods of Florida beggarweed control have been evaluated, including mowing (Wehtje et al., 1999) and wick-bar (Johnson et al., 1999), but cost limit practical use of either method.
Florida beggarweed is the most common and troublesome weed species in Southeastern peanut production. Studies that have compared chemical and mechanical control of Florida beggarweed in the same experiment at different times of emergence have not been reported. Thus, research was conducted to evaluate chemical verses mechanical control methods, and time of emergence effects on Florida beggarweed establishment, growth, and competition in peanut.
Materials and Methods
Experiments were conducted in Georgia in 1992, 1993, and 1994 at the Bledsoe Research farm near Williamson in different parts of the same field. Soil was a Cecil sandy clay loam (clayey, kaolinitic, thermic, Typic Hapludults) with 1 to 2% organic matter and pH 6.1 to 6.5.
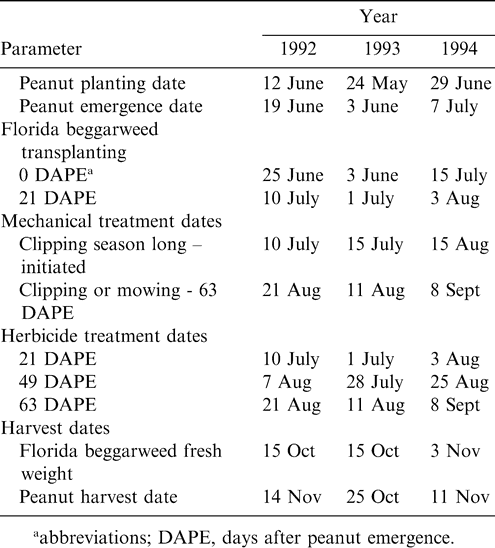
Ethalfluralin (0.8 kg/ha) was preplant incorporated to a depth of 5 to 7 cm over the entire test area. Peanut was planted 4 to 5 cm deep and spaced 5 to 6 cm apart. Individual plots consisted of four 76-cm wide by 12 m long rows. Plots were irrigated as needed. Fertilizer, fungicides, and insecticides were applied based on University of Georgia extension recommendations for peanut production (Brown, 1990). Plots were maintained weed-free by hand hoeing for the entire season to remove all weeds except Florida beggarweed. Peanut ‘Florunner' planting and emergence, Florida beggarweed transplanting, clipping, mowing, spray application, and harvest dates are presented in Table 1.
In a greenhouse, Florida beggarweed seed (Adams-Briscoe Seed Co. 325 East 2nd Street, Jackson, GA 30233) were sown in soil from the same field where the test was conducted. Flats of Florida beggarweed were established at 3 wk intervals in order to have the appropriate size transplants when needed for field establishment. Two-day-old, cotyledon stage Florida beggarweed were then hand transplanted at peanut emergence or 21 d after peanut emergence (DAPE) (Table 1). Florida beggarweed were established in the peanut row at one plant per 1.3 m2 beginning 0.5 m from the front of the plot for a total of 48 plants per plot.
Treatments included a weed-free check and a nontreated weedy check of Florida beggarweed established at peanut emergence. Five treatments were applied to Florida beggarweed established at peanut emergence and at 21 DAPE. These included chlorimuron at 4.0 g ai/ha applied 49 or 63 DAPE; Florida beggarweed clipped weekly to the crop canopy height after reaching 15 cm; Florida beggarweed clipped starting 63 DAPE; and mowing of Florida beggarweed at 63 DAPE. In addition Florida beggarweed planting at peanut emergence was followed by paraquat at 64.0 g ai/ha applied at 63 DAPE. Additional treatments were tested on plots established with Florida beggarweed at 21 DAPE including paraquat at 64 g ai/ha 21 DAPE , and paraquat at 64 g ai/ha 21 DAPE in combination with chlorimuron 63 DAPE. The last two treatments consisted of a Florida beggarweed infested (weedy check) and weed-free check also were evaluated. This treatment served to mimic Florida beggarweed emergence after a paraquat treatment at 21 DAPE EPOST. For this, treatments were applied in the morning and Florida beggarweed was established in the afternoon.
Herbicides were applied with a CO2-pressurized backpack sprayer calibrated to deliver 187 L/ha at 210 kPa. Treatments were applied with a nonionic surfactant at 0.25% v/v. Clipping treatments were applied when Florida beggarweed branching stems were 15 cm above the peanut canopy. Branches were hand clipped back to peanut canopy height with scissors, and removed from the plot. Similarly, mowing treatments were performed to the top of the canopy with a string trimmer but stems were left in the plot.
For all treatments, Florida beggarweed was 3 to 8 cm tall at 21 DAPE, 40 to 60 cm tall at 49 DAPE, and 75 to 91 cm tall at 63 DAPE. Peanut height ranged from 4 to 8 cm tall at 21 DAPE, 28 to 35 cm at 49 DAPE, and 46 cm at 63 DAPE.
Final Florida beggarweed counts were taken on whole plots in October of each year. Survival of treatments was calculated based on the number of Florida beggarweed per plot as compared to survival for the weedy check. Florida beggarweed was hand harvested from the entire plot by cutting plants to the soil surface prior to peanut harvest and height and fresh weight recorded. These data were used to establish Florida beggarweed g/plant, kg/ha, and biomass as a percentage of the weed-free-check, and cm height. Florida beggarweed biomass is reported as a percentage of the nontreated check to reduce the variation associated with differences between treatments in biomass yield. Peanut was dug and inverted based on mesocarp pod color (William and Drexter 1981). Pods were combined 10 to 20 d later with conventional harvesting equipment.
The experimental design was a randomized complete block with three replications. Data were subjected to ANOVA using SAS and tested for year by treatment interactions. Treatments means were separated by Fisher's Protected LSD Test at P ≤ 0.05.
Results and Discussion
Significant year-by-treatment interactions were noted for final Florida beggarweed percent survival, stand, fresh weight on a per-plant and -ha basis, height, and peanut pod yield. Thus, data for these variables are presented for individual experiments. No year-by-treatment interaction for Florida beggarweed fresh weight as a percentage of the nontreated check was noted so data for this variable were combined.
Florida Beggarweed Survival
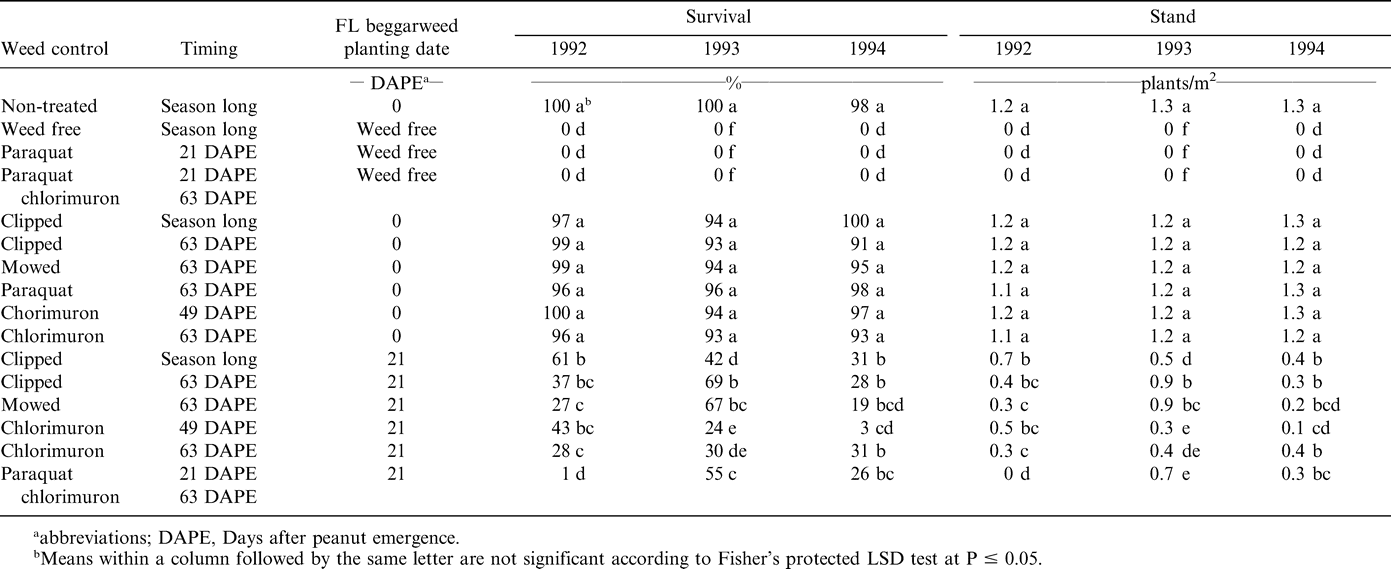
Treatments with Florida beggarweed transplanted at peanut emergence did not differ in percent survival (Table 2). Survival was 91% or greater in the non-treated control, and in treatments clipped season long, clipped beginning at 63 DAPE, mowed at 63 DAPE, paraquat at 63 DAPE, and chlorimuron at 49 or 63 DAPE Stand was 1.2 plants/m2 or greater for all years (Table 2). These responses indicate that a single mechanical or chemical control method did not reduce survival of Florida beggarweed that was established at peanut emergence. These data indicate that chlorimuron applied at 49 or 63 DAPE will not kill Florida beggarweed that emerge with the crop. In contrast, Florida beggarweed survival was 69% or less and stand was 0.9 plants/m2 or less in all treatments when Florida beggarweed was transplanted 21 DAPE followed by clipping, mowing, or paraquat 21 DAPE plus chlorimuron 63 DAPE. These data indicate that Florida beggarweed emerging after paraquat was applied 2 to 3 wks after peanut emergence, which is a standard herbicide practice in the Southeast (Wilcut et al., 1995), should be controlled with chlorimuron. Wehtje et al. (2000a) indicated that control of emerged Florida beggarweed is often achieved with paraquat applied 15 to 20 DAPE and that a subsequent application of chlorimuron at 63 DAPE can significantly improve control.
Florida Beggarweed Fresh Weight
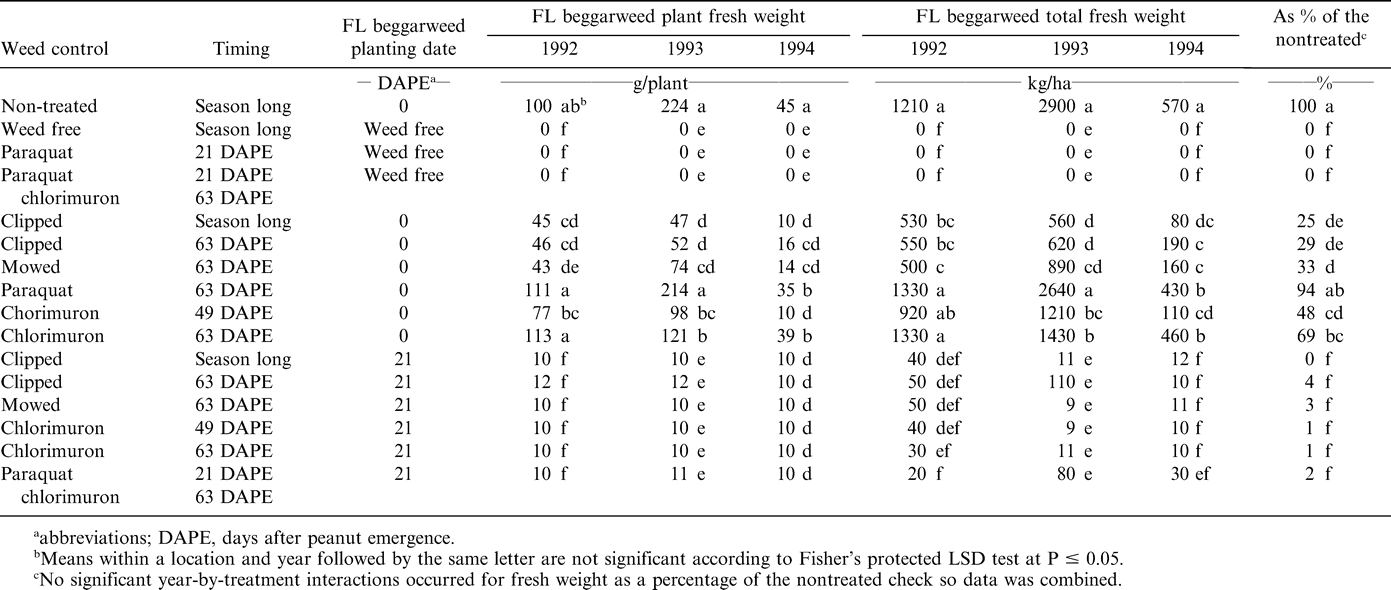
At the end of the season, fresh weight biomass of Florida beggarweed established at peanut emergence was 100 g/plant in 1992, 224 g/plant in 1993, and 45 g/plant in 1994 (Table 3). When Florida beggarweed was established at peanut emergence was clipped or mowed, weights ranged from 43 to 74 g/plant in 1992 and 1993 but 16 g or less in 1994. These differences were attributed to the late planting of Florida beggarweed in 1994 (Table 1). Florida beggarweed branched from nodes when clipped or mowed. Fresh weights of clipped or mowed plants for the treatments established at peanut emergence were significantly different from the nontreated control. Florida beggarweed total biomass mirrored average biomass in that the longer the plants survived, the greater the biomass accumulated by the end of the season (Table 3).
Florida beggarweed was not susceptible to paraquat at 63 DAPE (Table 3). Although paraquat was effective when applied up to 28 DAPE, it was ineffective when applied 63 DAPE. Poor control for paraquat applied at 63 DAPE was attributed to lack of translocation, and the large size of Florida beggarweed prevented good coverage.
When chlorimuron was applied at 49 DAPE to Florida beggarweed established at peanut emergence, biomass ranged from 77 g/plant in 1992 and 98 g/plant in 1993 to 10 g/plant 1994 (Table 3). When chlorimuron was applied at 63 DAPE biomass was 113 g/plant in 1992, 121 g/plant in 1993, and 39 g/plant in 1994. While survival of Florida beggarweed was not affected by either treatment, applying chlorimuron at 49 DAPE (14 days earlier than the 63 DAPE treatment) reduced Florida beggarweed plant biomass each year. None of the treatments affected biomass of Florida beggarweed established 21 DAPE (Table 3). As a percentage of the nontreated control, biomass of Florida beggarweed established at 21 DAPE for any treatment was always less than any of the 0 DAPE established treatments.
Hauser et al. (1975) reported season ending Florida beggarweed fresh weight of 6840 kg/ha for 8 plants/m2 for season-long interference. When Florida beggarweed was removed the first 2 weeks of the season then allowed to compete the remainder of the season, fresh weight decreased to 4140 kg/ha for 7 plants/m2. As peanut were kept free of Florida beggarweed for longer periods of time, the average fresh weight decreased. This same effect was noted for this experiment; chemical and mechanical control methods significantly reduced average plant fresh weight when compared to the non-treated check.
Mechanical destruction of Florida beggarweed reduced biomass to 33% or less of the non-treated control (Table 3). This response indicates that a significant proportion of the weed's biomass can be removed by mowing or clipping irregardless of time of emergence. However, Florida beggarweed established at peanut emergence had 48, 69, and 94% of the non-treated biomass when chlorimuron was applied at 49 and 63 DAPE, and paraquat was applied at 63 DAPE, respectively. In comparison, Florida beggarweed transplanted 21 DAPE fresh weights had 1, 1, and 2% of the control when chlorimuron was applied at 49 or 63 DAPE, or paraquat was applied at 21 DAPE followed by chlorimuron at 63 DAPE.
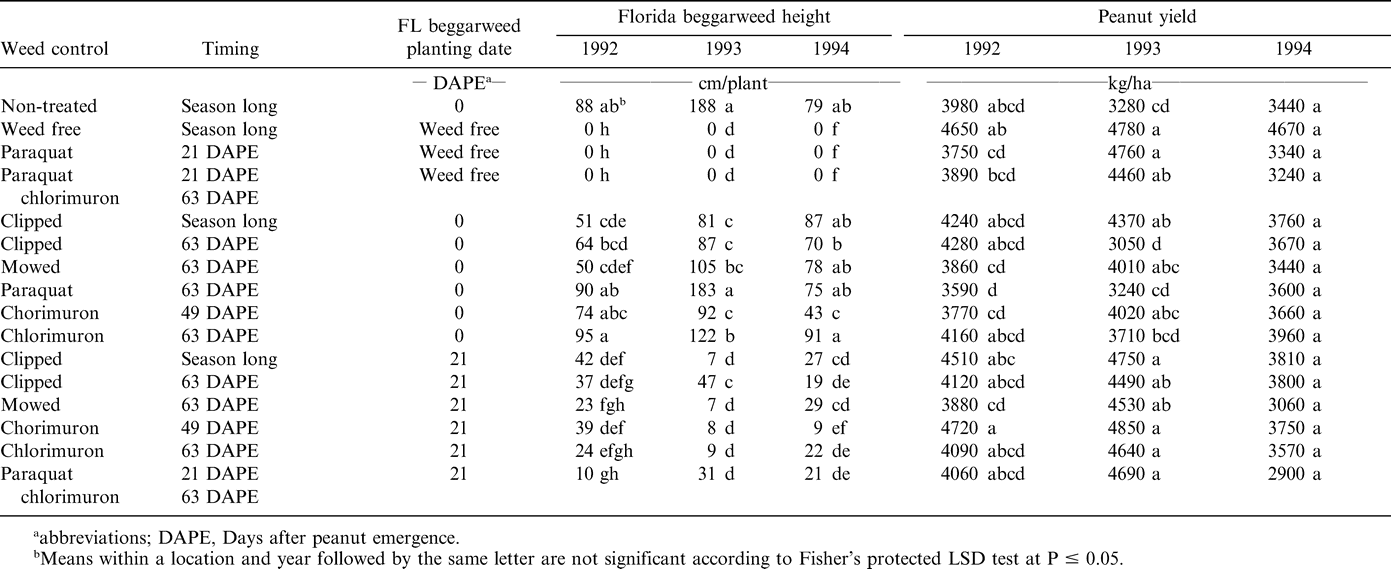
Florida beggarweed plant height was reflective of stand and biomass. Non-treated Florida beggarweed established at peanut emergence were 79 to 188 cm tall (Table 4). For all treatments established at peanut emergence, Florida beggarweed heights exceeded 43 cm. In contrast, Florida beggarweed was 47 cm or less for all 21 DAPE treatments. Previous research indicated that as wild poinsettia grew taller, it caused interference which reduced peanut yield (Barbour and Bridges, 1995) while other research (Royal et al., 1997) indicated the interception of fungicides occurred with larger weeds. For our research, the effective control of Florida beggarweed began with an application of paraquat 21 DAPE. Even though paraquat has no residual activity, the almost complete burndown control it provided then set the stage for effective control of later emerging Florida beggarweed with chlorimuron at 49 and 63 DAPE. The biomass reductions plus control likely increased harvesting efficiency. The root systems of treatments that clipped Florida beggarweed at 63 DAPE caused no problem with digging, inversion, or combining as this treatment occurred at least 63 days prior to these operations (Table 1).
Peanut Yield
Yield from the weed-free check was more than 4600 kg/ha in each year (Table 4). Florida beggarweed established at peanut emergence in 1992 and 1993 consistently reduced yield even when a mechanical or chemical method was used for control. No significant yield reduction occurred for any treatment in 1994 as there was variability in yield associated with the late planting date due to adverse environmental conditions. Yields in the non-treated control with Florida beggarweed transplanted at peanut emergence were 3980, 3280, and 3440 kg/ha in 1992, 1993, and 1994, respectively. This corresponds to the previous reports of 20 to 40% yield reduction from season long Florida beggarweed competition models (Barbour and Bridges, 1995). Buchanan et al. (1976) reported biomass correlated more closely with the weights of the weeds rather than with their populations.
Paraquat applied 21 DAPE to weed free peanut yield was 3750 and 3340 kg/ha as compared to the weed free of 4650 and 4670 kg/ha in 1992 and 1994, respectively (Table 4). These data indicate that in the absence of Florida beggarweed, paraquat applied 21 DAPE reduced yield, although not significant in 1994, as much as Florida beggarweed established at peanut emergence. This same trend for peanut yield reduction occurred for paraquat applied at 21 DAPE followed by chlorimuron 63 DAPE in 1992 and 1994. These trends were not observed in 1993.
Yields were 4720, 4850, and 3750 kg/ha in 1992, 1993, and 1994 respectively, when chlorimuron was applied 49 DAPE (Table 4). Although not significant, peanut yield for chlorimuron applied at 63 DAPE was less in 1992, 1993, and 1994 than when applied at 49 DAPE. Since these studies were conducted, research has indicated no difference for peanut yield of six cultivars treated with chlorimuron at 49 or 63 DAPE (Wehtje and Grey, 2004).
Conclusions
Mechanically controlling Florida beggarweed established at peanut emergence was as effective as chlorimuron applied at either 49 or 63 DAPE. Excellent Florida beggarweed control was achieved with chlorimuron applied at 63 DAPE if treatment was targeted to control plants emerging 21 DAPE. However, cost associated with mechanical control could limit use (Wehtje et al., 1999). For all variables, Florida beggarweed established 21 DAPE responded differently from the Florida beggarweed established at peanut emergence. EPOST applications of paraquat controlled Florida beggarweed as previously noted (Wehtje et al 2000). Florida beggarweed control can be improved with chlorimuron at 49 DAPE versus 63 DAPE. Herbicide timings and combinations affect on competition should be further investigated.
Literature Cited
Barbour J. C. and Bridges D. C. 1995 A model of competition for light between peanut and broadleaf weeds. Weed Sci 43 : 247 – 257 .
Brown S. M. 1990 Peanut pest management. 71 In Brown E. A. ed. 1990 Georgia Pest Control Handbook Univ. Georgia, Coop. Ext. Serv Athens, GA .
Buchanan G. A. , Murray D. S. , and Hauser E. W. 1982 Weeds and their control in peanuts. 206 – 249 In Pattee H. E. and Young C. T. eds. Peanut Science and Technology American Peanut Research and Education Society, Inc Yoakum, TX .
Buchanan G. A. , Hauser E. W. , Ethredge W. J. , and Cecil S. R. 1976 Competition of Florida beggarweed and sicklepod with peanuts. II. Effects of cultivation, weeds, and SADH. Weed Sci 24 : 29 – 39 .
Cardina J. and Brecke B. J. 1991 Florida beggarweed (Desmodium tortuosum) growth and development in peanuts (Arachis hypogaea). Weed Technol 5 : 147 – 153 .
Cardina J. and Brecke B. J. 1989 Growth and development of Florida beggarweed (Desmodium tortuosum) selections. Weed Sci 37 : 207 – 210 .
Dowler C. C. 1992 Weed survey - southern states. Proc. South. Weed Sci. Soc 45 : 396 .
Dowler C. C. 1998 Weed survey - southern states. Proc. South. Weed Sci. Soc 51 : 304 .
Elmore C. D. 1989 Weed survey - southern states. Proc. South. Weed Sci. Soc 42 : 413 .
Grey T. L. , Bridges D. C. , Eastin E. F. , Prostko E. P. , Vencill W. K. , Johnson W. C. , Brecke B. J. , MacDonald G. E. , Dacur J. A. , Everest J. W. , Wehtje G. R. , and Wilcut J. W. 2003 Residual weed control with imazapic, diclosulam, and flumioxazin in southeastern peanut (Arachis hypogaea). Peanut Sci 30 : 23 – 28 .
Hauser E. W. , Buchanan G. A. , and Ethredge W. J. 1975 Competition of Florida beggarweed and sicklepod with peanuts. I. Effects of periods of weed-free maintenance or weed competition. Weed Sci 23 : 368 – 372 .
Hauser E. W. , Buchanan G. A. , and Ethredge W. J. 1982 Effects of Florida beggarweed (Desmodium tortuosum) and sicklepod (Cassia obtusifolia) on peanuts. Weed Sci 30 : 602 – 604 .
Johnson W. C. , Colvin D. L. , Littlefield T. A. , and Mullinix B. G. 1999 Florida beggarweed (Desmodium tortuosum) and sicklepod (Senna obtusifolia) control in peanut using herbicides applied through a wick-bar. Peanut Sci 26 : 18 – 23 .
Johnson W. C. , Holbrook C. C. , Mullinix B. G. , and Cardina J. 1992a Response of eight genetically diverse peanut genotypes to chlorimuron. Peanut Sci 19 : 111 – 115 .
Johnson W. C. , Mullinix B. G. , and Brown S. M. 1992b Phytotoxicity of chlorimuron and tank mixtures on peanut (Arachis hypogaea). Weed Technol 6 : 404 – 408 .
National Agricultural Statistics Service (NASS) 2003 National Agricultural Statistics Service U.S. Dept. Agri. Published Estimates Database NASS-USDA, Washington, DC .
Prostko E. P. 2001 Weed survey - southern states. Proc. South. Weed Sci. Soc 54 : 249 .
Royal S. S. , Brecke B. J. , Shokes F. M. , and Colvin D. L. 1997 Influence of broadleaf weeds on chlorothalonil deposition, foliar disease incidence, and peanut (Arachis hypogaea) yield. Weed Technol 11 : 51 – 58 .
Webster T. M. and MacDonald G. E. 2001 A survey of weeds in various crops in Georgia. Weed Technol 15 : 771 – 790 .
Webster T. M. , Wilcut J. W. , and Coble H. D. 1997 Influence of AC 263,222 rate and application method on weed management in peanut (Arachis hypogaea). Weed Technol 11 : 520 – 526 .
Wehtje G. R. and Grey T. L. 2004 Response of New peanut cultivars to early postemergence chlorimuron applications. Peanut Sci 31 : 119 – 123 .
Wehtje G. R. , McGuire J. A. , Walker R. H. , and Paterson M. G. 1986 Texas panicum control in peanuts with paraquat. Weed Sci 34 : 308 – 311 .
Wehtje G. R. , Padgett D. , and Martin N. R. 2000a Performance and economic benefit of herbicides used for broadleaf weed control in peanut. Peanut Sci 27 : 11 – 16 .
Wehtje G. R. , Padgett D. , and Martin N. R. 2000b Imazapic-based herbicide systems for peanut and factors affecting activity on Florida beggarweed. Peanut Sci 27 : 17 – 22 .
Wehtje G. R. , Wells L. W. , Choate J. H. , Martin N. R. , and Curtis J. M. 1999 Mowing as a weed control supplement to herbicides and cultivation in peanut (Arachis hypogaea). Weed Technol 13 : 139 – 143 .
Wilcut J. W. , York A. C. , Grichar W. J. , and Wehtje G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea). 207 – 244 In Pattee H. E. and Stalker H. T. eds. Advances in Peanut Science American Peanut Research and Education Society, Inc Stillwater, OK .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Notes
- 1Asst. Prof. and Asst. Dean and Prof, Dept. of Crop and Soil Sci., Coastal Plain Experiment Station, University of Georgia, P.O. Box 748, 115 Coastal Way, Tifton, GA 31793. [^] *Corresponding author - Tim Grey.