Introduction
Cylindrocladium black rot (CBR) caused by the soilborne pathogen Cylindrocladium parasiticum Crous, Wingfield, and Alfenas, syn C. crotalariae (Loos) Bell and Sobers has long been a major disease problem in U.S. peanut (Arachis hypogaea L.) production. Consequently, new and improved CBR-resistant cultivars are continually needed to minimize damage, reduce chemical control costs, and increase yield, grade, and dollar value returns. These same cultivars also need to be resistant to tomato spotted wilt virus (TSWV) which is likewise another major peanut disease problem in the U.S.
In an earlier 3-yr extensive study under uniformly heavy disease pressure at the University of Georgia, Southeast Research and Education Center near Midville, GA (Branch and Brenneman, 2003), three CBR-resistant runner market type peanut cultivars, ‘Georgia-01R’ (Branch, 2002), ‘Georgia-02C’ (Branch, 2003), and ‘Carver’ (Gorbet, 2006) were all found to be similar in disease ratings as the CBR-resistant virginia-type checks, NC 3033 (Beute et al., 1976), ‘NC 8C’ (Wynne and Beute, 1983), and ‘Perry’ (Isleib et al., 2003), and produced significantly higher pod yields. However, ‘C-99R’ (Gorbet and Shokes, 2002), ‘Southern Runner’ (Gorbet et al., 1987), and ‘Florida MDR 98’ (Gorbet and Shokes, 2002) had among the highest percentage of CBR disease incidence and were considered to be among the most CBR susceptible.
Since this earlier study, several newer runner-type peanut cultivars have been developed and released. Among these, the nematode-resistant, runner-type cultivar, ‘Tifguard’ (Holbrook et al., 2008) was reported to be similar to Georgia-02C for CBR resistance in greenhouse and microplot studies (Dong et al., 2009a). So, the objective of the present study was to determine if potentially new sources of CBR resistance exist among more recently released runner-type peanut cultivars under C. parasiticum inoculated field conditions.
Materials and Methods
During 2008 and 2009, field tests were conducted at the Gibbs farm and in 2009 and 2010 at the Blackshank farm near the UGA, Coastal Plain Experiment Station, Tifton, GA. Each year, several different genotypes were evaluated at these two locations for CBR resistance compared to the previously found CBR-resistant check (ck) cultivars, Georgia-01R, Georgia-02C, Carver, Perry and the CBR-susceptible check (ck) cultivar, C-99R. The Gibbs farm test site has a long history of continuous peanut production, and the soil type at both locations was a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults).
Planting dates at the Gibbs Farm were April 15, 2008 and April 17, 2009, and the seeding rates were six seed per 30.5-cm of row. Recommended cultural practices with irrigation were used throughout each growing season, except no fungicides were applied with activity against CBR. All plots were artificially inoculated with microsclerotia of C. parasiticum in early June at approximately 50 days after planting (DAP) each year to avoid summer heat stress during the inoculation process.
Several isolates of C. parasiticum collected the previous year were grown for 4–6 weeks on 90-cm-dia petri plates with potato dextrose agar. Approximately 2 plates per plot were ground in a Waring blender with water and the slurry poured through cheese cloth. Enough water was added to give a total volume of 100 ml per plot, and this suspension of microsclerotia and hyphae was sprayed on the plant crowns with a pump-up garden sprayer. All plots were irrigated following inoculation.
A randomized complete block design was used each year with six replications. Plots consisted of two rows 6.10 m long × 1.83 m wide (0.81 m within and 1.02 m between rows on adjacent plots). Percentages (0–100%) of early-season TSWV disease incidence were scored at approximately 60 days after planting (DAP) when TSWV is usually the only disease occurring. Since some symptoms of CBR and TSWV are very similar, both mid-season TSWV + CBR were evaluated at ca. 100 DAP, and again just prior to harvest. The symptoms of CBR are most evident on plant roots, so CBR disease was also rated immediately after digging each year. A disease hit equaled one or more symptomatic plants within a 30.5-cm section row. Individual plots were harvested near optimum maturity according to visual above-ground disease pressure in conjunction with the hull-scrape maturity method from adjoining border plots (Williams and Drexler, 1981). Plots were mechanically harvested, and pods were dried with forced warm air to 6% moisture content, then hand-cleaned over a screen table before weighing for pod yield. Data from each test was subjected to analysis of variance. Waller-Duncan's T-test (k-ratio = 100) was used for mean separation of significant differences (P≤0.05).
Planting dates at the Blackshank Farm were May 20, 2009 and May 20, 2010, and the seeding rates were five seed per 30.5-cm of rows. Several weeks before planting, the field was treated with methyl bromide and chloropicrin at a rate of 448 kg/ha. A plastic tarp was used to contain the gas for several days and was then removed. This treatment will result in the elimination of most soilborne pathogens. Recommended cultural practices with irrigation were used throughout each growing season, and the only fungicide applied was chlorothalonil to control foliar diseases, but it has no activity against CBR. Inoculated plots were artificially inoculated with microsclerotia of C. parasiticum also in early June at approximately 15 DAP each year as described previously to avoid summer heat stress during the inoculation process. A split-plot design was used with genotypes as whole plots and non-inoculated vs. inoculated with C. parasiticum as sub plots with four replications. Plots consisted of two rows 7.62 m long × 1.83 m wide with 0.91 m spacing between rows. The CBR disease incidence was scored immediately after digging each year. A disease hit equaled one or more symptomatic plants within a 30.5-cm section of inverted row. All plots were dug at the same time, 152 DAP in 2009. However in 2010, digging was delayed until 166 DAP due to extremely hot conditions during the summer which inhibited CBR development until temperatures declined in the fall. Plots were mechanically harvested, and pods were dried with forced warm air to 6% moisture content, then hand-cleaned over a screen table before weighing for pod yield. Data from each test was subjected to analysis of variance, and least significant differences (LSD) was used for mean separation (P≤0.05).
Results and Discussion
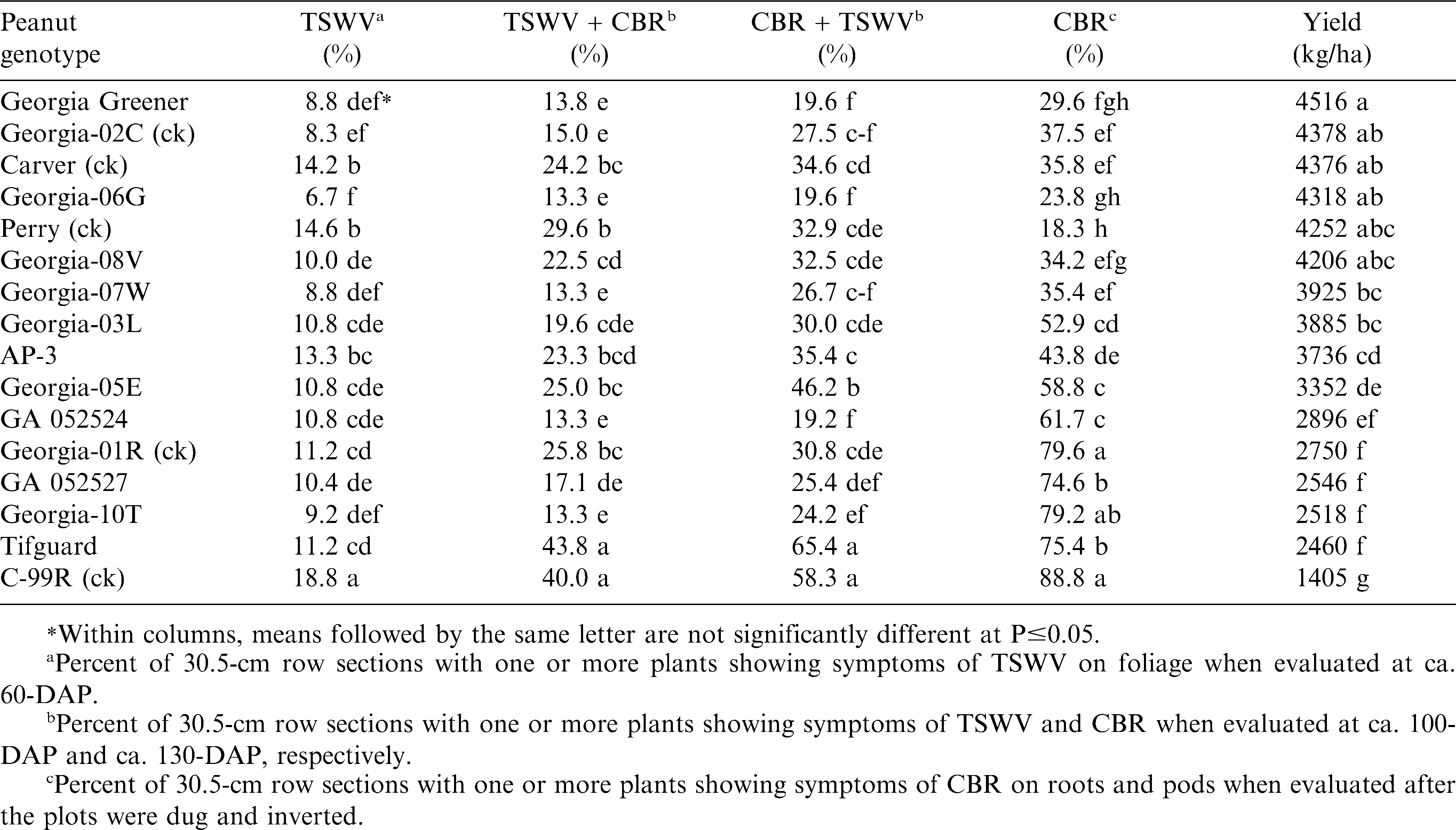
During 2008 at the Gibbs Farm (Table 1), ‘Georgia-06G’ (Branch, 2007) had the lowest early-season TSWV disease incidence, but it was not significantly (P≤0.05) different from ‘Georgia Greener’ (Branch, 2007), Georgia-02C, ‘Georgia-07W’ (Branch and Brenneman, 2008), and ‘Georgia-10T’ (Branch and Culbreath, 2011). At both mid-season and late-season, TSWV and CBR incidence was also lowest for these same five cultivars, and GA 052524 and GA 052527, two sister lines of Georgia-10T. However after digging, CBR incidence was the lowest with Perry, the virginia-type resistant check; whereas, CBR incidence was highest (most susceptible) with C-99R. These findings agree with the previous report by Branch and Brenneman (2003). However, both Georgia-06G and Georgia Greener were also found in this 2008 study to have CBR resistance and high yields, and the late-maturing genotypes, Georgia-01R, Georgia-10T, GA 052524, GA 052527, and the medium-maturing cultivar, Tifguard were all found to be CBR susceptible. These findings with Tifguard and Georgia-01R disagree with earlier reports (Dong et al., 2009a and Branch and Brenneman, 2003), where both cultivars had previously shown some CBR resistance. Since the tests in this study were inoculated, it is understandable that later-maturing genotypes, such as Georgia-01R, were exposed longer to high-levels of CBR before digging as compared to non-inoculated evaluations. However, this would not be the same for Tifguard which is reported to be a medium-maturing cultivar (Holbrook et al., 2008), and as such was dug earlier than Georgia-01R.
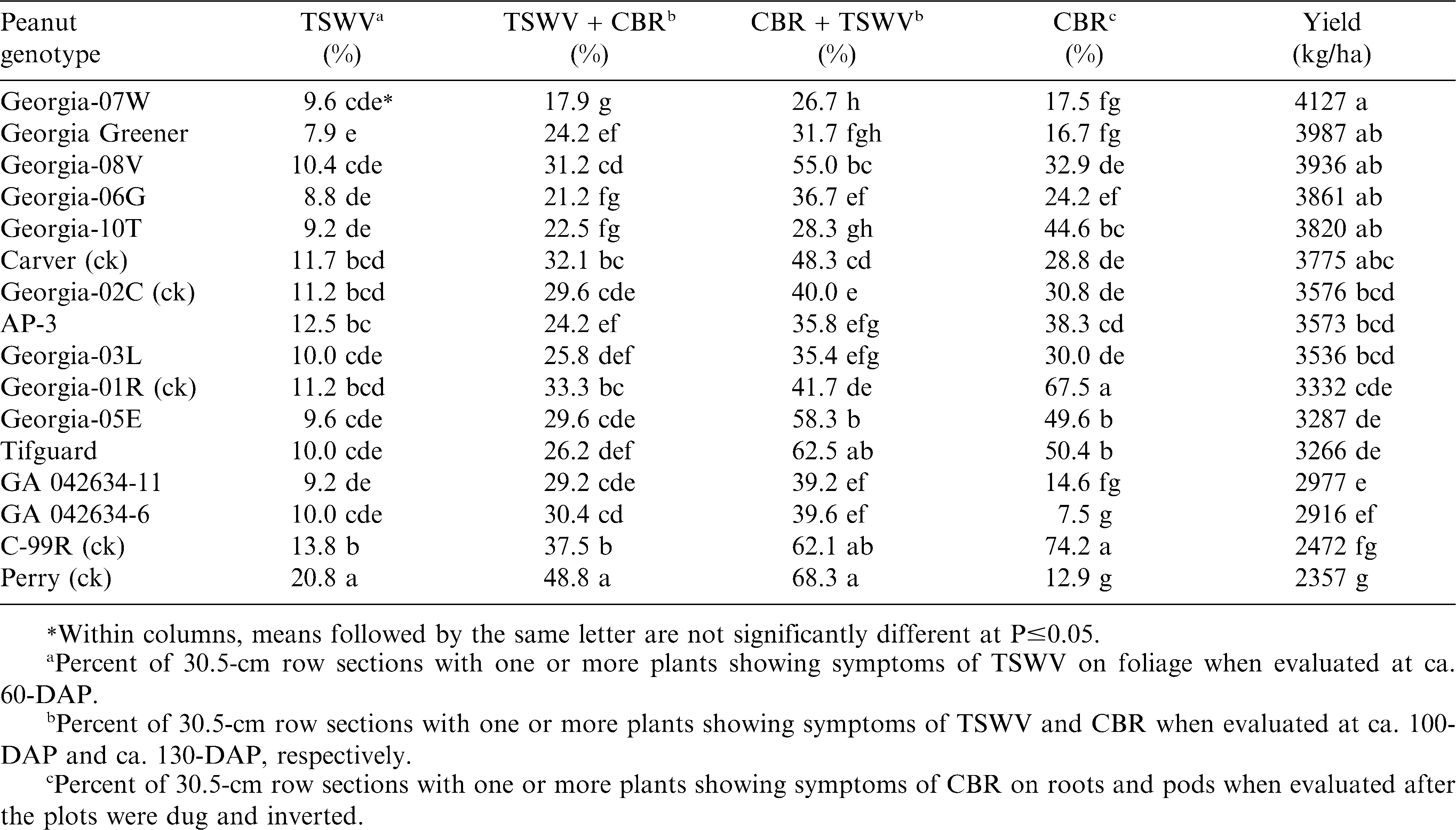
During 2009 at the Gibbs Farm (Table 2), Georgia Greener, Georgia-06G, Georgia-10T, Georgia-07W, GA 042634-11, and ‘Georgia-05E’ (Branch, 2006) had the lowest TSWV incidence, and Perry had the highest TSWV incidence earlier in the season. These results agree with a previous 3-yr maximum and minimum production input study for TSWV disease resistance (Branch and Fletcher, 2010). Again at both mid and late-season, TSWV and CBR incidence was found to be the lowest with Georgia-07W, Georgia-06G, Georgia Greener, and Georgia-10T. After digging, CBR incidence was again lowest with Perry, Georgia Greener, Georgia-07W, and two advanced Georgia breeding lines, GA 042634-6 and GA 042634-11. However, high CBR incidence and low-pod yield was again found for C-99R, Georgia-01R, and Tifguard which agrees with the 2008 results.
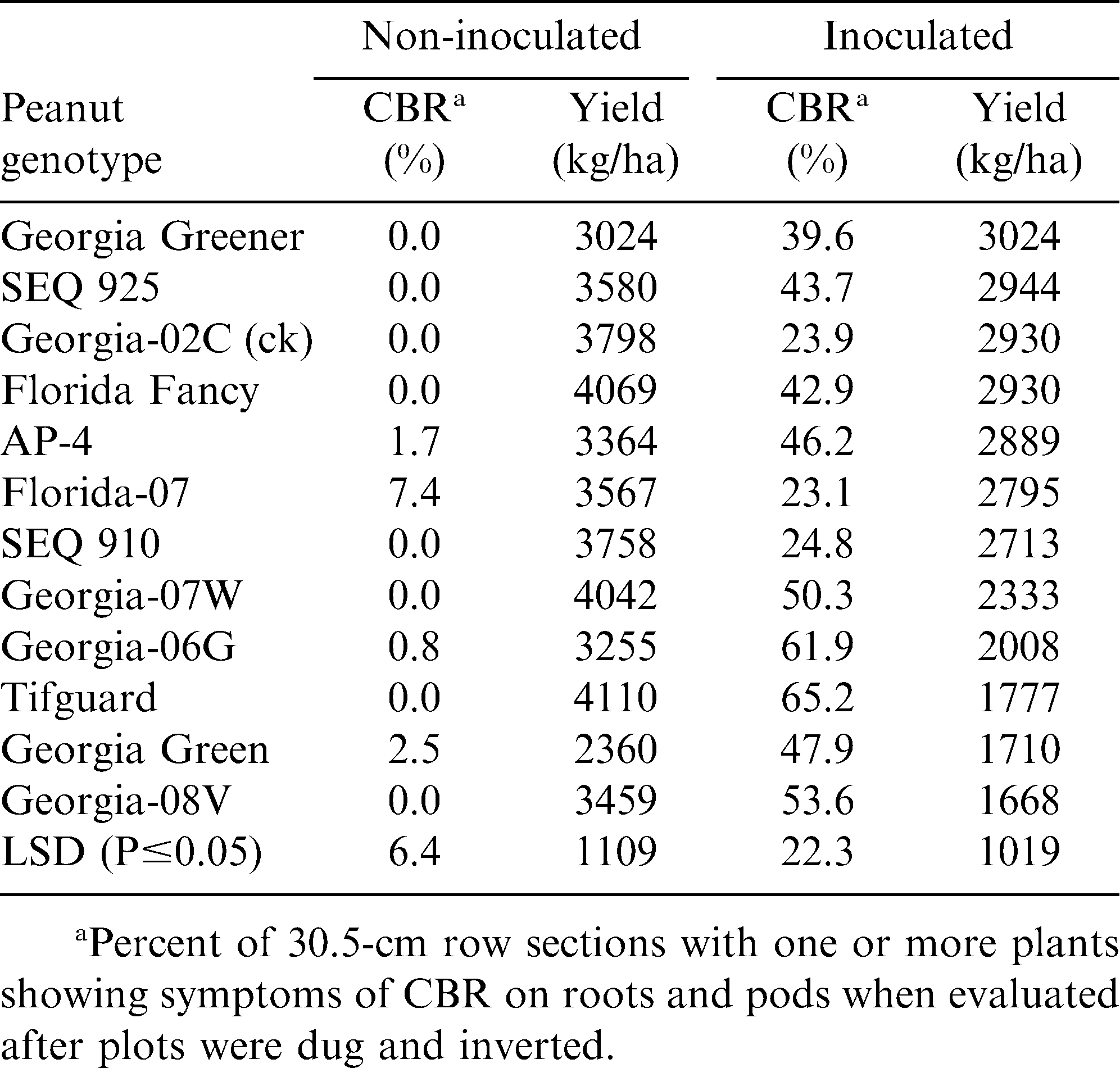
At the Blackshank Farm in 2009 (Table 3), non-inoculated vs. C. parasiticum inoculated plots showed significant differences between many of these same peanut genotypes. Georgia Greener, Florida-07, and SEQ 910 were found to have similar CBR resistance, comparable to Georgia-02C; whereas, Tifguard was found to have the highest CBR incidence after inoculation and low pod yield. Pod yield for Tifguard was very high in non-inoculated plots. However, Georgia Greener had the same yield in both non-inoculated and inoculated field trials, and Tifguard had the greatest yield difference (2333 kg/ha), again reflecting its susceptibility to CBR.
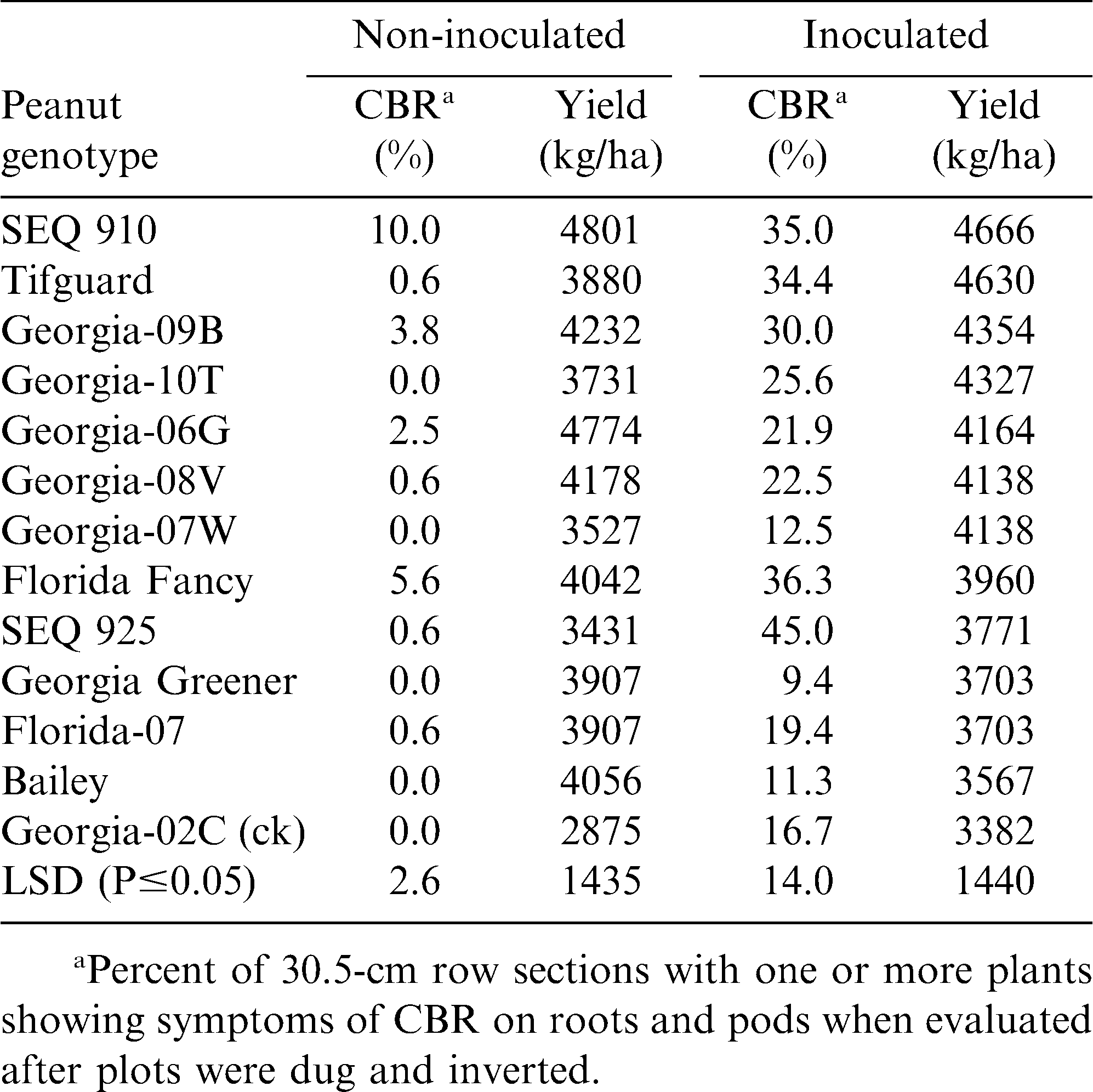
When the C. parasiticum inoculated vs. non-inoculated tests were repeated in 2010 at the Blackshank Farm (Table 4), similar results were found with Georgia Greener showing CBR resistance and Tifguard having CBR susceptibility. Yields were not significant in the 2010 inoculated tests because of delaying harvest until very late fall in an attempt to allow some disease to develop. Overall there was more variability in this test, and disease development was greatly delayed due to the extremely hot, dry weather in 2010. Although significant disease levels eventually developed; symptoms were primarily pod discoloration and had little effect on yield.
Summary
Results from three years (2008-10) at two Tifton, GA locations (Gibbs Farm and Blackshank Farm) clearly show new sources of CBR resistance with Georgia Greener being the most consistent in low disease incidence and high pod yield. However, these findings also show that Tifguard is highly susceptible to CBR, and is not CBR resistant as previously reported from microplot studies by Dong et al., 2009a. However as Dong et al., (2009b) concluded, CBR screening of peanut genotypes are most reliably done in the field with inoculation or using uniformly infested field sites, as previously described by Branch and Brenneman (2003).
Literature Cited
Beute M.K Wynne J.C and Emery D.A 1976 Registration of NC 3033 peanut germplasm (Reg. No. GP 9) Crop Sci. 16 : 887 .
Branch W.D 2002 Registration of ‘Georgia-01R’ peanut Crop Sci. 42 : 1750 – 1751 .
Branch W.D 2003 Registration of ‘Georgia-02C’ peanut Crop Sci. 43 : 1883 – 1884 .
Branch W.D 2006 Registration of ‘Georgia-05E’ peanut Crop Sci. 46 : 2305 .
Branch W.D 2007 Registration of ‘Georgia-06G’ peanut J. Plant Reg. 1 : 120 .
Branch W.D 2007 Registration of ‘Georgia Greener’ peanut J. Plant Reg. 1 : 121 .
Branch W.D and Brenneman T.B 2003 Field resistance to Cylindrocladium black rot and tomato spotted wilt virus among advanced runner-type peanut breeding lines Crop Protection 22 : 729 – 734 .
Branch W.D and Brenneman T.B 2008 Registration of ‘Georgia-07W’ peanut J. Plant Reg. 2 : 88 – 91 .
Branch W.D and Culbreath A.K 2011 Registration of ‘Georgia-10T’ peanut J. Plant Reg. 5 : 279 – 281 .
Branch W.D and Fletcher S.M 2010 Agronomic performance and economic return among peanut genotypes with maximum and minimum production inputs Peanut Sci. 37 : 83 – 91 .
Dong W.B Brenneman T.B Holbrook C.C Timper P and Culbreath A.K 2009a The interaction between Meloidogyne arenaria and Cylindrocladium parasiticum in runner peanut Plant Path. 58 : 71 – 79 .
Dong W.B Brenneman T.B Holbrook C.C and Culbreath A.K 2009b Evaluation of resistance to Cylindrocladium parasiticum of runner-type peanut in the greenhouse and field Peanut Sci. 35 : 139 – 148 .
Gorbet D.W 2006 Registration of ‘Carver’ Peanut Crop Sci. 46 : 2713 – 2714 .
Gorbet D.W and Shokes F.M 2002 Registration of ‘C-99R’ peanut Crop Sci. 42 : 2207 .
Gorbet D.W and Shokes F.M 2002 Registration of ‘Florida MDR 98’ peanut Crop Sci. 42 : 2207 – 2208 .
Gorbet D.W Norden A.J Shokes F.M and Knauft D.A 1987 Registration of ‘Southern Runner’ peanut Crop Sci. 27 : 817 .
Holbrook C.C Timper P Culbreath A.K and Kvien C.K 2008 Registration of ‘Tifguard’ peanut J. Plant Reg. 2 : 92 – 94 .
Isleib T.G Rice P.W Mozingo R.W Bailey J.E Mozingo R.W and Pattee H.E 2003 Registration of ‘Perry’ peanut Crop Sci. 43 : 739 – 740 .
William E.J and Drexler J.S 1981 A non-destructive method for determining peanut pod maturity Peanut Sci. 8 : 134 – 141 .
Wynne J.C and Beute M.K 1983 Registration of NC 8C peanut (Reg. No. 27) Crop Sci. 23 : 183 – 184 .
Notes
- University of Georgia, Dept. of Crop & Soil Sciences, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793-5766
- University of Georgia, Dept. of Plant Pathology, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793-5766 *Corresponding author (email: wdbranch@uga.edu)
Author Affiliations