Introduction
The effect of drought stress in the cultivated peanut (Arachis hypogaea L.) has long been shown to be dependent upon different stages of development during the growing season (Klepper, 1973; Boote et al, 1982; and Williams and Boote, 1995). For example, the 'Florunner' cultivar (Norden et al., 1969) is a medium-maturity runner-type that typically matures at approximately 140 d after planting (DAP) in south Georgia, and it was the predominant peanut cultivar grown in the southeastern U.S. for two-decades (mid-1970 to mid-1990). Drought stress had the least yield and grade effect from 106 to 140 DAP, greatest at 71 to 105 DAP, and intermediate for 36 to 70 DAP in the Florunner cultivar (Stansell and Pallas, 1985). However, the highest pod yield combined across years was found to be the full-season irrigation treatment without any drought stress as might be expected.
The detrimental effects of the 1980 drought resulted in the Georgia peanut breeding programs including drought tolerance as an objective in the development of new cultivars (Branch and Kvien, 1992). Recently, field and greenhouse screening of several peanut genotypes have resulted in naturally and artificially drought-induced small as well as large (normal) phenotypes within advanced breeding lines and released cultivars. Both plant-types with apparent drought tolerance during early to mid-season development were selected within the pure-line runner-type peanut cultivar, 'Georgia-10T' (Branch and Culbreath, 2011) for subsequent evaluation.
The objective of this study was to field evaluate these small and large drought tolerant plants for several agronomic traits. A greenhouse study was also conducted to explain the possible cause for these small-plant phenotypes.
Materials and Methods
Several different genotypes have been screened in the field and greenhouse for drought tolerance, and most have resulted in both small and normal plant types. During the 2011 growing season at Tifton and Plains, GA, there was an early-season drought stress period during May and June of 1.5 and 61.5 mm vs. long-term average of 82.8 vs 116.6 mm of rainfall at Tifton and 1.0 and 33.0 mm vs. long-term average of 78.7 and 116.1 mm of rainfall at Plains, respectively (Branch and Fletcher, 2017). A few drought-tolerant plants were identified and tagged which appeared green and turgid amongst otherwise dry and severely-wilted plants within the Georgia-10T runner-type peanut cultivar (Fig. 1). Peanut pod and seeds were harvested from these drought-tolerant individual plant selections (IPS) for increase and testing.
During the autumn and winter of 2014 to 2015, a greenhouse drought study was utilized to test and compare the IPS's to the parental check cultivar, Georgia-10T. Ninety-six 56.8 L heavy-duty rubber tubs were used with several small (ca. 3 to 4 mm) holes drilled in the bottom and concaved serrated metal disks covered with cheesecloth were placed face-down inside each tub to allow for drainage without soil loss. The growth medium was a 1:1 mixture of field soil and germination mixture, obtained earlier in the spring, and watered before tubs were sealed with clear plastic for solar-sterilization during the hot summer months. One hundred seed per IPS and parent were taken to the Georgia Department of Agriculture, State Seed Lab in Tifton, GA. Ten (5 d old) pre-germinated seedlings were then transplanted into the soil containing tubs in the greenhouse. All seedlings were watered and fertilized to obtain maximum growth and development between transplanting and initiation of drought stress periods. A few green and turgid plants were again found within the check cultivar after exposing the plants to an early-season drought stress period for two weeks at 60 and 90 d after planting (Fig. 2). Seed from IPS's of both naturally occurring and artificially drought-induced phenotypes produced similar normal and small-plants. Several individual plant selections were made within this true-breeding cultivar for comparison under three preliminary yield tests (PYT) during 2017 (PYT1 = mid-April planting date, PYT2 = mid-May planting date with irrigation and recommended input production practices; and PYT3 = mid-May planting date with reduced-input production practices without irrigation).
The PYT1, PYT2, and PYT3 were conducted on a Tifton loamy sand soil type (fine-loamy, siliceous, thermic Plinthic Kandiudult) at the University of Georgia Gibbs Research Farm (latitude: 31.43 N and longitude: -83.59 W) near Tifton, GA. Plots consisted of two-rows spaced 1.8 m apart by 6.1 m long. Field tests were planted 13 April, 16 May, and 12 May, 2017 for the mid-April and mid-May recommended-input irrigated PYT, and mid-May for the reduced-input dryland PYT, respectively. Each PYT were in a three-yr crop rotation following corn (Zea mays L.) and cotton (Gossypium hirsutum L.). Individual entries were dug near optimum maturity based upon the hull-scrape method determined from adjacent border rows (Williams and Drexler, 1981).
After digging with a two-row digger and picked with a small-plot thresher, pods were dried with forced warm air to 6% moisture. Pod samples were then hand-cleaned over a screen table before weighing for yield determination. Total sound mature kernels (TSMK) grade percentages were determined according to federal-state inspection service procedures for runner-type peanut (USDA-AMS, 1998). One hundred pod and sound mature seed weight were also counted and weighed for each genotype.
The experimental design was a randomized complete block with six replications. Data from each individual test was subjected to analyses of variance. Waller-Duncan's t-Test (k-ratio = 100) was used for mean separation of significant differences (P ≤ 0.05).
A greenhouse experiment was conducted in the autumn and winter (2017 to 2018) to evaluate the effect of a gibberellic acid (GA3) solution of 35.6 ppm (0.13g GA3/3.8 L), 1.3% 2-propanol, 0.1% nonionic surfactant, and 98.6% water on young seedlings from small plants originating from natural (GA 113385-1-2-2) and artificial (GA 10T-1-1) selections of Georgia-10T. Four seedlings per treatment and control (untreated) with three replications were used for evaluation of GA3 effect. The treated plants were sprayed by hand each morning with GA3 solution. Spray treatment started at 14 d after planting (DAP) and continued to 60 DAP.
Main stem height (MS), cotyledonary lateral (CL) length, and main stem internode length (IL) were measured during this GA3 study. Least significant difference (LSD) t-Test was used to compare treated plants versus the small plant controls at P ≤ 0.05.
During 2018, seed from both unsprayed control and GA3 spray treated small plants were space-planted approximately 122 cm apart in one-row field plots. The experimental design had five seed per replication for treatment and control, in a randomized complete block with four field replications were used to evaluate the effect of GA3 on the next generation. Measurements at harvest included main stem height, maximum diameter of plants, leaflet length and width, number and weight of peanut pods and seed.
Results and Discussion
Field Evaluations
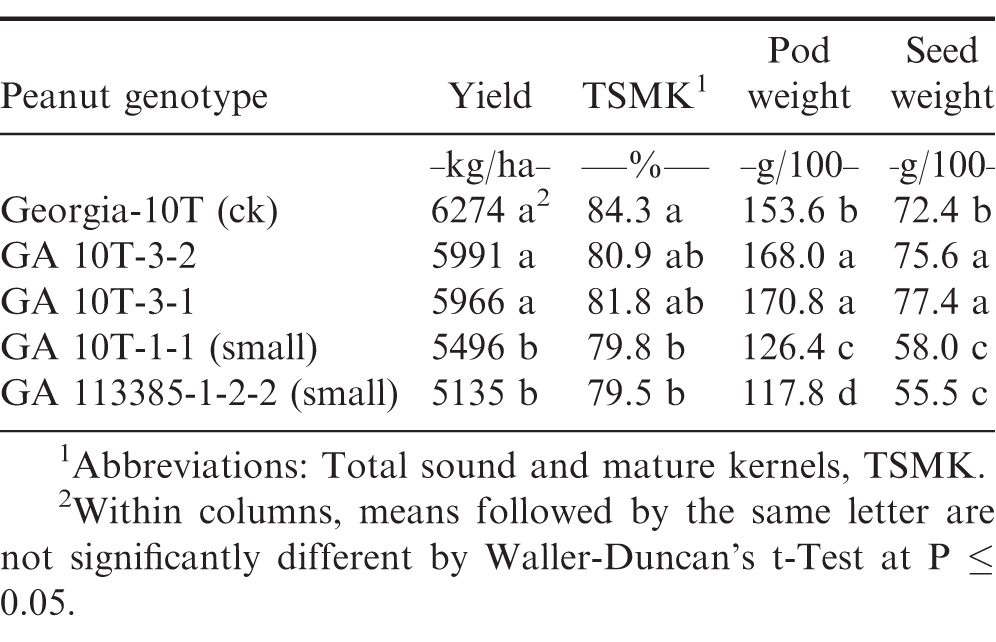
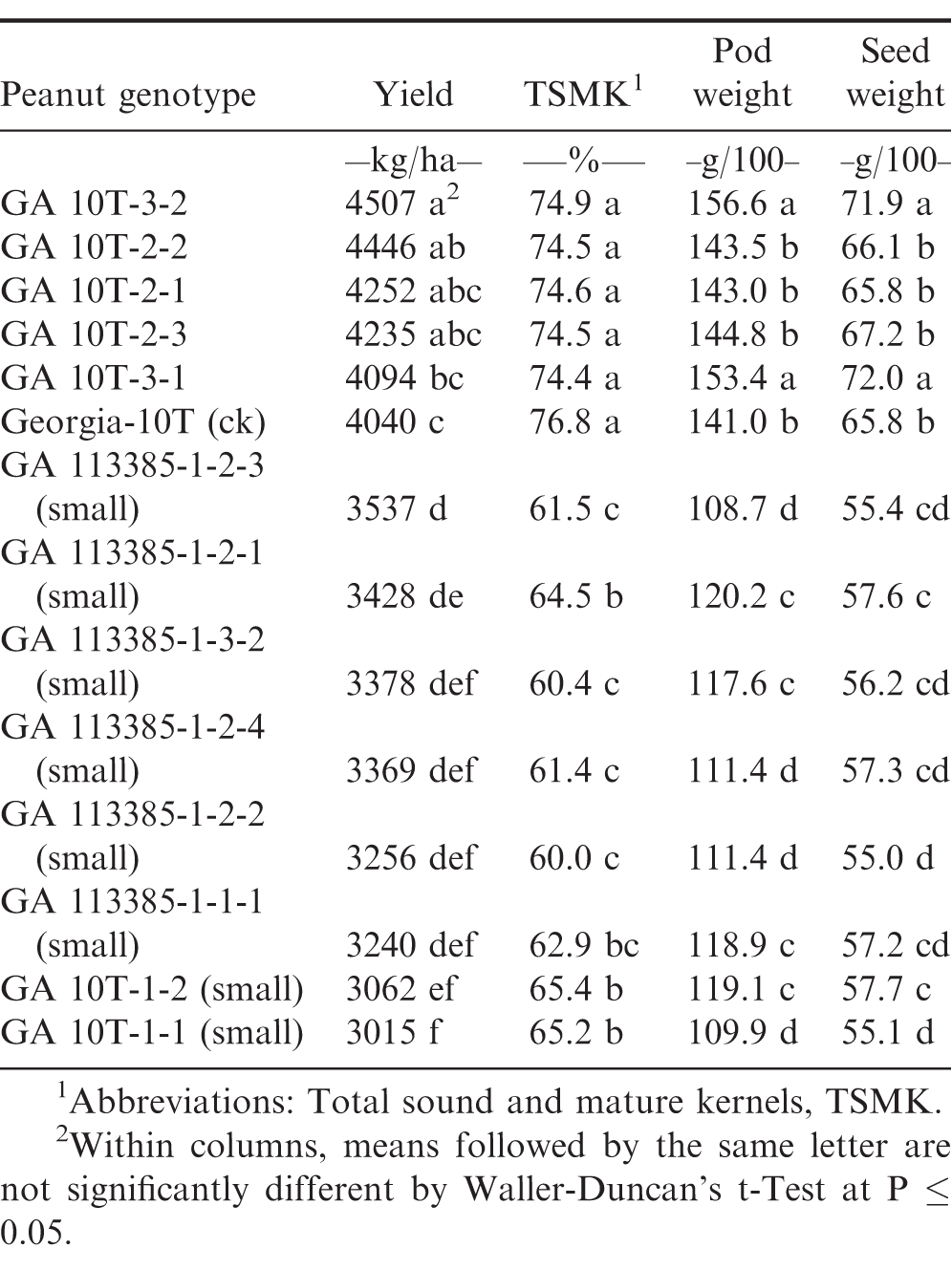
Pod yield, total sound and mature kernel (TSMK) grade, 100 pod weight, and 100 seed weight were each significantly (P ≤ 0.05) different between the two normal selections and check cultivar Georgia-10T compared to the two small-plant selections derived from both naturally and artificially induced drought stress (Tables 1-3). Genotypic comparisons were quite similar across all three 2017 preliminary yield tests (PYT).
In the early (mid-April) planting PYT1 with recommended input production practices and irrigation, Georgia-10T and the artificially-induced normal plant selections (GA 10T-3-1 and GA 10T-3-2) had significantly higher pod yield than the naturally-induced (GA 113385-1-2-2) small plant selection and the artificially-induced small plant selection GA 10T-1-1 (Table 1). Likewise, these small-plant selections had lower TSMK grade percentages and significantly smaller 100 pod and seed weights compared to the normal plant selections.
However, in the optimum (mid-May) planting PYT2 with recommended input production practices and irrigation, Georgia-10T had a significantly lower pod yield compared to the two normal plant selections, GA 10T-3-1 and GA 10T-3-2 (Table 2). All three normal plant genotypes had higher yield, TSMK grade, pod weight, and seed weight compared to the two small plant selections, GA 10T-1-1 and GA 113385-1-2-2.
Table 3 included several additional large and small plant selections compared to Tables 1 and 2; however, Table 3 was likewise an optimum-planting (mid-May) PYT3 but with reduced-input production practices and without irrigation. Drought stress conditions only occurred late during the 2017 growing season. Under these environmental conditions, Georgia-10T and the five other GA 10T large-plant selections, all were found to be significantly higher in pod yield, TSMK grade, pod weight, and seed weight compared to each of the naturally and artificially induced small-plant drought stress selections (Table 3).
Greenhouse Experiment
In an earlier report with dwarf mutants of pea (Pisum sativum L.), Brian and Hemming (1955) reported that GA3 increased the growth rate of shorter internode in the dwarf peas. Consequently, GA3 elongated the internode length and changed the dwarf mutants into normal plant types. Likewise, the current peanut greenhouse experiment was utilized to evaluate the effect of GA3 on these small-plant selections obtained by induced-drought, both naturally (GA 113385-1-2-2) and artificially (GA 10T-1-1).
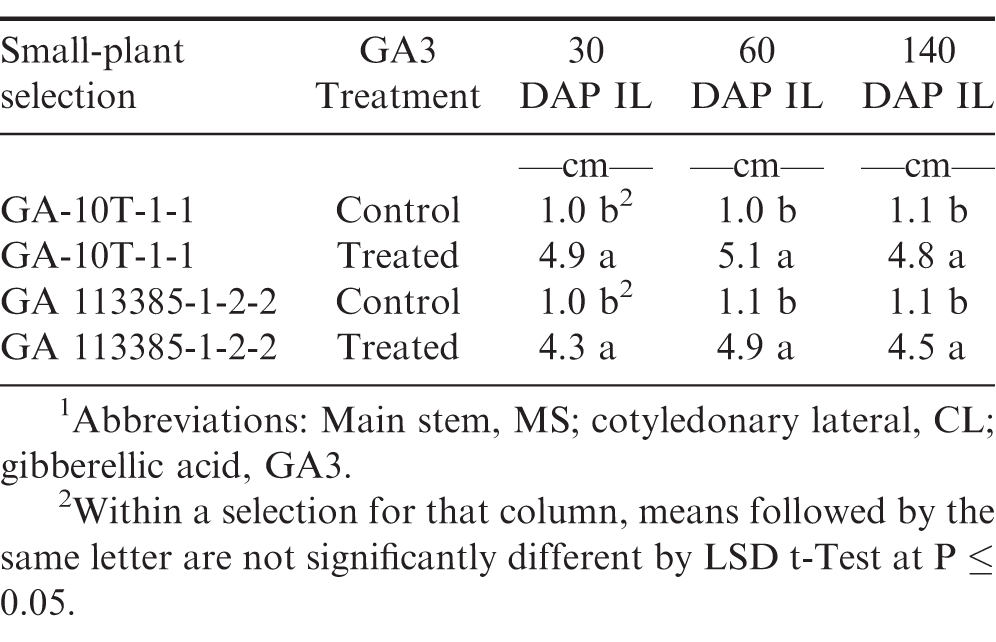
Artificially and naturally induced individual small-plants had similar short main stem (MS) length at 14 DAP for both control and treated plants prior to treatment (Table 4). However at 30, 60, and 140 DAP, the plants treated with GA3 had significantly (P≤0.05) taller MS height and longer cotyledonary lateral (CL) branches at 60 and 140 DAP, respectively.
MS internode lengths (IL) were significantly longer in the GA3 treated plants of artificially and naturally induced small-plants at 30, 60, and 140 DAP as compared to the control plants (Table 5). Obviously, GA3 increased the IL, MS, and CL, of these small-plants. The results of this study agrees with the earlier mentioned report with regard to GA3 increasing growth rates in pea mutants with short internodes (Brian and Hemming, 1955).
No differences were noted in the following generation between GA3 treated vs. control (nontreated) small-plants for plant size, leaflet size, pod number and weight, or number and weight. The temporary changes of GA3 effect only occurred for the small-plants treated in the greenhouse study and did not persist.
Summary and Conclusions
These findings show the importance of avoiding severe early-season drought-stress especially for breeders and growers of foundation, registered, and certified seed of peanut cultivars. Such severe drought-stress can induce development of counterproductive phenotypes such as small-plants with lower pod yield, TSMK grade, pod weight, and seed weight than normal size plants. Development of plants with short internodes appears to be related to hormonal imbalance, possible GA3, since exogeneous GA3 applications only temporary reversed the internode length of the drought stress induced small-plants. This could be a good example of epigenetic inheritance in the cultivated peanut.
Literature Cited
K. J., Boote, J. R Stansell, A. M Schubert, and J. F Stowe (1982). Chap. 7. Irrigation, water use, and water relations. Pp. 164- 205 In:. H.E Patteeand C.T Young (eds.). Peanut Science and Technology. Amer. Peanut Res. And Educ. Soc., Inc. .
W. D Branch, and A. K Culbreath (2011). Registration of 'Georgia-10T' peanut. J. Plant Reg 5: 279- 281.
W. D Branch, and S. M Fletcher (2017). Combination of disease resistance, drought tolerance, and dollar values among runner and virginia-type peanut cultivars in Georgia. Peanut Sci 44: 42- 46.
W. D Branch, and C. K Kvien (1992). Peanut breeding for drought resistance. Peanut Sci 19: 44- 46.
P. W Brian, and H. G Hemming (1955). The effect of gibberellic acid on shoot growth of pea seedlings. Physiol. Plantarum 8: 669- 681.
Klepper B 1973 Water relations of peanut plants Pp 263- 269 In: Peanuts - Culture and Uses, A Symposium. Amer. Peanut Res. & Educ. Assoc., Stillwater, OK.
A. J., Norden, R. W Lipscomb, and W. A Carver (1969). Registration of Florunner peanuts. (Reg. No. 2). Crop Sci 9: 850.
J. R Stansall, and J. E Pallas, (1985). Yield and quality response of Florunner peanut to applied drought at several growth stages. Peanut Sci 12: 64- 70.
USDA-Agricultural Marketing Service 1998 Farmer's stock peanut inspection instructions U.S. Dept. of Agric. Agric. Mkt. Serv. Fruit and Veg. Div. USDA-ARS, Washington, D.C.
Williams, J. H and K. J Boote 1995 Chap. 9 Physiology and modelling - predicting the "unpredictable legume" Pp 301- 353 In: H. E Pattee and H. T Stalker (eds.) Advances in Peanut Science Amer. Peanut Res. and Educ. Soc. Stillwater, OK.
J. E Williams, and J. S Drexler (1981). A non-destructive method for determining peanut pod maturity. Peanut Sci 8: 134- 141.
Notes
- Professor and 2Professor, Dept of Crop & Soil Sciences, and 3Professor, Dept. of Plant Pathology, Coastal Plain Expt. Station, 2360 Rainwater Rd., Tifton, GA 31793-5766 [^] *Corresponding author email: wdbranch@uga.edu
Author Affiliations