Introduction
The combination of improving genetic resources and increasing farmer access to new cultivars is considered one of the most important components of sustainable crop production, especially in circumstances where other components such as pesticides are unavailable or too expensive to purchase (Nigam et al., 2018). Resistance to pests and protection from the damage they cause and the ability of crops to withstand stresses resulting from drought, elevated salt, and other abiotic factors are important components of crop breeding programs in both private and public sectors. Agronomists, pest management specialists, and practitioners develop new strategies and/or refine current practices based on the inherent resistance of cultivars. Integrated Pest Management (IPM) strategies often focus on genetic resistance of cultivars as a first step in minimizing the negative impact of insects, nematodes, fungal pathogens, and viruses on crop yield. Use of pesticides can be decreased when pathogen-resistant cultivars are grown in many crops including peanut (Arachis hypogaea L.). Pest-resistant cultivars are often considered the most cost effective tool for farmers to adopt if yield can be maintained or increased above traditional cultivars and if quality attributes for markets are not compromised.
Developing and maintaining breeding programs can be expensive in both private and public sectors. In many resource-poor countries, investment in these programs is inadequate or only marginally effective in providing a local, robust breeding program that delivers improved cultivars across diverse environments to farmers. To address this issue, organizations like ICRISAT and other CGIAR centers provide germplasm to scientists in resource-poor countries that offer potential improvement over traditional cultivars or land races. Partnerships are often developed among the CGIAR centers, government agencies and national programs associated with agriculture and rural development, funding agencies such as USAID with public and private cooperative agreements including universities, and non-governmental organizations (NGOs). In this note we provide information on the timeline, research methodology, and other metrics used in evaluation and release of two cultivars (Yenyawoso and Otuhia) in Ghana through partnerships among CSIR-CRI, ICRISAT, the Ministry of Food and Agriculture (MoFA) in Ghana, NCSU, USAID Peanut CRSP and PMIL.
Materials and Methods
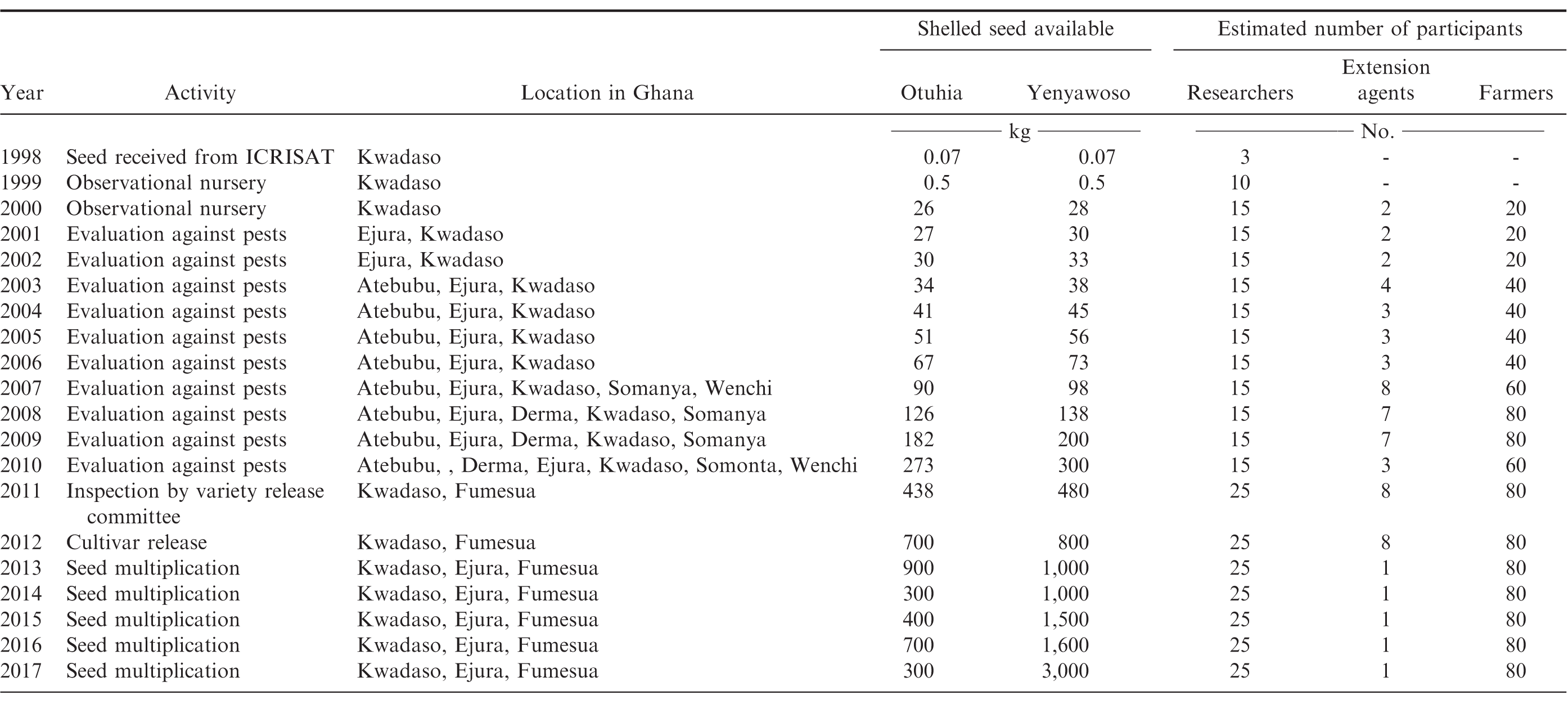
Approximately twenty seeds of the ICRISAT lines ICGX SM 87057 and ICGV SM 88709 (non-segregating, F7 generation) were received by scientists at CSIR-CRI in 1998. The pedigree for ICGX SM 87057 constitutes a cross between ICGM 197 and ICGM 751/754 (Pasupuleti, ICRISAT, personal communication). The genetic background of ICGV-SM 88709 is (SP 1 × RMP 91)-P12-P1-B2 (Pasupuleti, ICRISAT, personal communication). Thirty lines including ICGX SM 87057 and ICGV SM 88709 were observed at the CSIR-CRI facility near Kwadaso for two years (referred to as Observational Nursery). Ten lines were selected for further testing over the following 10 years including two locations during 2001 and 2002, 3 locations from 2003-2006, five locations from 2007-2009, and six locations during 2010 (Table 1). Experiments were conducted on research stations within the CSIR-CRI system and in farmer fields to determine the response of these lines to biotic and abiotic stresses in Ghana. Commercially-available cultivars were included in many of these experiments depending on seed availability and space allocation. These lines were also selected for advanced testing in cropping systems research with various levels of production and pest management Seed multiplication has continued since the release of these cultivars in 2012. Purity of these lines was maintained, and seed were dried after harvest and stored in a manner that maintained germination and seedling vigor. Participatory Variety Selection, primarily through Farmer Field Schools, was an important platform for evaluation of these experimental lines in Ghana, especially in obtaining input from farmers, MoFA, the NGO community, and the agricultural business sector (Mochiah, CSIR-CSI, personal communication).
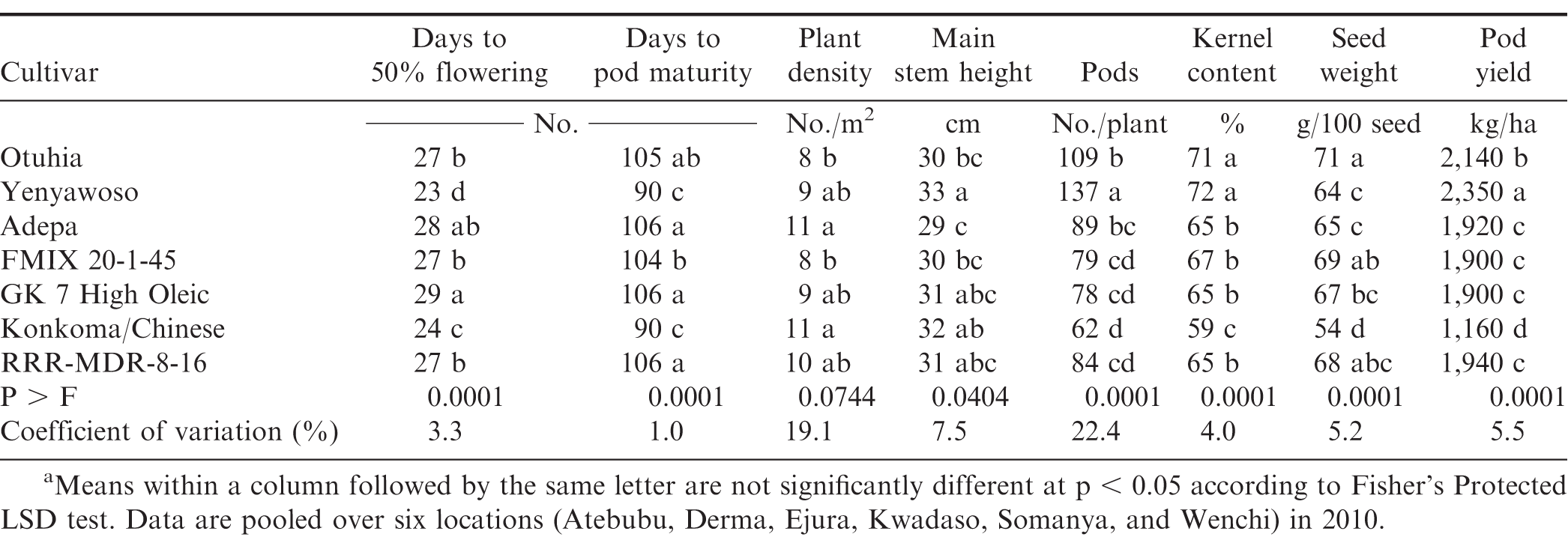
Data collected during the evaluation of these lines included days from planting to 50% flowering, d from planting to 70% of pods in the brown and black mesocarp color category as defined by Williams and Drexler (1981), plant density, main stem height, number of pods per plant, kernel content at harvest presented as percent of farmer stock mass weight, seed weight, and pod yield adjusted to 8% moisture. Data for leaf spot disease [caused by Cercospora arachidicola Hori or Cercosporidium personatum (Berk. & M.A. Curtis) Deighton], rust (caused by Puccinia arachidis Speg.), damage from arthropods, and plants expressing symptoms of disease caused by rosette virus (Tombusviridae umbravirus) were recorded. An example of agronomic characteristics for these cultivars from six locations for one growing season (2010) are presented compared with commercially-available cultivars planted in the region. Means were separated using Fisher's Protected LSD at p ≤ 0.10.
Results and Discussion
Two ICRISAT breeding lines were released as cultivars Otuhia (virginia botanical classification) and Yenyawoso (Spanish botanical classification) by CSIR-CRI in 2012. These cultivars provided several improved characteristics relative to agronomic performance and market attributes compared with the traditional cultivar Konkoma (Table 2).The cultivar Konkoma is also referred to as Chinese depending on the region of Ghana where peanut is grown. The descriptor for Otuhia (ICGV SM 88709), in the Akan language is translated "drives away poverty" in the English language. The phenotypic expression of this cultivar includes a prostrate growth habit with leaflets elliptical in shape. Flowering occurs approximately 27 d after emergence with no flowers present on the main stem. Lateral branches attached to the main stem present alternate flowering pattern and require approximately 110 d from planting to optimum pod maturity. Pods contain two seeds that are slightly beaked with slight constriction. This cultivar expressed field tolerance to rosette virus vectored by aphids (Aphis craccivora), early and late leaf spot, and rust (Anonymous, 2019; Lamptey et al., 2014). The descriptor for the cultivar Yenyawoso (ICGX SM 87057) in the Akan language is translated "there is no one like you" in the English language. This cultivar is characterized as semi-erect with obovate-shaped leaflets. Flowering is observed approximately 28 d after emergence on main stems with lateral branches expressing a sequential flowering pattern and requiring approximately 90 d from planting to optimum pod maturity. Unlike Otuhia, this cultivar expressed a modest level of field tolerance to pests compared with Konkoma but was no more effective in resisting pests than currently released cultivars. In contrast, Yenyawoso reaches optimum maturity similar to Konkoma and more quickly than Otuhia and comparison cultivars (Table 2). This trait is of value when rainfall toward the end of the growth cycle is limited.
The expense incurred by ICRISAT relative to developing a single experimental line that has potential to become a cultivar release is difficult to estimate. The cultivars discussed here derived from ICRISAT lines were a part of a much larger system of testing prior to being made available to scientists in Ghana. While resources and investment by the national program have increased in Ghana since 1999, the availability of germplasm through the ICRISAT system continues to be an important part of cultivar development in Ghana. The majority of resources enabling testing of these cultivars prior to and following release on research stations and in farmer fields were obtained through the Peanut CRSP and more recently the PMIL.
Attempts are underway to scale up seed of the cultivars discussed in this note for farmers across climate zones in Ghana. In 2017, enough seed was available for purchase to plant 75 ha of Yenyawoso based on the recommended seeding rate of 60 kg/ha to obtain a density of 100,000 seed/ha. Yield of Yenyawoso was twice that of the locally-grown cultivar Chinese. Currently there is less interest in planting the cultivar Otuhia (5 ha), which is most likely a reflection of the larger kernel size of this cultivar, the longer period of time required to reach optimum maturity, and the lower yield potential compared with Yenyawoso (Table 2). However, the larger kernel size could be a positive characteristic for specific aggregators and processors as the peanut industry in Ghana becomes more discriminating based on consumer demand. It is possible that these calculations underestimate the presence of these cultivars with farmers. A common practice in these research programs is to provide farmers with a small quantity of seed for their use during the following year, and that effect on distribution has not been quantified.
While these cultivars are improvements over traditional cultivars and cultivars released during the previous two decades, access to these cultivars is limited in Ghana because of challenges associated with the current national seed chain for peanut and complications in shifting away from traditional cultivars with well-established production systems and processor and consumer preferences. For example, even though some of the improved cultivars listed in Table 2 have been available for a reasonable amount of time, 80 to 90% of land area devoted to peanut in Ghana is planted with Konkoma and Chinese or locally-produced seed of unknown origin (J. Y. Asibuo, CSIR-CRI, and R. Oteng-Frimprong, CSIR-Savanna Agricultural Research Institute, personal communication). Oppong-Sekyere et al. (2015) reported that the cultivars Otuhia and Yenyawoso were grown by no more than 0.5% of farmers in northern Ghana in 2015. None-the-less, efforts are underway to promote and distribute improved cultivars by several groups in Ghana. For example, Yenyawoso was promoted by the Feed the Future project SPRING (SPRING, 2017), and the Feed the Future Scaling project in cooperation with ICRISAT (Anonymous, 2018) received more than 5 metric tons of Yenyawoso foundation seed from CSIR-CRI for multiplication. These seed will be certified for distribution to farmers in the three Northern Regions as an alternative to traditional cultivars (J. Y. Asibu, CSIR-CRI, personal communication). Improving seed storage and delivery strategies and maintaining the purity and quality that ensure high germination and seedling vigor have the potential to increase peanut production and profitability in Ghana, especially when combined with improved cultivars like Otuhia and Yenyawoso. These cultivars may also provide traits that are valuable for future releases of cultivars through breeding programs in Ghana (Desmae et al., 2018; Gaikpa et al., 2015).
Acknowledgements
The original experimental lines ultimately released were provided by ICRISAT. This publication was made possible through support provided by the Office of Agriculture, Research and Policy, Bureau of Food Security, U.S. Agency for International Development, under the terms of Award No. AID-ECG-A-00-07-0001 to The University of Georgia as management entity for the Peanut Collaborative Research Support Program (1999-2012) and U.S. Feed the Future Innovation Lab on Peanut Productivity and Mycotoxin Control (2012-2017). The opinions expressed herein are those of the author(s) and do not necessarily reflect the views of the U.S. Agency for International Development. Appreciation is expressed to staff and farmers for assistance in developing these cultivars in Ghana. P. Janila assisted with information on the pedigree of lines from ICRISAT.
Literature Cited
Anonymous 2018 Increasing groundnut productivity of smallholder farmers - Groundnut technologies upscaling (USAID) Available at: http://exploriet.icrisat.org/resource.data/project_indetail/15171 Accessed October 2, 2018.
Anonymous 2019 CSIR Crop varieties released and registered in Ghana Document available at: https://www.csir.org.gh/images/Doc/NewsLetter/CROP%20VARIETIES%20RELEASED20AND%20REGISTERED%20IN%20GHANA.pdf Accessed November 30, 2018.
Desmae, H., J Pasupuleti, B. N Motagi, E Monyo, R Oteng-Frimpong, C Ojiewo, and R. K Varshney 2018 Genetics, genomics and breeding of groundnut (Arachis hypogaea L.) Plant Breeding DOI: 10.1111/pbr.12645.
D. S., Gaikpa, R Akromah, J. Y Asibuo, and D Nyadanu (2015). Studies on molecular variation in commercially cultivated groundnuts (Arachis hypogaea L.) using SSR markers. The International Journal of Science and Technoledge 3: 80- 85.
Lamptey, J. N. L., F Anno-Nyako, M Owusu-Akyaw, M. B Mochiah, G Bolfrey-Arku, K Osei, R. L Brandenburg, and D. L Jordan 2014 Incidence and management of peanut (Arachis hypogaea L.) rosette disease in southern Ghana International Journal of Agriculture Innovations and Research Vol. 2, Issue 4, ISSN (Online) 2319-1473.
Nigam, S. N., D. L Jordan, and P Janila 2018 Improving cultivation of groundnuts Pages 155- 180 in S Sivasankar, D Berguinson, P Gaur, S Kumar, S Beebe, and M Tamó, eds., Achieving Sustainable Cultivation of Grain Legumes; Improving Cultivation of Particular Grain Legumes. Volume 2 Burleigh Dodds Series in Agricultural Science, Burleigh Dodds Science Publishing, Cambridge, UK.
D., Oppong-Sekyere, R Akromah, M. M Akpalu, A. D Ninfaa, E. Y Nyamah, M. M Braimah, and S Salifu (2015). Participatory rural appraisal of constraints to groundnut (Arachis hypogaea L.) production in northern Ghana. International J. Current Research and Academic Review 3 ((10)): 54- 76.
SPRING [Strengthening Partnerships and Innovation in Nutrition Globally] 2017 Farmer field school curriculum: good agronomic practices for groundnut Available at: https://www.jsi.com/JSIInternet/Inc/Common/_download_pub.cfm?id=19155&lid=3 Accessed September 30, 2018.
E.J., Williams, and J.S Drexler (1981). A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification. Peanut Sci 8: 134- 141.
Notes
- Crops Research Institute, Council for Scientific and Industrial Research, Kumasi, Ghana. [^]
- Savanna Agricultural Research Institute, Council for Scientific and Industrial Research, Tamale, Ghana. [^]
- Savanna Agricultural Research Institute, Council for Scientific and Industrial Research, Wa, Ghana. [^]
- Kwame Nkrumah University of Science and Technology, Kumasi, Ghana. [^]
- Department of Entomology and Plant Pathology, North Carolina State University, Raleigh, NC 27695. [^]
- Department of Crop and Soil Sciences, North Carolina State University, Raleigh, NC 27695. [^]
- USAID Peanut Collaborative Research Support Program (1999-2012), University of Georgia, Griffin, GA. [^]
- Feed the Future Innovation Lab for Peanut, University of Georgia, Athens, GA. [^] *Corresponding author's email: david_jordan@ncsu.edu
Author Affiliations