Introduction
Paraquat is often used in peanut for postemergence (POST) broadleaf weed control of sicklepod (Senna obtusifolia L.), Florida beggerweed (Desmodium tortosum D.C) and morningglory species (Ipomoea spp.) in the southern US (Wilcut et al. 1990; Wilcut et al. 1989; Wilcut and Swann 1990). Paraquat is labeled in peanuts up to 28 d after ground cracking with up to two applications at a total of 280 g ai/ha (Anonymous 2016). Broadcast applications of paraquat, prior to 28 day restriction, causes foliar injury to peanuts, however, it does not lead to yield loss (Wehtje et al. 1986). Other research has also confirmed runner type and virginia market-type peanuts are tolerant to paraquat if applied prior to pegging and fruit development at a rate less than 280 g ai/ha (Grichar and Dotray 2012; Wehtje et al. 1991; Wilcut and Swann 1990).
Paraquat tank mixed with 2,4-DB and/or bentazon is a frequently utilized POST program in peanut (Brecke and Colvin 1991; Wilcut et al. 1989, 1994b). Paraquat plus bentazon tank mixes control more broadleaf weeds including bristly starbur (Aeanthospermum hispidum DC.), coffee senna (Cassia oecidentalis L.), prickly sida (Sida spinosa L.), and smallflower morningglory (Jacquemontia tamifolia L.) than either herbicide applied alone (Wehtje et al. 1992; Wilcut et al. 1994a). Using 2,4-DB in combination with acifluorfen, bentazon, and paraquat will improve control of broadleaf weeds larger than the recommended size for treatment (Wilcut et al. 1994b). Additionally, bentazon acts as an antagonist to paraquat reducing paraquat efficacy on weed control; however it reduces peanut injury. Bentazon, a photosynthetic inhibitor, inhibits the Hill reaction in photosystem II and reduces the flow of electrons into photosystem I (Mine and Matsunaka 1975; Shaner 2014). Paraquat inhibits photosynthesis at photosystem I by diverting electrons creating oxygen singlets (Shaner, 2014). It has been shown that herbicides that inhibit photosystems II, such as bentazon, can cause herbicides that inhibit efficacy of photosystem I, such as paraquat. (Hogue and Warren 1970; Moore and Banks 1991). Another study reported bentazon interferes and reduces the absorption of paraquat on the leaf surface (Wehtje et al. 1992).
Lack of residual activity and a short window for application are the two main drawbacks for paraquat (Wilcut et al. 1995). Therefore, producers frequently apply residual herbicides in combination with paraquat to broaden the spectrum of weed control, provide residual control and to prevent the development of herbicide resistant weeds (Jordan et al. 2011; Wilcut et al. 1995). Chloroacetamides are residual herbicides that are often used to control annual grasses, yellow nutsedge (Cyperus exculentus L.) and broadleaf weeds in peanuts (Brecke and Colvin 1991; Wilcut et al. 1994). Previous studies in Virginia, Texas, North Carolina, and Alabama have observed peanut injury from the application of chloroacetamide herbicides (Cardina and Swan 1988; Grichar et al. 1996; Jordan et al. 2003; Wehtje et al. 1988). Soil pH, moisture, organic matter, as well as, herbicide rates can affect chloroacetamide injury on peanuts (Cardina and Swann 1988; Wehtje et al. 1988). However, none of the observed injury in these studies led to yield loss when applied at the labeled rates. Therefore, tank mixing a chloroacetamide herbicide with paraquat and 2,4-DB or bentazon may increase peanut injury under certain environmental conditions, but it will provide longer weed control and should not decrease peanut yield.
Previous studies have determined runner-type peanut tolerance to paraquat is neither cultivar dependent nor influenced by seed size (Johnson et al. 1993; Wehtje et al. 1991; 1994). New runner-type peanut cultivars with different growth characteristics and greater yield potential are being released; however, they have not been sufficiently evaluated for tolerance to frequently used paraquat based herbicide programs in the southeastern US. Therefore, the objective of this study was to evaluate the tolerance runner-type peanut cultivars (Georgia 06G, Georgia 12Y, Georgia 14N, and TufRunner 511) to paraquat based programs and determine if these programs may result in growth suppression and yield losses in peanut.
Materials and Methods
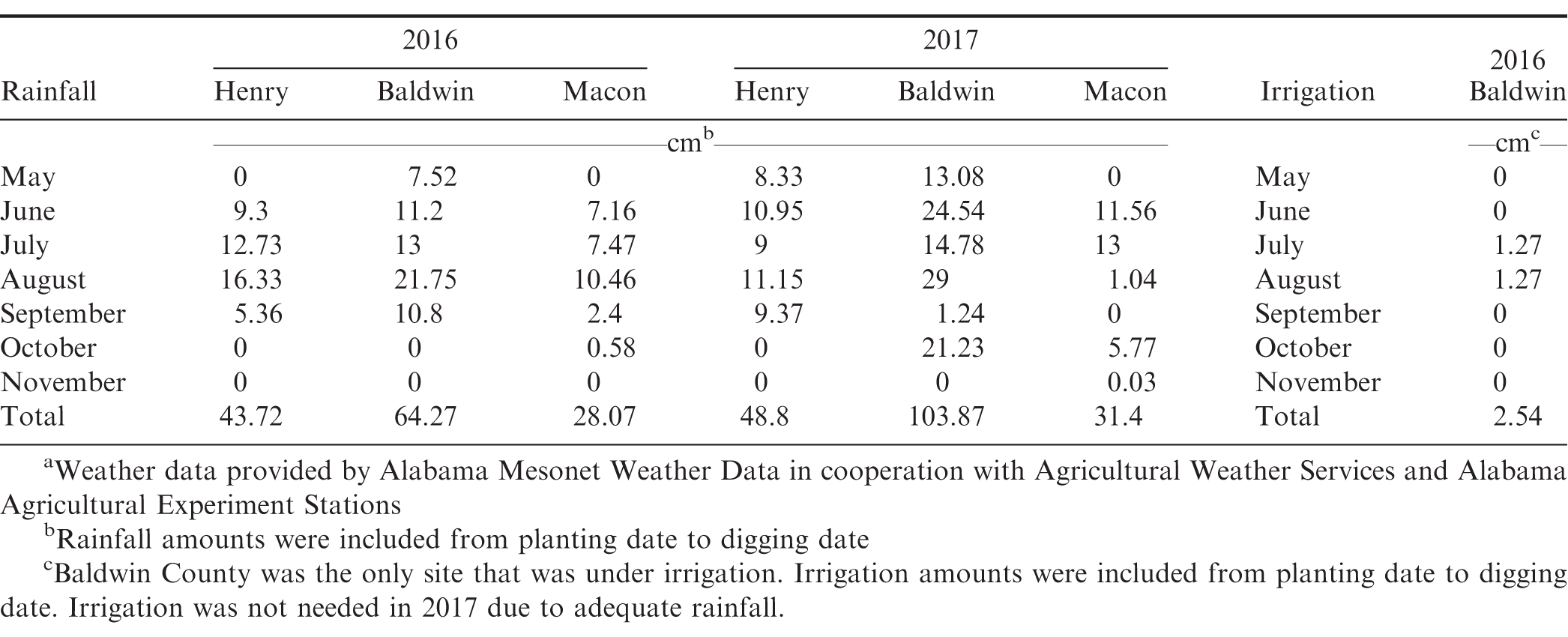
Field trials were conducted in Macon County (32°29'45.6"N 85°53'25.2"W), Baldwin County (30°32'45.7"N 87°52'52.2"W), and Henry County (31°21'17.1"N 85°19'35.3"W), Alabama in 2016 and 2017. Soils at the Macon County location were kalmia sandy loam (fine-loamy over sandy or sandy-skeletal, siliceous, semiactive, thermic Typic Hapludult), soils at the Henry County location were Dothan fine sandy loam, (fine-loamy, kaolinitic, thermic Plinthic Kandiudult), while soils at the Baldwin County location were a red bay fine sandy loam (fine-loamy, kaolinitic, thermic Rhodic Kandiudult). Soils at the Macon County location had a pH of 6.1 and organic matter (OM) of 0.9%, Henry County Location had a pH of 6.2 and OM 1.2% and the Baldwin County location had a pH of 5.6 and OM of 1.6%.
Fields were conventional tilled and experiments were set up as a split plot design with four replications. The main plot was herbicide treatment and the subplot was peanut cultivar. Each subplot contained two rows while a whole plot had eight rows of peanut. Peanut cultivars evaluated were Georgia 06G (Branch 2007), Georgia 12Y (Branch 2013), Georgia 14N (Branch and Brenneman 2015), and TufRunner 511 (Tillman and Gobert 2017). Subplots were 7.6 m long in Headland in 2016 and at all locations in 2017. Subplots in Macon and Baldwin counties in 2016 were 9.1m long. Peanuts were planted on 0.9 m wide rows at all locations. Henry, Macon, and Baldwin County trials were planted on May 25, May 27, and May 16, 2016, respectively, while in 2017, Henry, Macon, and Baldwin County trials were planted on May 9, June 9, and May 10 respectively. Flumioxazin at 107 g ai/ha was applied at planting and imazapic at 70 g ai/ha plus 2,4-DB 280 at g ai/ha plus S-metolachlor at 1470 g ai/ha were applied POST as needed to all treatments including non-treated check to provide season-long weed control. Hand-weeding was used whenever needed to maintain a weed-free trial.
Treatments were applied using a backpack sprayer with a six-nozzle boom (Teejet TT110025 wide angle flat nozzles, Teejet®, Spraying Systems Co. Wheaton, IL. 60187) using compressed CO2 at a spray volume of 187 L/ha. POST treatments were applied June 17, June 22, and June 13, 2016 in Henry, Macon and Baldwin County respectively; May 31, July 3, and June 2, 2017 in Henry, Macon and Baldwin County respectively. Henry and Macon County trials were planted dryland while Baldwin County trials were under irrigation. Table 1 includes rainfall and irrigation amounts for each location in 2016 and 2017. Although similar herbicide treatments were used each year, the paraquat rate was higher in 2017 since peanut varieties demonstrated sufficient tolerance, with little to no foliar burn, to paraquat at the recommended rate of 210 g ai/ha in 2016. While paraquat rates were different in 2016 (210 g ai/ha) and 2017 (280 g ai/ha), they fall within the labeled registration (Anonymous 2016). Additionally, several herbicide rates that were utilized in tank mixes with paraquat were changed from 2016 to 2017. In 2016, pyroxasulfone was applied at a higher than label rate of 179 g ai/ha, because at the time of the application it was not registered in peanuts so an estimated rate was used. In 2017, the newly registered labeled rate of 125 g ai/ha for peanut on sandy soils was used. Pre-mixture of pyroxasulfone plus carfentrazone (Anthem Flex®, FMC Corporation, Philadelphia, PA) was only used in 2016 study. Treatments can be found in Table 2 for 2016 and Table 3 for 2017. All treatments included a nonionic surfactant at 0.25 % v/v (Top Surf®, Winfield Solutions LLC. St. Paul, MN. 55164). Stand counts were recorded prior to POST treatment applications to ensure consistency between cultivars. In 2016, ten plant heights were randomly recorded in each subplot at 50 to 55 days after planting (DAP) and 72 to 78 DAP. In 2017, ten plant heights and canopy widths were randomly recorded in each subplot at 45 to 48 DAP and 66 to 68 DAP. Heights were measured from base of the plant at soil line to the highest growing point. Canopy widths were measured from furthest leaf tips horizontally across the peanut canopy at a spot randomly selected in the row. Based on peanut pod maturity (Williams and Drexler 1981) peanuts were dug October 3, October 6, October 24 in Henry, Baldwin, and Macon respectively in 2016. In 2017, peanuts were dug September 21, October 20, and November 8 in Henry, Baldwin, and Macon respectively. Pod yield was determined 4 to 7 d after digging utilizing a combine for each subplot.
Statistical analysis
All data was converted to a percentage of NTC prior to statistical analysis. Then, converted data was processed with PROC GLIMMIX procedure in SAS® 9.4 (SAS Institute Inc. Cary, NC. 27513). Cultivar, treatment, location and block were subjected to analyses of variance for a split plot treatment arrangement. Combined analysis over years was not conducted due to herbicide treatment differences among year. Treatment, location, and cultivar were considered fixed effects, while block was a random effect. If treatment by location was not significant, then location was used as a random effect and data was combined over location for analysis. If the interaction was significant, data was analyzed and presented by location. All means were separated using the Fisher's Protected LSD (P≤ 0.05) to reveal statistical differences.
Results and Discussion
Data was combined over peanut cultivars, as there was no significant cultivar by treatment interaction (P≤ 0.05) for any of the parameters evaluated during 2016 and 2017. Therefore, the results of this study indicate response of four cultivars to paraquat is not cultivar specific. This agrees with previous research that concluded paraquat tolerance in peanut was not cultivar dependent (Johnson et al. 1993; Wehtje et al. 1991; Wehtje et al.1994). Irrigation and dryland effects were considered a part of the location effects and not considered for analysis due to irrigation only being located at Baldwin County. Peanut stands prior to application were not significantly different at any location over both years; the stands were healthy and consistent and did not influence the results.
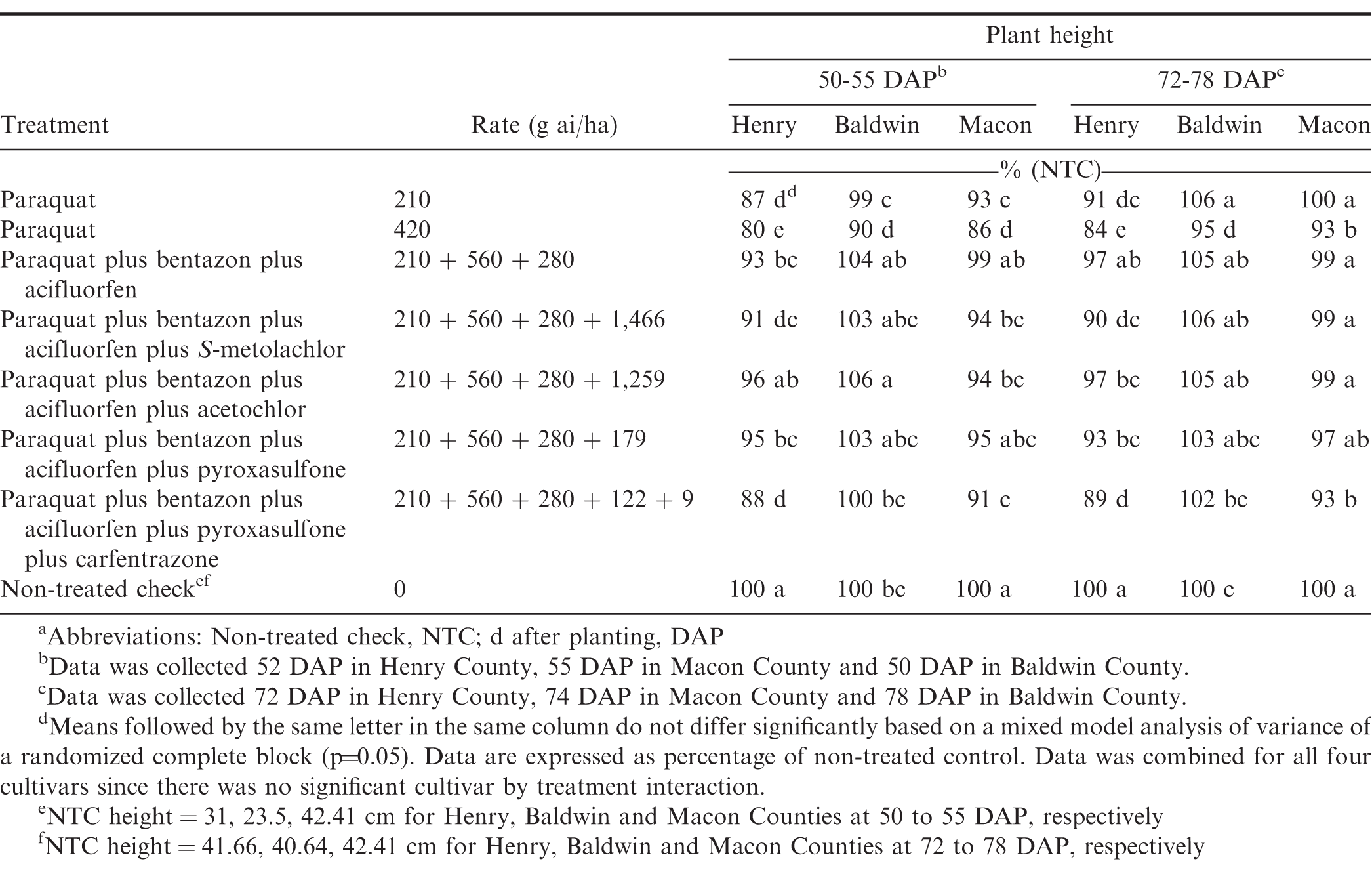
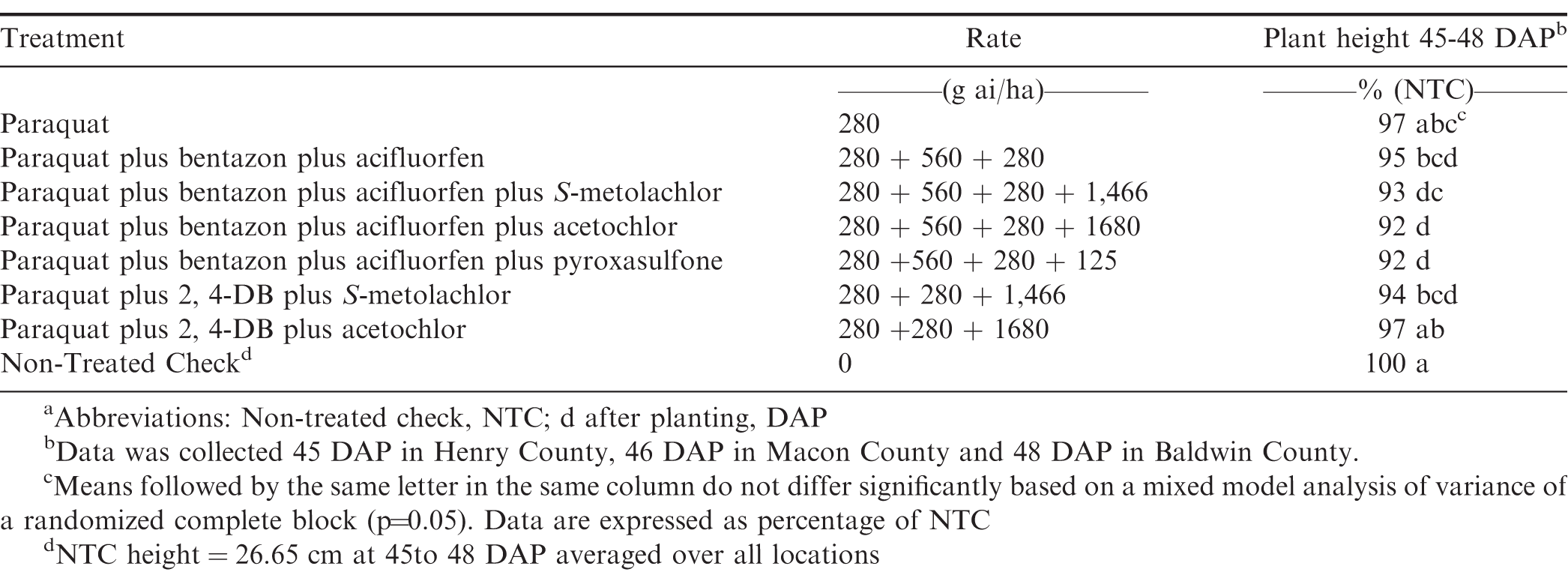
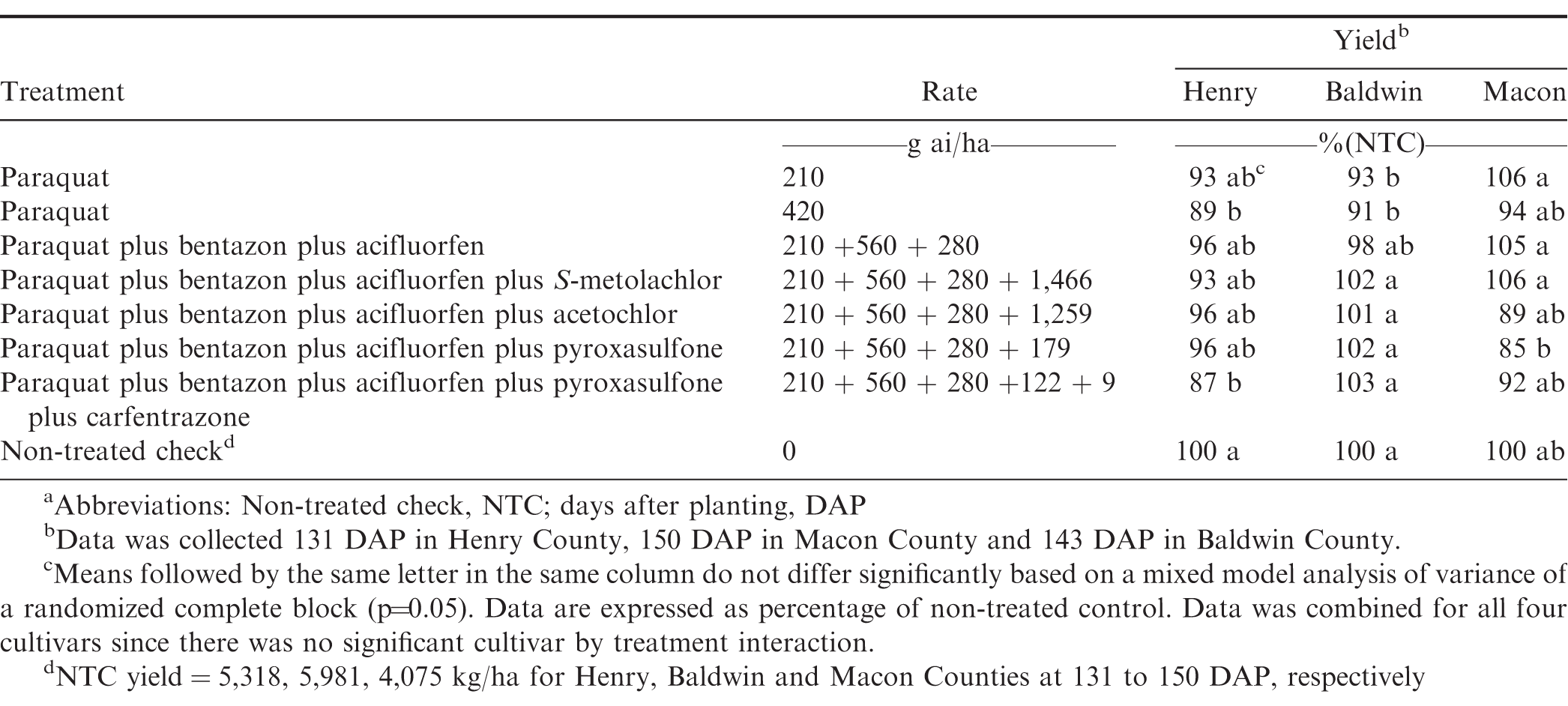
In 2016, there was a significant treatment by location interaction (P=0.03) for heights therefore, locations were analyzed separately. Paraquat 420 g ai/ha reduced plant height by 10 to 20% at 50 to 55 DAP, and 5 to 16% at 72 to 78 DAP, and it was the only treatment that reduced plant height at both timings across all locations in 2016 (Table 2). The greatest overall height reductions of 10%, 14%, 20% were observed for paraquat at 420 g ai/ha in Baldwin, Macon and Henry County, respectively at 50-55 DAP. At 72 to 78 DAP, paraquat tank mixed with bentazon plus acifluorfen was the only treatment that did not generate reduced heights compared to the NTC across all locations. In 2017, there was no location by treatment interaction (P=0.22) for peanut heights therefore, this data was pooled over all locations (Table 3). Paraquat alone at 280 g ai/ha and paraquat plus 2,4-DB plus acetochlor were the only treatments evaluated at 45 to 48 DAP which did not have height reductions compared to the NTC over all locations. Paraquat plus bentazon plus acifluorfen plus either pyroxasulfone or acetochlor had the highest height reduction of 8% over all locations in 2017. Peanuts with early season height reductions recovered and no reductions were observed when evaluated 66 to 68 DAP (data not shown).
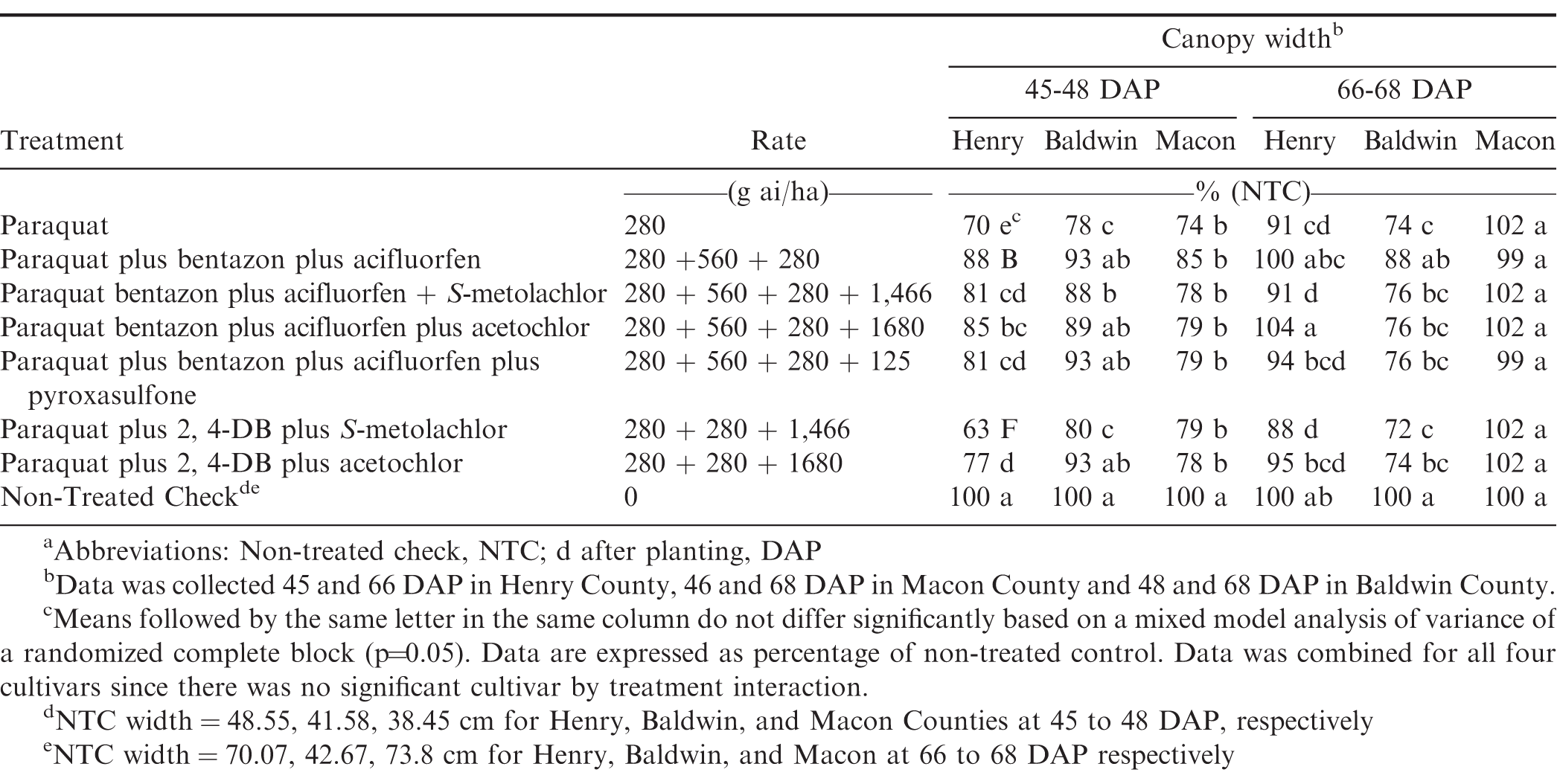
Peanut canopy widths, collected only in 2017, showed a significant treatment by location interaction (P=0.032) and were analyzed separately (Table 4). The Henry and Macon County locations showed reduced widths of 12 to 37% and 15 to 26% respectively, for all treatments evaluated 45 to 48 DAP. At the Baldwin County location, width reductions of 22%, 12%, and 20% with paraquat at 280 g ai/ha, paraquat plus bentazon plus acifluorfen plus S-metolachlor, and paraquat plus 2,4-DB plus S-metolachlor, respectively, were noted 45-48 DAP. At the Henry County location, paraquat plus 2,4-DB plus S-metolachlor showed the largest width reduction of 37% 45-48 DAP. Paraquat at 280 g ai/ha alone resulted in width reductions at the Baldwin and Macon County locations by 22% and 28%, respectively, 45 to 48 DAP.
At 66 to 68 DAP, the Macon County location no width reductions with any treatments were noted. At the Henry County location, paraquat plus bentazon plus acifluorfen, paraquat plus bentazon plus acifluorfen plus acetochlor, paraquat plus bentazon plus acifluorfen plus pyroxasulfone and paraquat plus 2,4-DB plus acetochlor all recovered from early season stunting and were no longer different from NTC. Paraquat plus 2,4-DB plus S-metolachlor showed a 12% width reduction 66 to 68 DAP while paraquat plus bentazon plus acifluorfen plus S-metolachlor showed a 9% reduction. At the Baldwin County location, more treatments had width reductions 66 to 68 DAP than 45 to 48 DAP with the exception of paraquat plus bentazon plus acifluorfen. Paraquat plus 2,4-DB plus S-metolachlor had the largest width reduction of 28% in Baldwin County at 66-68. It is likely an environmental factor, such as higher soil moisture and wetter conditions, may have prolonged herbicide injury and crop stunting at Baldwin County that did not occur in Macon and Henry County.
Yield losses were rare and did not occur over multiple locations or years. In 2016, there was a treatment by location interaction for yield (P=0.023) therefore, locations were analyzed separately (Table 5). In Henry and Baldwin County trials, paraquat at 420 g ai/ha resulted in a yield loss of 11% and 9% respectively. Paraquat at 420 g ai/ha, was included for research purpose only, but data does show that paraquat does not always have a 1.5 times safety margin; therefore, applications of higher than labeled rates will could result in height and up to 11% yield reductions as it did in 2016. Peanut producers should use caution when spraying paraquat to avoid spraying errors, miscalculations, or overlapping, as peanuts in this study showed sensitivity to paraquat over the labeled rate. In Henry County, a 13% yield loss was observed with paraquat plus bentazon plus acifluorfen plus pyroxasulfone plus carfentrazone, which also had the highest height reduction among all of the tank mixes evaluated. The Baldwin County location also had a 7% yield reduction for paraquat at 210 g ai/ha; however, this treatment did not have any height reductions at either evaluation timing. It is rare for paraquat to have caused a yield loss at a labeled rate of 210 g ai/ha as was observed in the Baldwin County trial. Other studies have not reported yield loss with labeled rate of paraquat on previous peanuts evaluated (Carley et al. 2009; Johnson et al. 1993; Wehtje et al. 1991). It is possible the herbicide application may have been applied too late in the peanut growth stage even though it was 28 DAP or an environmental factor influenced the herbicide injury in this rare case.
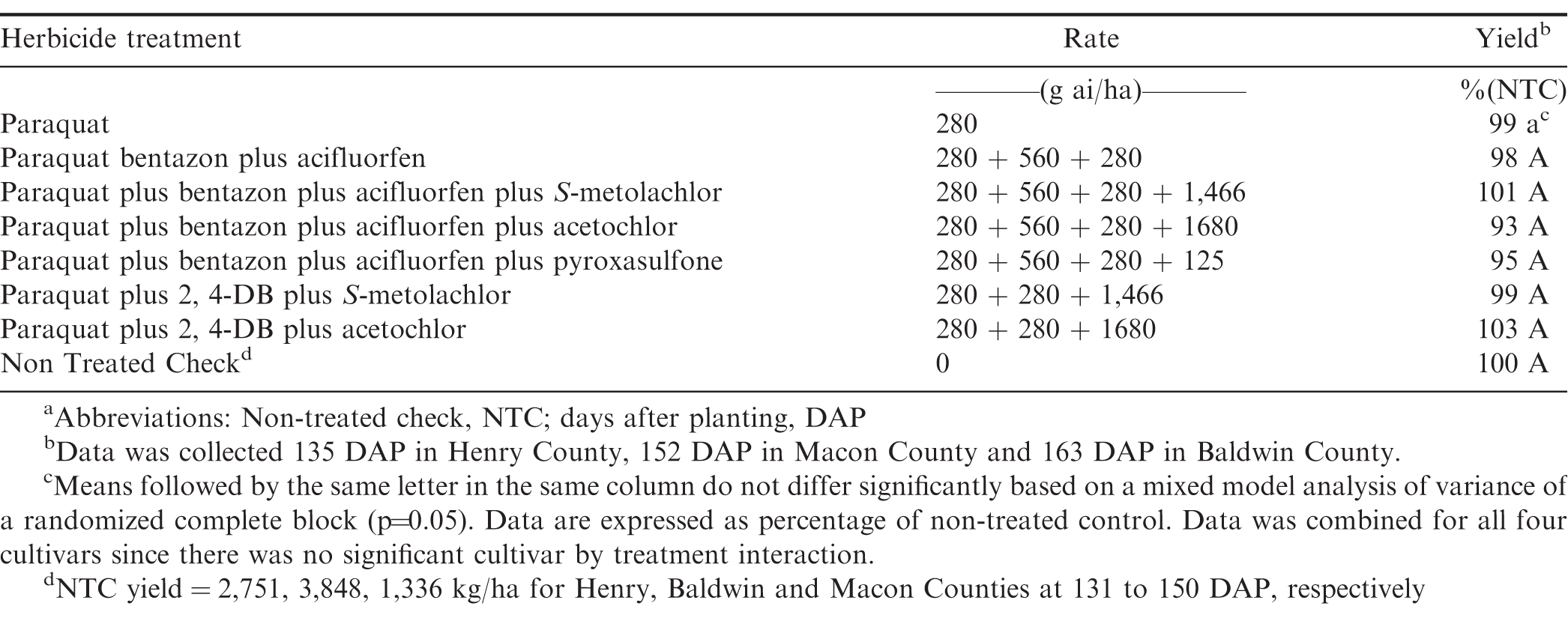
In 2017, there was no treatment by location interaction for yield (P=0.49), therefore data was analyzed together (Table 6). There were no significant yield reductions for any of the treatments evaluated in 2017. Based on these data, significant height and width reductions are not always indicative of a yield loss and peanuts can recover from initial stunting. Overall, these data and previous research indicates paraquat is safe to use on the peanuts and it is extremely rare to observe yield losses.
Treatments that included chloroacetamides were more likely to result in height and width reductions than those without. Jordan et al. (2003) observed more peanut injury with POST tank mixes of acifluorfen plus bentazon or acifluorfen plus bentazon plus 2,4-DB which included metolachlor compared to tank mixes including diclosulam, dimethenamid, and flumioxazin. This increased injury, however, did not lead to a significant yield loss (Jordan et al. 2003). Another study reported that the highest amount of stunting when S-metolachlor and paraquat were combined in a tank mix rather than either herbicide applied alone, however, this injury was only observed in one year (Grichar and Dotray 2012). Other studies have shown that chloroacetamide herbicides applied POST did not cause significant peanut injury (Grichar et al. 1996; Jordan et al. 2003). In 2017, it is possible that rainfall following application increased peanut injury when using S-metolachlor. During the first 7 d after application in 2016, Henry, Macon and Baldwin County trials had 3.5 cm, 0.03 cm, and 1 cm of rainfall respectively. In 2017, Henry, Macon and Baldwin County trials had 1.6 cm, 4.6 cm, and 7.3 cm respectively in that period. Therefore, it is likely that during our multi-location study, some field conditions, possibly rainfall, resulted in increased chloroacetamide injury and peanut stunting.
Overall, peanut in Henry County showed most sensitivity to paraquat tank mix treatments with greater height and width reductions than any other location for both years, while peanut in Baldwin County showed the most tolerance to the tank mix treatments evaluated in 2016. However, Baldwin County had more height and width reductions observed with paraquat alone than with any tank mix treatments in 2017. No treatment using labeled rates caused a significant yield loss compared to NTC over all locations in 2017. Meanwhile, for both years, we observed 10-20% less foliar injury to peanuts when bentazon was tank mixed with paraquat (data not shown). While the addition of bentazon increases broadleaf weed control and reduces injury to peanuts, it also increases herbicide cost per acre. Previous studies have shown bentazon combined with paraquat reduces peanut injury, however, this reduction in injury does not often result in a greater yield (Wehtje et al.1986; Wilcut et al. 1989). Therefore, unless a producer is trying to broaden their weed control spectrum and increase efficacy on certain weeds such as smallflower morningglory, the use of bentazon only for safening effect with paraquat may not be cost effective.
Different crop cultivar tolerance to herbicides has been noted for years. Several older peanut cultivars, such as 'Early Bunch' and 'Southern Runner', exhibited significant more sensitivity to paraquat compared to 'Florunner' (Brecke 1989). Another study showed reduced yields of 'Sunrunner', 'Southern Runner', and 'Florunner' peanut cultivars from two applications of paraquat (Knauft et al. 1990). As new cultivars are introduced herbicide programs need to be continually evaluated. In this study, 'Georgia 06G', 'Georgia 12Y', 'Georgia 14N', and 'TufRunner 511' were equivalently tolerant to labeled rates of paraquat and paraquat tank mixes evaluated. Overall, paraquat based tank mixes can be safely applied on the peanut cultivars tested in this study when using labeled rates. However, producers should expect some early season stunting, especially when tank mixing paraquat and chloroacetamides herbicides. These situations warrant further investigation to determining the environmental factors, such as rainfall, that contribute to increased chloroacetamide injury on peanuts when tank mixed with paraquat.
Acknowledgements
The authors would like to thank the National Peanut Board and Alabama Peanut Commission for funding support and Alabama Agricultural Experiment Stations for assistance with field trial management. No conflicts of interest have been declared.
Literature Cited
Anonymous 2016 Gramoxone label. Syngenta Crop Protection. Greensboro, NC. 27419 'Available at http://www.syngenta-us.com/current-label/gramoxone-sl-2.0 (verified 6 February 2016).
W.D Branch (2007). Registration of 'Georgia-06G' peanut. J. Plant Registration 1 ((2)): 120- 120.
W.D Branch (2013). Registration of 'Georgia-12Y' peanut. J. Plant Registration 7 ((2)): 151- 153.
W.D Branchand T.B Brenneman (2015). Registration of 'Gerogia-14N' peanut. J. Plant Registration 9 ((2)): 159- 161.
B.J Brecke (1989). Response of peanut cultivars to selected herbicide treatments. Proceedings Southern Weed Science Society 42: 28.
Brecke B.J and D.L Colvin 1991 Weed management in peanuts pp 239-251 'In Pimentel D 'ed CRC Handbook of Pest Management in Agriculture. Vol. 3 2nd Ed. Boca Raton: CRC Press
J Cardinaand C.W Swann (1988). Metolachlor effects on peanut growth and development. Peanut Sci 15: 57- 60.
D.S., Carley D.L Jordan, E.L Brandenburgand L.C Dharmasri (2009). Factors influencing response of virginia market type peanut (Arachis hypogae) to paraquat under weed free conditions. Peanut Sci 36: 180- 189.
W.J Gricharand P.A Dotray (2012). Peanut cultivar response to S-metolachlor and paraquat alone and in combination. Peanut Sci 39: 15- 21.
W.J., Grichar A.E Colburn, and P.A Baughman (1996). Yellow nutsedge (Cyperus esculentus) control in peanut (Arachis hypogaea) as influenced by method of metolachlor application. Weed Technol 10: 278- 281.
E.J., Hougeand G.F Warren (1970). Comparisons of dihydroxybenzenes and paraquat. Weed Sci 18 ((1)): 179- 182.
W.C., Johnson D.L Calvin, B.G. Mullinix (1993). Comparative response of three peanut cultivars to multiple herbicide applications. Peanut Sci 20: 17- 20.
Jordan D.L., G.S Chahal, S.H Lancaster, J.B Beam, A.C York, and W.N Reynolds 2011 Defining Interactions of Herbicides with Other Crop Protection Products Applied to Peanut. 'Pp 73- 93 'In S. Sololoneski S, Larramedy ML 'eds Herbicides, Theory and Applications. In Tech.
D.L., Jordan J.F Spears, and J.W Wilcut (2003). Tolerance of peanut (Arachis Hypodaea L.) to herbicides applied postemergence. Peanut Sci 30: 8- 13.
D.A., Knauft D.L Colvinand D.W Gorbet (1990). Effect of paraquat on yield and market grade of peanut (Archis hypogaea) genotypes. Weed Technol 4: 866- 870.
A Mineand S Matsunaka (1975). Mode of action of bentazon: effect on photosynthesis. Pest. Bio. Phy 5: 44- 450.
J.D Mooreand P.A Banks (1991). Interactions of foliar applied herbicides on three weed species in peanut (Arachis hypogaea). Weed Sci 39 ((4)): 614- 621.
Shaner D.L 'ed 2014 Herbicide Handbook. 10th ed. Lawrence, KS; Weed Science Society of America Pp 15, 337- 338.
B.L Tillmanand D.W Gorbet (2017). Registration for 'TUFRunner '511' peanut. J. Plant Registrations 11 ((3)): 235- 239.
G., Wehtje B Breckeand J.P Bostick (1994). Peanut tolerance to paraquat as influenced by seed size. Peanut Sci 21: 12- 16.
G., Wehtje J McGuire, R.H Walkerand M.G Patterson (1986). Texas panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat. Weed Sci 34: 308- 311.
G., Wehtje J.W Wilcut, T.V Hicks, and J McGuire (1988). Relative tolerance of peanuts to alachlor and metolachlor. Peanut Sci 15: 53- 56.
G. J.W Wehtje J.A Wilcut J McGuire (1992). Influence of bentazon on the phytotoxicity of paraquat to peanuts (Arachis hypogaea) and associated weeds. Weed Sci 40: 90- 95.
G., Wehtje J.W Wilcut, J.A McGuireand T.V Hicks (1991). Foliar penetration and phytotoxicity of paraquat as influenced by peanut cultivar. Peanut Sci 18: 67- 71.
Wilcut J.W., J.S Richburg III, E.F Eastin, G.R Wiley, F.R Walls Jr and S Newell 1994 'a Imazethapyr and paraquat systems for weed management in peanut (Arachis hypogaea) Weed Sci 42: 601- 607.
J.W Wilcutand C.W Swann (1990). Timing of paraquat applications for weed control in virginia-type peanuts (Arachis hypogaea). Weed Sci 38: 558- 562.
J.W., Wilcut G.R Wehtje, T.A Cole, T.V Hicks, and J.A McGuire (1989). Postemergence weed control systems without dinoseb for peanuts (Arachis hypogaea). Weed Sci 37: 385- 391.
J.W., Wilcut G.R. Wehtje T.V Hicks, and T.A Cole (1990). Postemergence weed management systems for peanuts. Weed Technol 4: 76- 80.
Wilcut J.W., A.C York, W.J Gricharand G.R Wehtje 1995 The biology and management of weeds in peanut (Arachis hypogaea). 'Pp 207- 244 'In Pattee HE, Stalker HT, 'eds Advances in Peanut Science Stillwater: American Peanut Research and Education Society.
Wilcut J.W., A.C York, and G.R Wehtje 1994 'b The control and interaction of weeds in peanut (Arachis hypogaea) Review Weed Sci 6: 177- 205.
E.J Williamsand S.J Drexler (1981). A non-destructive method for determining peanut pod maturity. Peanut Sci 8: 134- 141.
Notes
- Research Associate, Department of Crop Soil and Environmental Sciences, Auburn University. Auburn, AL 36849. [^]
- Extension Specialist, Assistant Professor, Department of Crop Soil and Environmental Sciences, Auburn University. Auburn, AL 36849. [^]
- Research Physiologist, USDA-ARS National Soil Dynamics Laboratory. Auburn, AL 36830. [^]
- Professor. Department of Crop Soil and Environmental Sciences, Auburn University. Auburn, AL 36849. [^]
- Professor. Department of Crop and Soil Sciences, University of Georgia. Tifton, GA. 31793. [^] *Corresponding author contact information: 201 Funchess Hall, Auburn University. Auburn. AL. 36849. steveli@auburn.edu
Author Affiliations