Introduction
Germplasm collections are valuable sources of desirable traits such as disease resistance (Hoisington et al., 1999; McCouch et al., 2013), but much work remains in characterizing the collections for useful traits. Considering the 10,065 peanut accessions currently maintained by the National Plant Germplasm System (NPGS) (USDA-ARS, 2019) and the 15,622 accessions held by the International Crops Research Institute for the Semi-Arid Tropics (ICRISAT Genebank, 2019), relatively few have been used by breeders (Isleib et al., 2001). Despite this, the impact of the few utilized plant introductions (PI) on the United States peanut industry is significant. For example, PI 203396-an accession of Brazilian origin with resistance to late leaf spot, southern blight, and tomato spotted wilt virus (TSWV)-is in the pedigrees of many cultivars including Florida-07, Georgia-12Y, Georgia Green, Southern Runner, and Tifguard (Isleib et al., 2001; Holbrook et al., 2008; Gorbet and Tillman, 2009; Branch, 2013).
For the USDA-NPGS peanut collection, disease phenotyping efforts have focused on the more manageable core (Holbrook et al., 1993) and mini-core (Holbrook and Dong, 2005) subsets. To date, all or parts of the core collection have been evaluated for resistance to late leaf spot (Anderson et al., 1993; Holbrook and Anderson, 1995), TSWV (Anderson et al., 1996), Rhizoctonia limb rot ((Franke et al., 1999), peanut root-knot nematode (Holbrook et al., 2000), Sclerotinia blight (Damicone et al., 2010; Bennett et al., 2018), pepper spot (Damicone et al., 2010), and web blotch (Damicone et al., 2010). One notable omission from this list of diseases is southern blight, caused by Athelia rolfsii (Curzi) C.C. Tu & Kimbr. (= Sclerotium rolfsii Sacc.; Xu et al., 2010). Southern blight (also called southern stem rot, stem rot, and white mold) is considered to be one the most economically damaging pathogens of peanut in the U.S. (Backman and Brenneman, 1997).
Many studies have screened peanuts in the field for resistance to A. rolfsii, but most have evaluated breeding lines and cultivars (Branch and Csinos, 1987; Shew et al., 1987; Grichar and Smith, 1992; Besler et al., 1997; Gorbet et al., 2004; Branch and Brenneman, 2009). The most comprehensive screening by far was conducted in India, where Mehan et al. (1995) evaluated 859 germplasm accessions, breeding lines, and interspecific hybrid derivatives over several years for southern blight and pod rot resistance. However, while 16 highly resistant interspecific hybrids and breeding lines were identified, this study only named a fraction of the entries that were evaluated. Bera et al. (2016) also screened 25 wild Arachis accessions and 178 F3 progenies for southern blight in India. One accession each of A. appresipila (ICG 8945) and A. pusilla (DGR 12047) and three F3 lines had less than 15% mortality when screened in pots or in the field.
The objective of the current study was to conduct field trials to evaluate a 71-accession subset of the U.S. mini-core collection (Bennett et al., 2018) to address the lack of information regarding sources of resistance to A. rolfsii in the U.S. germplasm collection. In the second and third years of evaluation, disease ratings were taken for web blotch caused by Didymella arachidicola (Khokhr.) Tomilin (=Phoma arachidicola Marasas, Pauer & Boerema; Chen et al., 2015), and pod rot ratings were also taken in the last year of the study.
Materials and Methods
Seventy-one accessions of the U.S. peanut mini-core collection were evaluated at Oklahoma State University's Caddo Research Station at Fort Cobb in 2016 to 2018. The A. rolfsii-susceptible, runner cultivar Georgia-06G (Branch, 2007), rated 20 by Peanut Rx (Kemerait et al., 2018) was used as a positive control. Runner cultivars Georgia-03L (Branch, 2004) and Georgia-07W (Branch and Brenneman, 2008), rated 10 and 15, respectively by Peanut Rx, served as resistant controls.
Field plots were in either Pond Creek fine sandy loam (fine-silty, mixed, superactive, thermic Pachic Argiustolls) or Binger fine sandy loam (fine-loamy, mixed, active, thermic Udic Rhodustalfs). A randomized complete block design with three replications (blocks) were used, and individual plots consisted of two 4.6-m-long rows with 0.9-m beds. Seeds were planted at a density of five per 0.3 m. Plots were planted on 12 May 2016, 3 June 2017, and 18 May 2018. Herbicide and fertilizer treatments were applied following Extension recommendations. Applications of azoxystrobin, chlorothalonil, chlorothalonil and difenoconazole, cyproconazole, and fluazinam were applied during the three seasons to manage other diseases such as leaf spots and Sclerotinia blight. Plots were dug 142 d after planting in 2016 (30 September), 139 d after planting in 2017 (19 October), and 152 d after planting in 2018 (16 October). Air temperature and rainfall were recorded by the Oklahoma Mesonet weather station at the Oklahoma Agricultural Experiment Station Caddo Research Station in Fort Cobb, OK (McPherson et al., 2007). The field plots were within 290 m of the weather station.
Plots were inoculated with A. rolfsii on 8 August 2016, 7 July 2017, and 17 July 2018. In 2016, 35 ml of oat groats colonized by A. rolfsii isolate Ar-15-1 were distributed along the interior of the plant canopy of each row. The oat inoculum was produced by soaking 600 ml of oat groats in 350 ml of reverse osmosis water in a stainless-steel tray (42 cm-long x 25 cm-wide x 6.4 cm-deep) for 4 hours. The tray was then covered with two layers of aluminium foil before autoclaving for 60 min at 121 C and 172 kPa on two consecutive days. After cooling to room temperature, the sterilized oats were inoculated with eight, 7.2-mm-diam. mycelial plugs, taken from the colony margins of a 3-day-old culture grown on potato dextrose agar. The inoculated oats were incubated at 27 C for 10 days, after which the grains were colonized by A. rolfsii. The trays were placed in a greenhouse set at approximately 29 C, the foil cover was removed, and the colonized oats were stirred daily until completely dry. In 2017 and 2018, 0.15 g of sclerotia (approximately 225 individual sclerotia) mixed with 0.15 g of sand to serve as a carrier were used to inoculate plots instead of colonized oats. Sclerotia were produced on peanut leaves and stems of U.S. peanut core collection accessions CC38 (PI 493581), CC41 (PI 493631), CC68 (PI 493880), CC384 (PI 155107), and CC787 (PI 429420). These accessions were chosen because they were found to be highly susceptible to A. rolfsii in growth chamber inoculation experiments (Bennett, in press). Half-size flatware racks (50.2-cm long x 25.4-cm wide x 10.2-cm deep, Cambro HFR258151, WebstaurantStore, Lancaster, PA) were lined with a damp cotton towel. Five mycelial plugs, produced as for the oat inoculum, were each placed on moist cosmetic cotton rounds along the middle of the racks. Eight-week-old plants, grown in 11-cm-diam. pots (ca. 600 ml), were cut at the soil line. Stems and leaves were cut into approximately 50-cm segments and were placed on the flatware rack, covering the mycelial plugs. Trays were placed in a growth chamber (PGR15; Controlled Environments Ltd., Winnipeg, MB) set at 28.5 C. Reverse osmosis water was applied to the trays for 5 sec, every 5 min, at a flow rate of 160 cm3 min-1 with misting system (Orbit Irrigation, North Salt Lake City, UT). After 1 or 2 weeks, when the peanut vegetation was covered with A. rolfsii mycelium, the mister was turned off. Numerous sclerotia formed as the plant material and towels slowly dried over 2 to 5 days. Sclerotia were separated from the dried vegetation by hand.
Southern blight disease evaluations were taken twice each year on the following dates: 22 August and 19 September in 2016, 11 and 28 September in 2017, and 24 August and 18 September in 2018. Disease incidence was measured by counting the number of 6-inch sections within each plot that had symptoms of southern blight. More disease was observed each year on the second disease rating, so only last rating date was analyzed. Incidence of web blotch was taken on 28 September 2017 and on 10 October 2018 by visually estimating the percentage of leaflets in each plot with symptoms. In 2017, plots with approximately an incidence of 5% or less of web blotch were recorded as 0%. In 2018, web blotch severity, or the percentage of leaf area in remaining leaves with symptoms, and percentage of defoliation were also estimated. In addition, plots were rated for pod rot in 2018 by estimating the percentage of pods with symptoms of pod rot within three hours after digging.
Data were analyzed using one-way ANOVA in PROC GLIMMIX of SAS Version 9.4 (SAS Institute, Cary, NC) using a split plot design, with year as the whole plot and entry as the subplot. Replication (block) was included as a random factor. The SLICE option of the LSMEANS statement was used to examine differences within entries among years. The Type I error rate for pairwise comparisons of breeding lines and cultivars was controlled at 𝜶 = 0.05 using the ADJUST=TUKEY option. Correlation between years for incidence of southern blight and web blotch, as well as between web blotch incidence, severity, and defoliation, were conducted using PROC CORR.
Results and Discussion
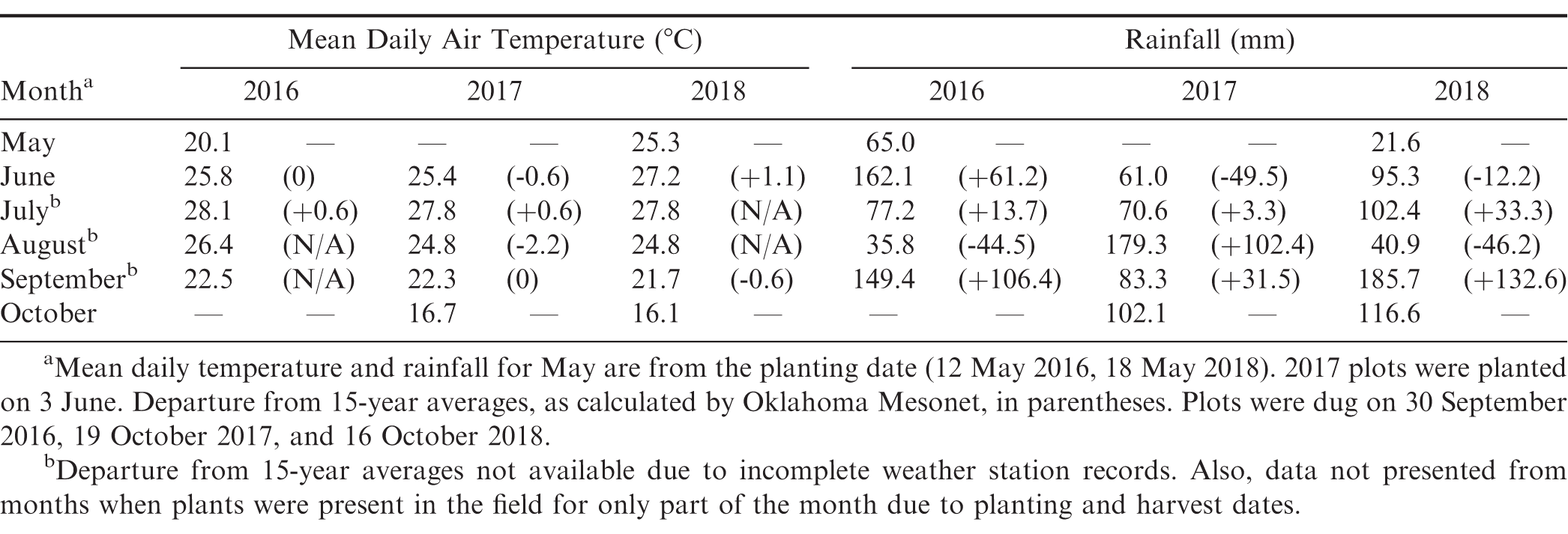
Over the duration of the experiment, temperatures were generally close to the 15-year average for Fort Cobb except for August 2017 when it was 2.2 C cooler (Table 1). For rainfall, the most notable deviations from 15-year averages were September 2016 and 2018, and August 2017. In these months, two to three times the 15-year average rainfall occurred (Table 1).
Monthly air temperature and rainfall in 2016-2018 at the Caddo Research Station, Fort Cobb, OK, from Oklahoma Mesonet (McPherson et al., 2007).
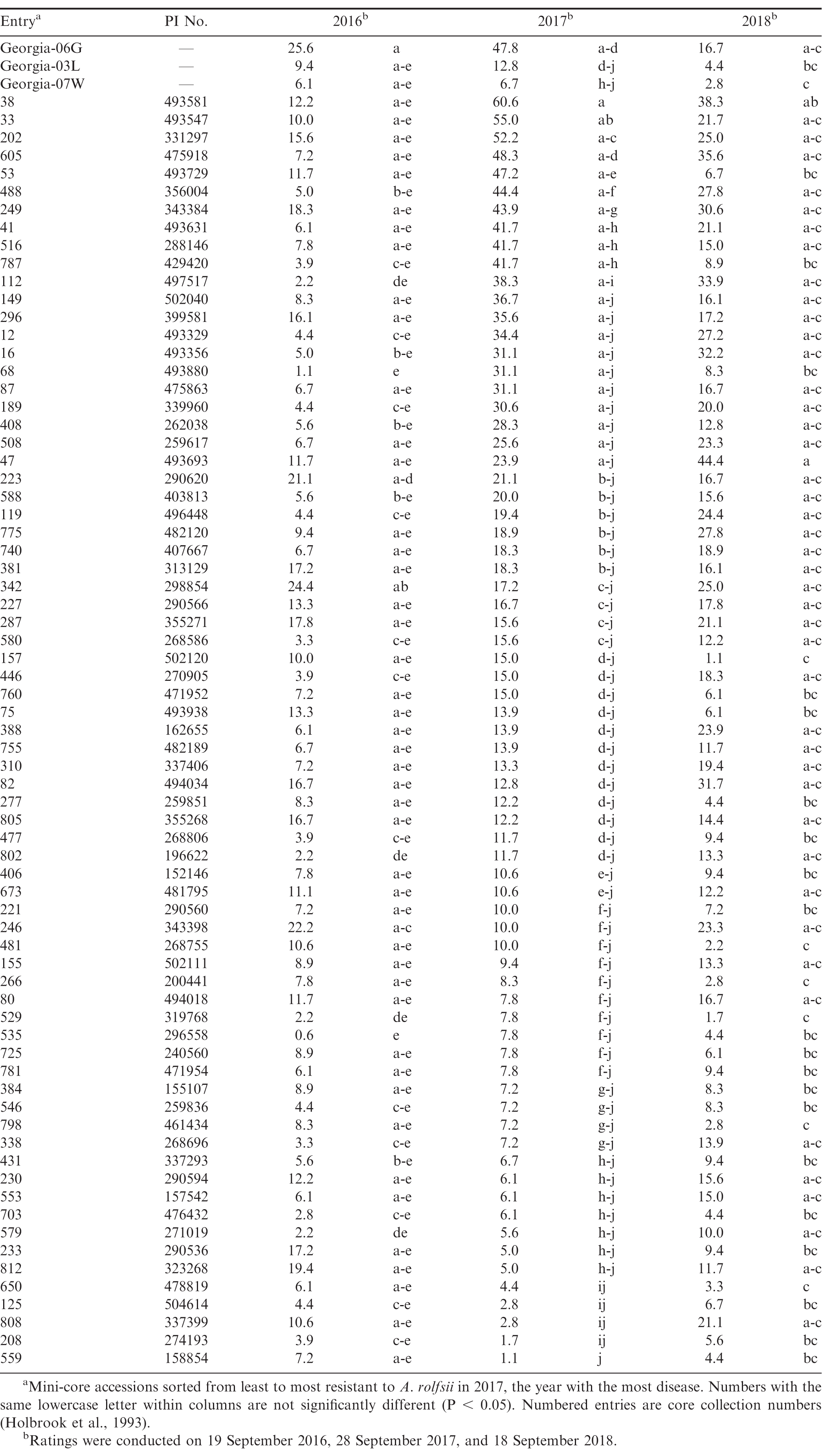
In general, moderate to low levels of southern blight were observed in this experiment relative to similar experiments conducted in the Southeast, where greater than 50% disease incidence has been observed in Georgia-06G over multiple years (Branch and Brenneman, 2009). In this experiment, southern blight was most severe in 2017, with nearly 50% disease incidence in Georgia-06G, but significantly less disease was observed in 2016 and 2018 (Table 2). The above-average rainfall received in August 2017 likely resulted in conditions more favourable for A. rolfsii. Analysis of southern blight incidence indicated significant effects of entry (F = 6.99; df = 73, 438; P < 0.01) and year (F = 34.84; df = 2, 4; P < 0.01), as well as a significant entry*year interaction (F = 3.03; df = 146, 438; P < 0.01). Entries differed in incidence of southern blight in 2016 (F = 3.08; df = 73, 146; P < 0.01), 2017 (F = 6.02; df = 73, 146; P < 0.01), and 2018 (F = 2.85; df = 73, 146; P < 0.01). In 2017, the best year for disease, there were three entries with over 50% disease incidence, 17 entries with 25 to 48%, 28 entries with 10 to 24%, 18 entries with 5 to 9%, and five entries with less than 5% disease incidence. As indicated by the significant entry*year interaction, disease incidence within entries varied with year, and results from 2017 and 2018 had the best correlation among years (r = 0.56, P < 0.01). CC38, CC33, and CC202, the three most susceptible entries in 2017, were among the top third and quarter most diseased entries in 2016 and 2018, respectively. Similarly, the five of the six most resistant entries in 2017-CC559, CC208, CC125, and CC650-were among the bottom quarter of least diseased entries in 2017. The resistant cultivar Georgia-07W was among the more resistant entries, and Georgia-03L had numerically, albeit not statistically, more disease than Georgia-07W in 2017 and 2018. A couple entries were notably inconsistent among years. CC808, which was among the most resistant entries in 2017 (4%), had moderate disease in 2016 and 2018 (11% and 21%, respectively). In addition, CC47 had the most southern blight of any entry in 2018 (44%) but had intermediate levels of disease in 2017 (24%). Finally, CC157 (2%) and CC481 (1%) were among the most resistant entries in 2018 but were only moderately resistant in 2017 (15% and 10%, respectively). Possible explanations for inconsistency between years could be genetically mixed entries, since these accessions were from the non-purified mini-core (Chen et al., 2014), in addition to uneven distribution of inoculum despite manual application of sclerotia.
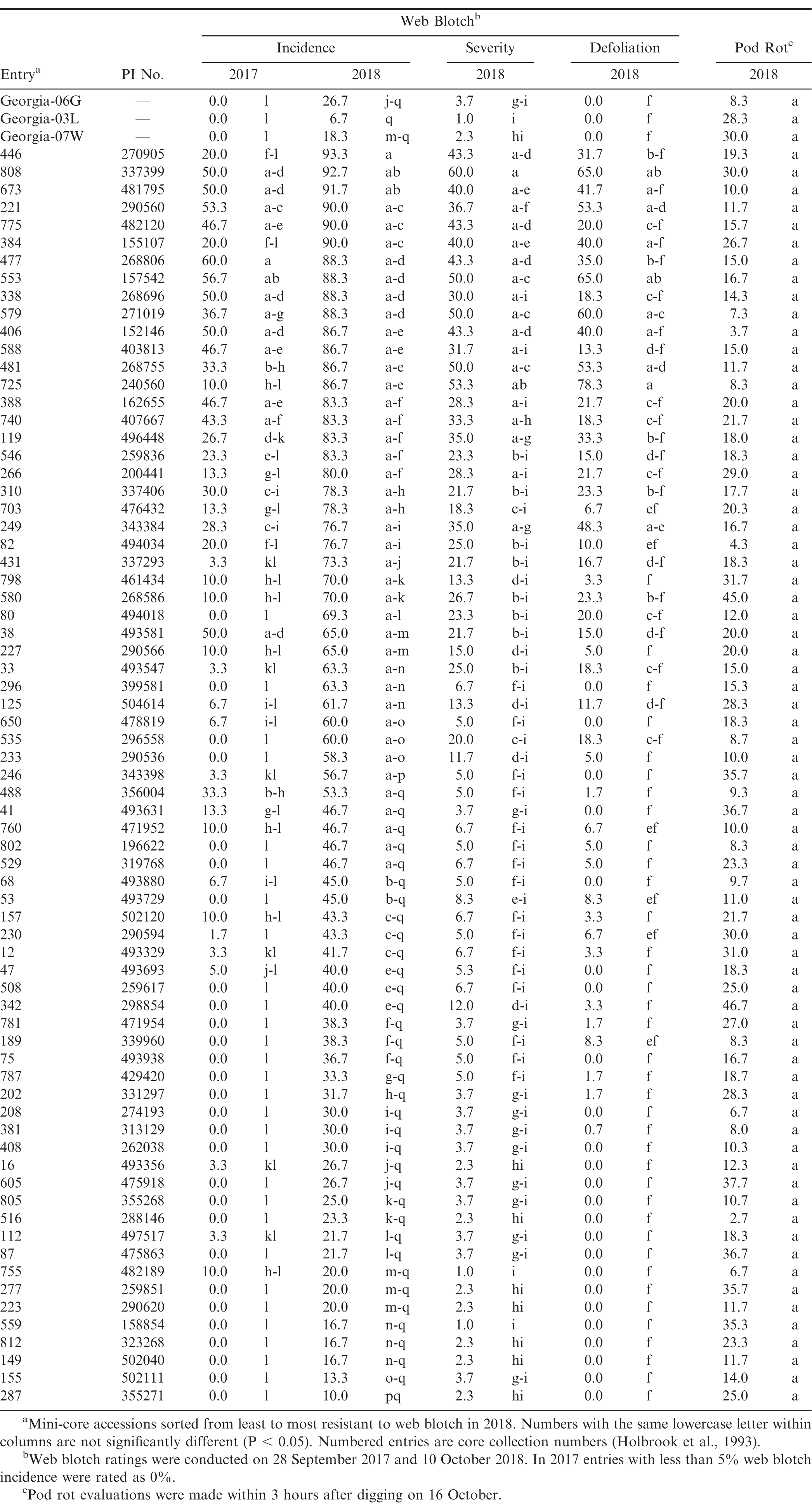
Little web blotch was observed in 2016 (data not shown), but 2017 had moderate levels of disease and considerable disease was observed in 2018 (Table 3). Incidence of web blotch was greatest in 2018, perhaps because disease evaluations were taken nearly two weeks later than in 2017. In the combined year analysis, significant effects were observed for year (F = 258.93; df = 1, 4; P < 0.01) and entry (F = 22.66; df = 73, 292; P < 0.01), as well as for the year*entry interaction (F = 3.73; df = 73, 292; P < 0.01). Despite the significant interaction, which indicated that web blotch incidence within entries depended upon year, there was a correlation between the two years in disease incidence (r = 0.76, P < 0.01). Statistical differences among entries in disease incidence were also observed within each year (F = 20.96 in 2017 and 11.14 in 2018; df = 73, 146; P < 0.01; Table 3). The Georgia cultivars were among the most resistant entries in both years and had low estimates for severity and defoliation. In 2018, incidence of web blotch greater than 75% was observed in 23 entries, 50-73% in 14 entries, 27-47% in 22 entries, and 25% or less in 12 entries (Table 3). Five entries-CC287, CC155, CC149, CC812, and CC559-had between 10% and 20% disease. The 23 most susceptible entries in 2018 were also among the most diseased in 2017. In 2018, there were significant differences among entries in web blotch severity (F = 9.52; df = 73, 146; P < 0.01) and defoliation (F = 7.36; df = 73, 146; P < 0.01). Strong correlations were also observed between disease incidence and severity (r = 0.89, P < 0.01), disease incidence and defoliation (r = 0.76, P < 0.01), and severity and defoliation (r = 0.93, P < 0.01). Three entries-CC33, CC208, and CC481-were also evaluated for web blotch by Damicone et al. at Fort Cobb in 2003 (2010). In that study, moderate to low levels of web blotch were observed, with the most susceptible entry, CC357, having 37.5% disease incidence. In 2003, CC33 and CC208 had 6.2% web blotch and CC481 had 30%, similar to what was observed in this study in 2017. While leaves with web blotch symptoms are not uncommon, severe outbreaks of web blotch do not occur regularly at Fort Cobb. Web blotch has been considered a minor disease in Oklahoma (Damicone et al., 2010).
Moderate levels of pod rot were observed in 2016, but disease ratings were not taken. Ratings were not taken in 2017 because the entire 2017 experiment was left in the ground in an attempt to increase soilborne inoculum for other pod rot experiments. Moderate levels of pod rot were observed in 2018, but differences among entries were not statistically significant after correcting for Type I error rate resulting from multiple comparisons (Table 3). There were eight entries with between 35% to 47% pod rot, 19 entries between 20% and 32%, 17 entries between 11% and 20%, and 18 entries with less than 11%. Among the cultivars, Georgia-06G had the least pod rot at 8% and Georgia-03L and Georgia-07W had 28% and 30%, respectively. More years of data are needed to definitively identify resistant accessions in the mini-core, but the data presented here provide a starting point. The need for host resistance to Pythium pod rot has increased with the European Union recently reducing the maximum residue limit for phosphite fungicides to 2 mg/kg (European Union Pesticides Database - European Commission, 2019).
The high levels of southern blight reported in Georgia (Branch and Brenneman, 2009, 2015) were not obtained in this study. However, these results identify A. rolfsii-susceptible accessions within the U.S. mini-core, in addition to promising resistant candidates for further testing in environments more conducive to southern blight. Of the four most resistant accessions identified in this study (CC559, CC208, CC125, and CC650), CC125 and CC650 are also highly resistant to Sclerotinia blight (Bennett et al., 2018). CC650 has also demonstrated high levels of resistance to A. rolfsii in growth chamber assays (Bennett, in press.) The current study identified susceptible and resistant accessions to web blotch. Web blotch first appeared in the U.S. in southern Texas in 1972 and has since spread to other peanut production regions throughout the country (Taber et al., 1984). Web blotch has been reported from most other peanut-producing countries and is one of the important diseases of peanut in southern Africa (Cole and Liddell, 1997). The study by Damicone et al. (2010), which evaluated 62 accessions from the core collection, and this study are the only reports to date regarding web blotch resistance in the U.S. germplasm collection. Peanut breeders may find these disease-resistant accessions useful for introducing new sources of resistance to A. rolfsii and web blotch into their breeding programs.
Acknowledgements
Angie Harting and Lisa Myers at USDA-ARS, and Robert Weidenmaier, Harley Houston, and Brennan Leighton at the OAES Caddo Research Station provided technical assistance. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the USDA. The USDA is an equal opportunity provider and employer.
Literature Cited
W.F., Anderson, C.C Holbrook, and T.B Brenneman (1993). Resistance to Cercosporidium personatum within peanut germplasm. Peanut Sci 20: 53- 57.
W.F., Anderson, C.C Holbrook, and A.K Culbreath (1996). Screening the peanut core collection for resistance to tomato spotted wilt virus. Peanut Sci 23: 57- 61.
Backman, P.A., and T.B Brenneman 1997 Stem rot. Pages 36- 37 In Compendium of Peanut Diseases. 2nd ed American Phytopathological Society, St. Paul, Minnesota.
Bennett, R.S 2020 Growth chamber assay for evaluating resistance to Athelia rolfsii, Peanut Sci. 47:(in press).
R.S., Bennett, K.D Chamberlin, and J.P Damicone (2018). Sclerotinia blight resistance in the US peanut mini-core collection. Crop Sci 58: 1306- 1317.
S.K., Bera, J.H Kamdar, S.V Kasundra, and P.P Thirumalaisami (2016). Identification of groundnut genotypes and wild species resistant to stem rot using an efficient field screening technique. Electron. J. Plant Breed 7: 61- 70.
B.A., Besler, W.J Grichar, and O.D Smith (1997). Reaction of selected peanut varieties and breeding lines to southern stem rot. Peanut Sci 24: 6- 9.
W.D Branch, (2004). Registration of 'Georgia-03L' peanut. Crop Sci 44: 1485- 1486.
W.D Branch, (2007). Registration of 'Georgia-06G' peanut. J. Plant Regist 1: 120.
W.D Branch, (2013). Registration of 'Georgia-12Y' peanut. J. Plant Regist 7: 151- 153.
W.D., Branch, and T.B Brenneman (2008). Registration of 'Georgia-07W' peanut. J. Plant Regist 2: 88- 91.
W.D., Branch, and T.B Brenneman (2009). Field evaluation for the combination of white mould and tomato spotted wilt disease resistance among peanut genotypes. Crop Prot 28: 595- 598.
W.D., Branch, and T.B Brenneman (2015). Stem rot (white mold) and tomato spotted wilt resistance among peanut genotypes. Peanut Sci 42: 18- 22.
W.D., Branch, and A.S Csinos (1987). Evaluation of peanut cultivars for resistance to field infection by Sclerotium rolfsii. Plant Dis 71: 268- 270.
C.Y., Chen, N.A Barkley, M.L Wang, C.C Holbrook, and P.M Dang (2014). Registration of purified accessions for the U.S. peanut mini-core germplasm collection. J. Plant Regist 8: 77- 85.
Q., Chen, J.R Jiang, G.Z Zhang, L Cai, and P.W Crous (2015). Resolving the Phoma enigma. Stud. Mycol 82: 137- 217.
Cole, D.L., and C Liddell 1997 Web blotch, Pages 39- 40 In Compendium of Peanut Diseases. 2nd ed American Phytopathological Society, St. Paul, Minnesota.
J.P., Damicone, C.C Holbrook, D.L Smith, H.A Melouk, and K.D Chamberlin (2010). Reaction of the core collection of peanut germplasm to Sclerotinia blight and pepper spot. Peanut Sci 37: 1- 11.
EU Pesticides database - European Commission 2019 https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/?event=activesubstance.detail&language=EN&selectedID=1747 (accessed 17 September 2019)
M.D., Franke, T.B Brenneman, and C.C Holbrook (1999). Identification of resistance to Rhizoctonia limb rot in a core collection of peanut germ plasm. Plant Dis 83: 944- 948.
D.W., Gorbet, T.A Kucharek, F.M Shokes, and T.B Brenneman (2004). Field evaluations of peanut germplasm for resistance to stem rot caused by Sclerotium rolfsii. Peanut Sci 31: 91- 95.
D.W., Gorbet, and B.L Tillman (2009). Registration of 'Florida-07' peanut. J. Plant Regist 3: 14- 18.
W.J., Grichar, and O.D Smith (1992). Variation in yield and resistance to southern stem rot among peanut (Arachis hypogaea L.) lines selected for Pythium pod rot resistance. Peanut Sci 19: 55- 58.
D., Hoisington, M Khairallah, T Reeves, J.-M Ribaut, B Skovmand, et al (1999). Plant genetic resources: What can they contribute toward increased crop productivity?. Proc. Natl. Acad. Sci 96: 5937- 5943.
C.C., Holbrook, and W.F Anderson (1995). Evaluation of a core collection to identify resistance to late leafspot in peanut. Crop Sci 35: 1700- 1702.
C.C., Holbrook, W.F Anderson, and R.N Pittman (1993). Selection of a core collection from the U.S. germplasm collection of peanut. Crop Sci 33: 859- 861.
C.C., Holbrook, and W Dong (2005). Development and evaluation of a mini core collection for the U.S. peanut germplasm collection. Crop Sci 45: 1540- 1544.
C.C., Holbrook, P Timper, A.K Culbreath, and C.K Kvien (2008). Registration of 'Tifguard' peanut. J. Plant Regist 2: 92- 94.
C.C., Holbrook, P Timper, and H.Q Xue (2000). Evaluation of the core collection approach for identifying resistance to Meloidogyne arenaria in peanut. Crop Sci 40: 1172.
ICRISAT Genebank 2019 http://genebank.icrisat.org/IND/Passport?Crop=Groundnut (accessed 13 September 2019) .
T.G., Isleib, C.C Holbrook, and D.W Gorbet (2001). Use of plant introductions in peanut cultivar development. Peanut Sci 28: 96- 113.
Kemerait, R., A Culbreath, E Prostko, T Brenneman, S Tubbs, R Srinivasan, M Abney, S Monfort, A Rabinowitz, B Tillman, N Dufault, D Rowland, M Mulvaney, I Small, A Hagan, J Sarver, D Anco, and N Smith 2018 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2018 version of the peanut disease risk index Univ. of Georgia Coop. Ext. Serv, Athens, GA.
S., McCouch, G.J Baute, J Bradeen, P Bramel, P.K Bretting, et al (2013). Agriculture: feeding the future. Nature 499: 23- 24.
R.A., McPherson, C.A Fiebrich, K.C Crawford, J.R Kilby, D.L Grimsley, J.E Martinez, J.B Basara, B.G Illston, D.A Morris, K.A Kloesel, A.D Melvin, H Shrivastava, J.M Wolfinbarger, J.P Bostic, and D.B Demko (2007). Statewide monitoring of the mesoscale environment: a technical update on the Oklahoma Mesonet. J. Atmospheric Ocean. Technol 24: 301- 321.
V.K., Mehan, C.D Mayee, D McDonald, N Ramakrishna, and S Jayanthi (1995). Resistance in groundnut to Sclerotium rolfsii-caused stem and pod rot. Int. J. Pest Manag 41: 79- 83.
B.B., Shew, J.C Wynne, and M.K Beute (1987). Field, microplot, and greenhouse evaluations of resistance to Sclerotium rolfsii in peanut. Plant Dis 71: 188- 191.
R.A., Taber, R.E Pettit, and G.L Philley (1984). Peanut web blotch: I. Cultural characteristics and identity of causal fungus. Peanut Sci 11: 109- 114.
USDA-ARS Germplasm Resources Information Network (GRIN) database 2019 Natl. Germplasm Resour. Lab., Beltsville, MD http://www.ars-grin.gov/npgs/acc/acc_queries.html (accessed 13 Sep 2019)
Z., Xu, T.C Harrington, M.L Gleason, and J.C Batzer (2010). Phylogenetic placement of plant pathogenic Sclerotium species among teleomorph genera. Mycologia 102: 337- 346.
Notes
- Research Plant Pathologist and Research Biologist, USDA-ARS, Wheat, Peanuts and Other Field Crops Research Unit, Stillwater, OK 74075. [^] *Corresponding author's E-mail: rebecca.bennett@usda.gov
Author Affiliations