Introduction
Peanut (Arachis hypogaea L.) has several unique features that contribute to challenging weed management. Peanut cultivars grown in the United States require a long growing season (140 to 160 d), depending on cultivar and geographical region [Henning et al., 1982; Wilcut et al., 1995]. Peanut also has a prostrate growth habit, a relatively shallow canopy, and is slow to shade inter-rows allowing weeds to be more competitive [Walker et al., 1989; Wilcut et al., 1995). Consequently, herbicides applied at planting may not provide season-long control and mid-to-late season weed emergence can occur. Additionally, peanut fruit develops underground on pegs originating from branches that grow along the soil surface. This prostrate growth habit and pattern of fruit development restricts cultivation to an early-season control option [Wilcut et al., 1995; Brecke and Colvin, 1991). With conventional row spacing (91 to 102 cm), complete ground cover may not be attained until 8 to 10 wk after planting. In some areas of the United States peanut growing region, complete canopy closure may never occur.
Weeds compete with peanut for sunlight, moisture, and nutrients and may reduce harvesting efficiency. Weeds are particularly troublesome during digging and inverting procedures (Young et al., 1982). Weed biomass slows field-drying of peanut vines and pods and increases the likelihood of exposure to rainfall, which may increase harvesting losses (Wilcut et al., 1995; Young et al., 1982). The fibrous root system of annual grasses is extremely difficult to separate from peanut (Wilcut et al., 1994).
Acetochlor is a chloroacetanilide herbicide and the mode of action is elongase inhibition and inhibition of geranylgeranyl pyrophosphate (GGPP) cyclization enzymes, which is part of the gibberellin biosynthetic pathway (Arregui et al., 2010) and controls weeds by inhibiting growth of seedling shoots (Ross and Childs, 1996). Acetochlor controls a broad spectrum of weeds in corn (Zea mays L.), cotton (Gossypium hirsutum L.), soybean (Glycine max L.), and various other crops (Anonymous, 2012; 2014b). Steckel et al. (2002) reported inconsistent control of common waterhemp (Amaranthus rudis L.) in corn with chloroacetamide herbicides. However, they also reported that encapsulated acetochlor formulations controlled common waterhemp at least 85% regardless of application method when evaluated 56 d after planting. Armel et al. (2003) reported that tank mixes of mesotrione plus acetochlor controlled smooth pigweed (Amaranthus hybridus L.) and giant foxtail (Setaria faberi Herrm.), but did not adequately control common ragweed (Ambrosia artemisiifolia L.), common lambsquarters (Chenopodium album L.), or morningglory species (Ipomoea spp.). Geier et al. (2009) found that acetochlor, in combination with atrazine, controlled large crabgrass [Digitaria sanguinalis (L.) Scop.] in grain sorghum [Sorghum bicolor (L.) Moench.] 55 to 76% in one yr but 94% or greater in two other yrs. However, shattercane [Sorghum bicolor (L.) Moench.] was controlled less than 20%.
Parker et al. (2005) compared acetochlor as an emulsifiable concentrate (EC) with two encapsulated formulations, capsule suspension (CS) and microencapsulated (ME). The CS formulation controlled giant foxtail 62 to 74% while the EC and ME formulations controlled 43 to 46%, 180 d after application. The encapsulated formulation of acetochlor (Warrant®) (Anonymous, 2010) is now commercially available and provides greater crop safety in several crops, including soybean and was designed to give PRE and postemergence (POST) weed control in acetolactate synthase (ALS) and glyphosate resistant weeds (Anonymous, 2010; 2014b). The encapsulated formulation requires limited moisture for activation (Anonymous, 2010), helps minimize crop injury, and also can extend weed control for up to 40 d (Anonymous, 2010; 2014b). Acetochlor recently received approval from the Environmental Protection Agency for use on peanut in the U. S. (Anonymous, 2014b).
The objectives of this research were: 1) to evaluate and compare weed efficacy of the new encapsulated acetochlor with flumioxazin, S-metolachlor, and pendimethalin alone or acetochlor in combination with pendimethalin or followed by POST applications of lactofen, and 2) to evaluate peanut cultivar response to the encapsulated acetochlor when applied at different rates and timings.
Materials and Methods
Studies were conducted during 2011 and 2012 at Texas A&M AgriLife Research site near Yoakum in south-central Texas (29.276o N, 97.123o W) and at the Texas A&M AgriLife Research and Extension Center at Halfway (34.188o N, 101.952o W) in the Texas High Plains. Soil at the Yoakum site was a Tremona loamy fine sand (thermic Aquic Arenic Palenstalf) with less than 1% organic matter and pH 7.2, while the soil at Halfway was a Pullman clay loam (fine, mixed, thermic Torrertic Paleustoll) with less than 1% organic matter and pH 7.7.
Weed Efficacy Studies
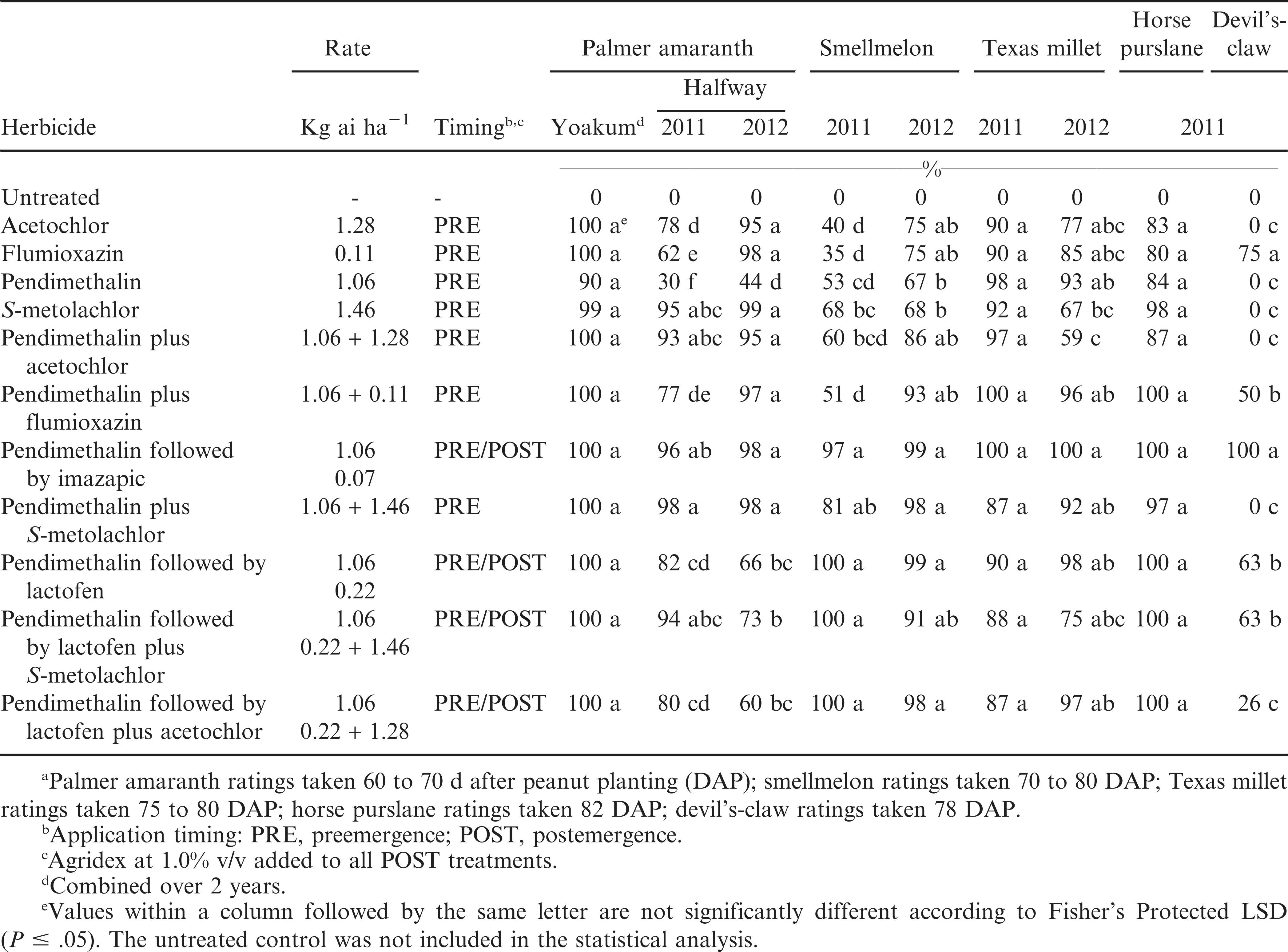
For the first weed efficacy study (Study 1), experimental design was a randomized complete block with 3 replications at Yoakum and 3 or 4 replications at the Halfway location. Plot dimensions at Yoakum were two rows (spaced 97 cm apart) by 7.9 m long and four rows (spaced 102 cm apart) by 9.5 m long at Halfway. Experimental sites contained natural infestations of devil’s-claw (at Halfway, densities of 2 to 3 plants/m2), horse purslane (at Yoakum, densities were 5 to 7 plants/m2), Palmer amaranth (densities at Yoakum were 1 to 2 plants/m2 while at Halfway populations were greater than 2 plants/m2), smellmelon (at Yoakum, denisities of 4 to 6 plants/m2), and Texas millet (at Yoakum, densities of 2 to 4 plants/m2).
Herbicide treatments included acetochlor, flumioxazin, pendimethalin, or S-metolachlor alone at 1.28, 0.11, 1.06, or 1.46 kg ai/ha, respectively, applied preemergence (PRE). Pendimethalin plus flumioxazin, pendimethalin plus S-metolachlor, and pendimethalin plus acetochlor were applied PRE in combination at the above mentioned rates. Pendimethalin applied PRE was followed by postemergence (POST) applications of either imazapic alone at 0.07 kg ai/ha, lactofen alone at 0.22 kg ai/ha, lactofen plus S-metolachlor or lactofen plus acetochlor at the above mentioned rates. Postemergence applications of imazapic, lactofen, lactofen plus S-metolachlor, or lactofen plus acetochlor included a crop oil concentrate (Agridex, Helena Chemical Co., Memphis, TN 38119) at 1% (v/v) at both locations. The pendimethalin followed by imazapic treatment was included because it is a commercial standard in many peanut growing areas of the state (Grichar 2007; 2008). An non-treated check was also included at both locations. Peanut yield was taken at the Halfway location but not Yoakum due to high weed density which made digging difficult (Buchanan et al., 1982).
For the second weed efficacy study (Study 2), conducted at Yoakum and Halfway, the experimental design was a factorial arrangement with a randomized complete block design and herbicide treatments were replicated 3 times. Treatments consisted of a factorial arrangement of PRE herbicide treatments (acetochlor at 1.26 kg/ha, flumioxazin at 0.11 kg/ha, and S-metochlor at 1.46 kg/ha) and POST herbicide (no POST herbicide or lactofen at 0.22 kg/ha). Lactofen treatments included a crop oil concentrate (Agridex, Helena Chemical Co., Memphis, TN 38119) at 1% (v/v) at both locations. A non-treated check was included at both locations. Row configurations and weed populations were similar to those mentioned in Study 1. Peanut yield was not collected at either location due to difficulty of digging plots with high weed populations as mentioned earlier (Buchanan et al., 1982).
Preemergence herbicides were applied within 24 h after peanut planting. Rainfall or irrigation followed within 7 to 14 d to activate PRE herbicides. Postemergence herbicides were applied 3 to 4 wks after planting at Yoakum or 6 to 8 wks after planting at Halfway and were applied when devil’s-claw, Palmer amaranth, Texas millet, or horse purslane were less than 42 cm in height while smellmelon was less than 30 cm in length.
Peanut Tolerance Studies
In the peanut tolerance studies (Study 3), plots were kept weed-free using a combination of hand hoeing or POST herbicides which did not impact crop growth and development. Clethodim at 0.11 kg ai/ha, lactofen at 0.22 kg/ha, or 2,4-DB at 0.28 kg ae/ha were applied with a crop oil concentrate (Agridex) to control annual grasses and broadleaf weeds, respectively.
Field studies were conducted at Yoakum in 2011 and 2012 and at Halfway in 2012. At Yoakum, three runner market type cultivars were evaluated, ‘Tamrun OL01’ (Simpson et al., 2003b) was planted both years while ‘Tamrun OL07’ (Baring et al., 2006) was planted in 2011 and ‘McCloud’ (Anonymous, 2014a; Beasley and Baldwin, 2009) was planted in 2012. At Halfway, the Spanish market type, ‘OLin’ (Simpson et al., 2003a) peanut was planted in 2012. Herbicide treatments consisted of a factorial arrangement of herbicide treatments (acetochlor at 1.26 and 2.52 kg/ha, S-metochlor at 1.46 kg/ha) and application timings [preplant incorporated (PPI), PRE, early postemergence (EPOST), and POST].
Variables for All Studies
Herbicides were applied in water using a CO2-pressurized backpack sprayer with TeeJet® 11002 DG (Spraying Systems Company, P.O. Box 7900, North Avenue, Wheaton, IL 60188) nozzles calibrated to deliver 190 L/ha at 180 kPa at Yoakum and TurboTee® 11002 nozzles in 2011 and TurboTee® 11003 calibrated to deliver 190 L/ha in 2011 or 140 L/ha in 2012 at 207 kPa at Halfway.
At Yoakum, PPI herbicides were incorporated immediately after application with a power-driven rotary tiller to a depth of approximately 6 cm while at the Halfway location herbicides were incorporated with a field cultivator, set to a depth of approximately 6 cm. Preemergence herbicides were applied within 24 h after peanut planting. Rainfall or irrigation followed within 7 to 14 d to activate PRE herbicides. Early POST herbicides were applied approximately 3 wks after peanut planting at Yoakum and 8 wks after planting at Halfway.
Peanut were planted at Yoakum using a Monosem® vacuum planter calibrated to plant 170,000 seed/ha while at Halfway a John Deere® JD 1700 Series MaxEmerge 4-row planter calibrated to plant 160,000 (2011) to 210,000 (2012) seed/ha was used. Planting depth was approximately 4 to 5 cm at both locations.
Weed control or peanut injury was estimated visually using a scale of 0 (no weed control or peanut injury) to 100 (complete weed control or plant death) (Frans et al., 1986). Weed control ratings were taken 28 to 150 d after peanut were planted depending on location while peanut injury (chlorosis/stunting) was rated 14 to 28 d after PRE herbicide application or 5 to 7 d after POST herbicide application.
Peanut yields were obtained by inverting each plot separately, air-drying in the field for 4 to 7 d, and harvesting peanut pods from each plot with a combine. Weights were recorded after soil and trash were removed from plot samples. Grade samples were determined using screens specified in USDA grading procedures (USDA, 1998).
Visual estimates of weed control and peanut injury were arcsine square root transformed prior to analysis of variance, but are expressed in their original form for clarity because the transformation did not alter interpretation. Means were compared with Fisher’s Protected LSD test at the 5% probability level (SAS Institute Inc., 2007). The non-treated check was not included in the weed control or peanut injury analysis but was included in peanut yield and grade analysis.
Results and Discussion
Peanut Injury
No peanut injury was observed with any PRE herbicides (data not shown). Many growers have reported peanut stunting when PPI or PRE applications of metolachlor have been followed by rain (Grichar et al., 1996). They also reported that POST applications of metolachlor followed by irrigation within 24 h could be effective for yellow nutsedge (Cyperus esculentus L.) control and reduce the chance of peanut injury from soil applications of metolachlor. Combinations of factors, such as herbicide rate, moisture conditions at planting, soil organic matter, and pH may affect peanut injury by chloroacetamide herbicides, such as acetochlor and S-metolachlor (Cardina and Swann, 1988; Mueller et al., 1999; Osborne et al., 1995; Wehtje et al., 1988). Cardina and Swann (1988) reported that metolachlor often delayed peanut emergence and reduced peanut growth when irrigation followed planting. However, yield loss was observed only when metolachlor was applied at rates three times higher than recommended by the manufacturer.
Flumioxazin can injure peanut, especially when the application is delayed until peanut emergence (Johnson et al., 2006; Jordan et al., 2009; Tredaway-Ducar et al., 2009). When applied soon after peanut planting (1 to 2 d), Grichar et al. (2004) reported that flumioxazin plus metolachlor combinations, under cool, wet conditions resulted in peanut stunting which was evident throughout the growing season. Injury was attributed to increased absorption of flumioxazin and metolachlor with the heavy rainfall and the slowed metabolism of these herbicides as a result of cool temperatures (Yoshida et al., 1991). Askew et al. (1999) reported that flumioxazin at 0.07 and 0.11 kg/ha injured peanut 45 and 62%, respectively, when evaluated 2 wks after peanut planting. Peanut stunting of greater than 60% was followed by as much as 35% leaflet discoloration, which was characterized as necrotic spots on foliage. Scott et al. (2001) reported that flumioxazin treated peanuts were injured 10% when evaluated 3 wks after planting. However, injury was transient and was not apparent 6 wk after planting. Flumioxazin enters plants mainly by shoot and root uptake, and plant injury can be mitigated by rapid metabolism (Yoshida et al., 1991; Anderson et al., 1994).
Leaf chlorosis and necrosis with lactofen never exceeded 15% 5 to 7 d after POST herbicide application (data not shown). This injury was evident for several wks after application on older tissue. Subsequent new growth did not show the effects of the lactofen applications and was 2% or less four wks after application (data not shown). Other studies have reported that lactofen injury is transient and subsequent growth does not show any effects of the herbicide (Grichar, 2007; 2008; Grichar and Dotray, 2011).
Weed Control
Study 1
Palmer amaranth
Since there was no treatment by year interaction at Yoakum for control of this weed, data were combined over years; at the Halfway location data are presented by year due to a treatment by year interaction.
At Yoakum, under low weed pressure, there was no difference in control between any of the herbicide treatments with control exceeding 90%. At the Halfway location in 2011, S-metolachlor alone and pendimethalin plus either S-metolachlor or acetochlor applied PRE controlled Palmer amaranth at least 93%, while pendimeathlin or flumioxazin alone provided less than 63% control (Table 1). Pendimethalin applied PRE followed by POST applications of either imazapic or lactofen plus S-metolachlor controlled Palmer amaranth at least 94%. In 2012, either pendimethalin alone or pendimethalin plus lactofen combinations provided 44 to 73% control.
Monoculture production systems and the repeated use of herbicides with the same mode of action have led to herbicide resistance in weeds, especially Palmer amaranth (Culpepper et al. 2006; Peterson, 1999; VanGessel, 2001). The use of soil-applied and POST herbicides with alternative modes of action are recommended to delay the rate of development of herbicide-resistant weed populations (Shaner et al., 1997; Ellis and Griffin, 2002; Craigmyle et al. 2013).
Smellmelon
There was a treatment by year interaction at Yoakum so data are presented separately by year. In 2011, smellmelon control with either acetochlor, flumioxazin, pendimethalin, or S-metolachlor alone was less than 70% while pendimethalin applied PRE followed by either imazapic or lactofen combinations applied POST provided at least 97% control (Table 1). In 2012, smellmelon control with acetochlor, flumioxazin, pendimethalin, or S-metolachlor alone ranged from 67 to 75% while all pendimethalin combinations provided 86 to 99% control. Grichar and Dotray (2013) reported inconsistent control of smellmelon with flumioxazin. In one year, flumioxazin at either 0.07 or 0.11 kg/ha controlled less than 55% while in another year smellmelon control was 77 and 96%, respectively.
Texas millet
Since there was a treatment by year interaction at Yoakum, each year is presented separately. Acetochlor, flumioxazin, S-metolachlor, and pendimethalin alone controlled this weed at least 90% while pendimethalin plus either flumioxazin or acetochlor applied PRE or followed by imazapic applied POST provided at least 97% control during 2011 (Table 1). In 2012, flumioxazin and pendimethalin alone controlled this weed 85 and 93%; respectively, while acetochlor and S-metolachlor provided 67 to 77% control. All pendimethalin combinations with the exception of pendimethalin plus acetochlor applied PRE or pendimethalin followed by lactofen plus S-metolachlor applied POST controlled Texas millet at least 92%.
Wilcut et al. (1995) reported that metolachlor provided little or no Texas millet control. In contrast, the dinitroaniline herbicides provide excellent control of annual grasses (Buchanan et al., 1982; Chamblee et al., 1982; Wilcut et al., 1994) including full-season control of Texas millet (Wilcut et al., 1987a; 1987b; 1995).
Horse purslane
This weed was present at Yoakum only in 2011. All herbicide systems controlled this weed at least 80% and the addition of imazapic or lactofen applied POST provided complete control (Table 1). Imazapic typically does not control horse purslane (Grichar, 2007; 2008). Grichar (2007) reported that pendimethalin, flumioxazin, imazethapyr, S-metolachlor, or dimethenamid-P provided 73% control of horse purslane when used alone.
Devil’s-claw
This weed was present at Halfway only in 2011. Pendimethalin applied PRE followed by imazapic applied POST provided acceptable control (Table 1). Combinations that included lactofen controlled this weed no better than 63%. Grichar and Dotray (2011) reported that lactofen alone failed to control devil’s-claw but a sequential application of lactofen followed by 2,4-DB controlled at least 88%.
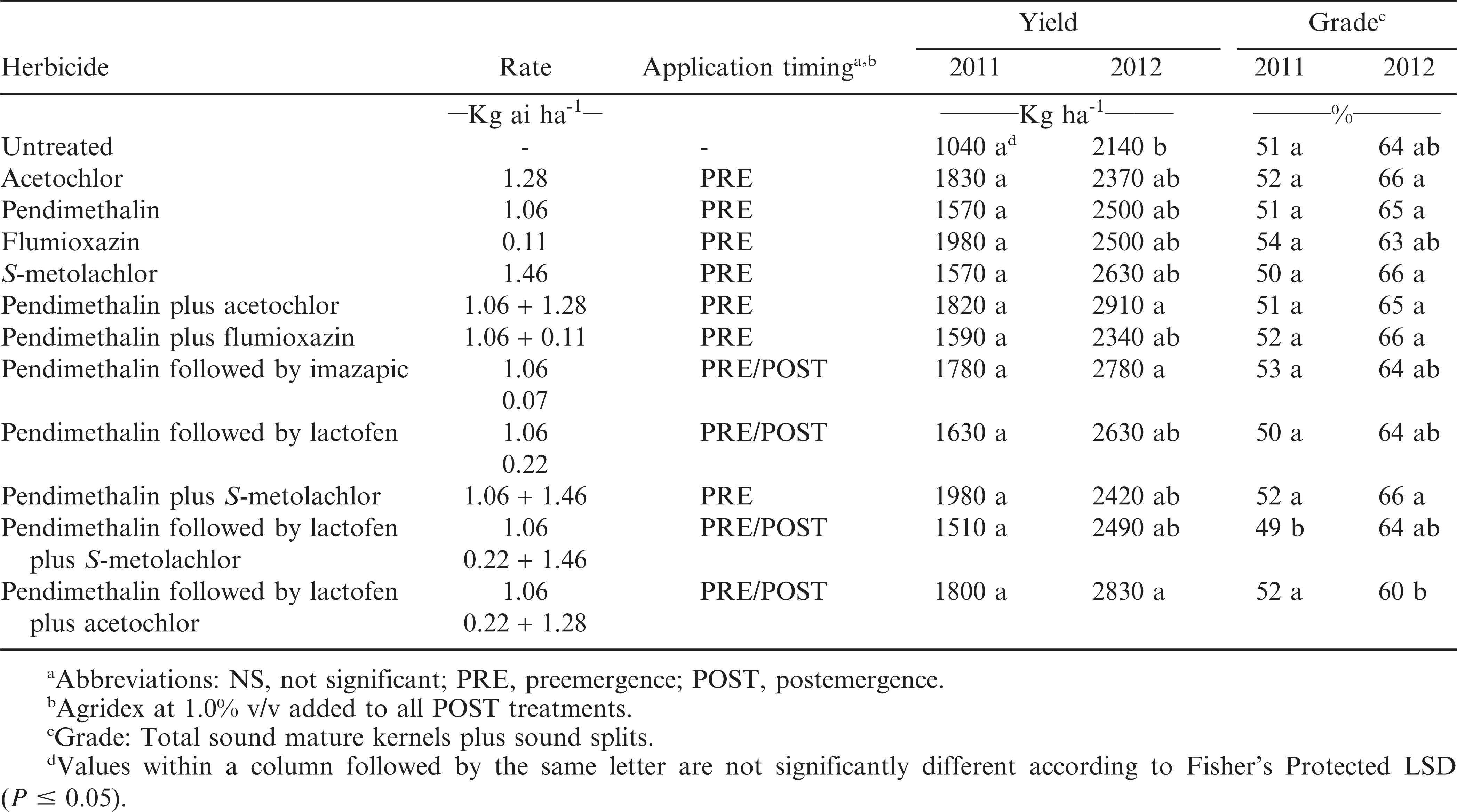
Peanut yield
In 2011, there was no difference in yield between any of the herbicide treatments and the non-treated check (Table 2). In 2012, pendimethalin plus acetochlor applied PRE produced the greatest yields while the non-treated check produced the lowest. Competition from Palmer amaranth can severely reduce peanut yield (Grichar, 2007; Grichar, 2008; Wilcut, et al. 1987a). Not only does the competition from these weeds reduce peanut yield but their extensive root system interferes with harvesting efficiency (Buchanan et al., 1982).
Peanut grade
In 2011, pendimethalin applied PRE followed by lactofen plus S-metolachlor produced the lowest grade (Table 2). In 2012, pendimethalin applied PRE followed by lactofen plus acetochlor applied POST produced the lowest grade while acetochlor and S-metochlor alone and pendimethalin plus either flumioxazin or S-metolachlor produced the highest.
Study 2
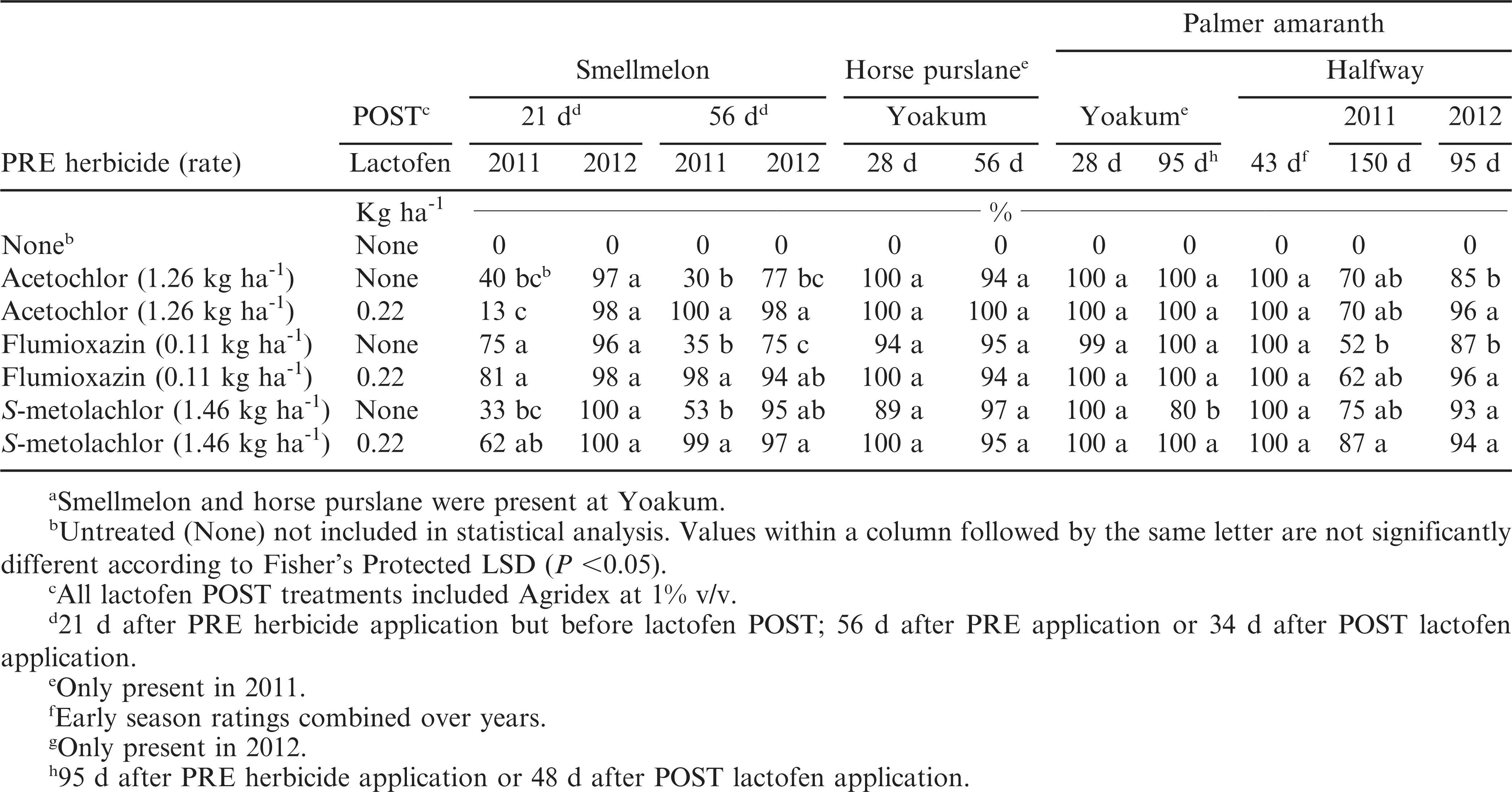
Smellmelon
There was a treatment by year interaction; therefore, each year is presented separately. When rated 21 d after PRE applications, only flumioxazin provided moderately acceptable control (75 to 81%) in 2011 while in 2012, all PRE herbicides alone controlled smellmelon at least 96% (Table 3). In 2012, 1.9 mm of rain fell within 4 d of PRE herbicide application and this accounted for the excellent control with all PRE herbicides. Since the PRE herbicides can photodegrade on the soil surface, these herbicides need to be mechanically incorporated or activated by rainfall or irrigation (Wilcut et al., 1995; Grichar et al., 1996) which explains the erratic control noted between the two years. When rated 56 d after PRE application in 2011, flumioxazin, S-metolachlor, or acetochlor alone failed to adequately control smellmelon while the addition of lactofen applied POST improved control to at least 98% for all combinations. In 2012 S-metolachlor alone provided 95% control and the addition of lactofen to the PRE herbicides provided at least 94% smellmelon control. In previous work, Grichar (2008) and Grichar and Dotray (2011) reported that smellmelon control with lactofen varied from 66 to 88% regardless of application timing. Also, Grichar and Dotray (2011) reported that lactofen controlled smellmelon 2 to 5 cm long at least 97% with either Agridex or Induce; however, smellmelon 15 to 20 cm long was controlled 96% with lactofen plus Agridex but only 82% with lactofen plus Induce. They also reported that high smellmelon densities in plots with poor smellmelon control can result in the smellmelon vines as well as the tight fibrous root system of the plant becoming intertwined with the peanut plant and digging equipment during the digging operation. As a result, many peanut pods can be stripped from the peanut plant during the digging process.
Horse purslane
This weed was present at Yoakum only in 2011. All herbicide systems controlled this weed at least 89% at either rating date (Table 3). Grichar (2007) and Grichar and Dotray (2011) reported that lactofen alone controlled horse purslane at least 88% regardless of application timing.
Palmer amaranth
This weed was present at Yoakum only in 2011 and at Halfway in both years. At Halfway, early season (43 d after PRE treatment) ratings were combined over years due a lack of treatment by year interaction while later season (95 and 150 d after PRE treatment) ratings are presented separately due to a treatment by year interaction.
At Yoakum, under low weed pressure (1 to 2 plants/m2), all herbicide systems provided nearly complete control when rated either 28 d or 56 d after PRE application with the exception of S-metolachlor alone which controlled this weed 80% (Table 3). At Halfway, when rated 43 d after PRE application, all herbicide systems provided 100% control. In 2011, when rated 150 d after PRE application, acetochlor or S-metolachlor either alone or followed by lactofen applied POST controlled Palmer amaranth 70 to 87% (Table 3). In 2012, when rated 95 d after PRE application, all systems provided at least 93% control with the exception of acetochlor or flumioxazin alone which controlled this weed 85 and 87%, respectively.
In previous work, pendimethalin alone controlled Palmer amaranth less than 42% while flumioxazin alone at 0.07 and 0.11 kg/ha provided 72 and 85% control, respectively (Grichar, 2008). In addition, lactofen applied POST following pendimethalin applied PPI improved Palmer amaranth control to 100%. Pigweed spp. can effectively be controlled with the dinitroaniline herbicides (Wilcut et al., 1994). Metolachlor applied PPI or PRE controls pigweed less consistently than dinitroaniline herbicides (Wilcut et al., 1994; 1995).
Peanut Cultivar Response
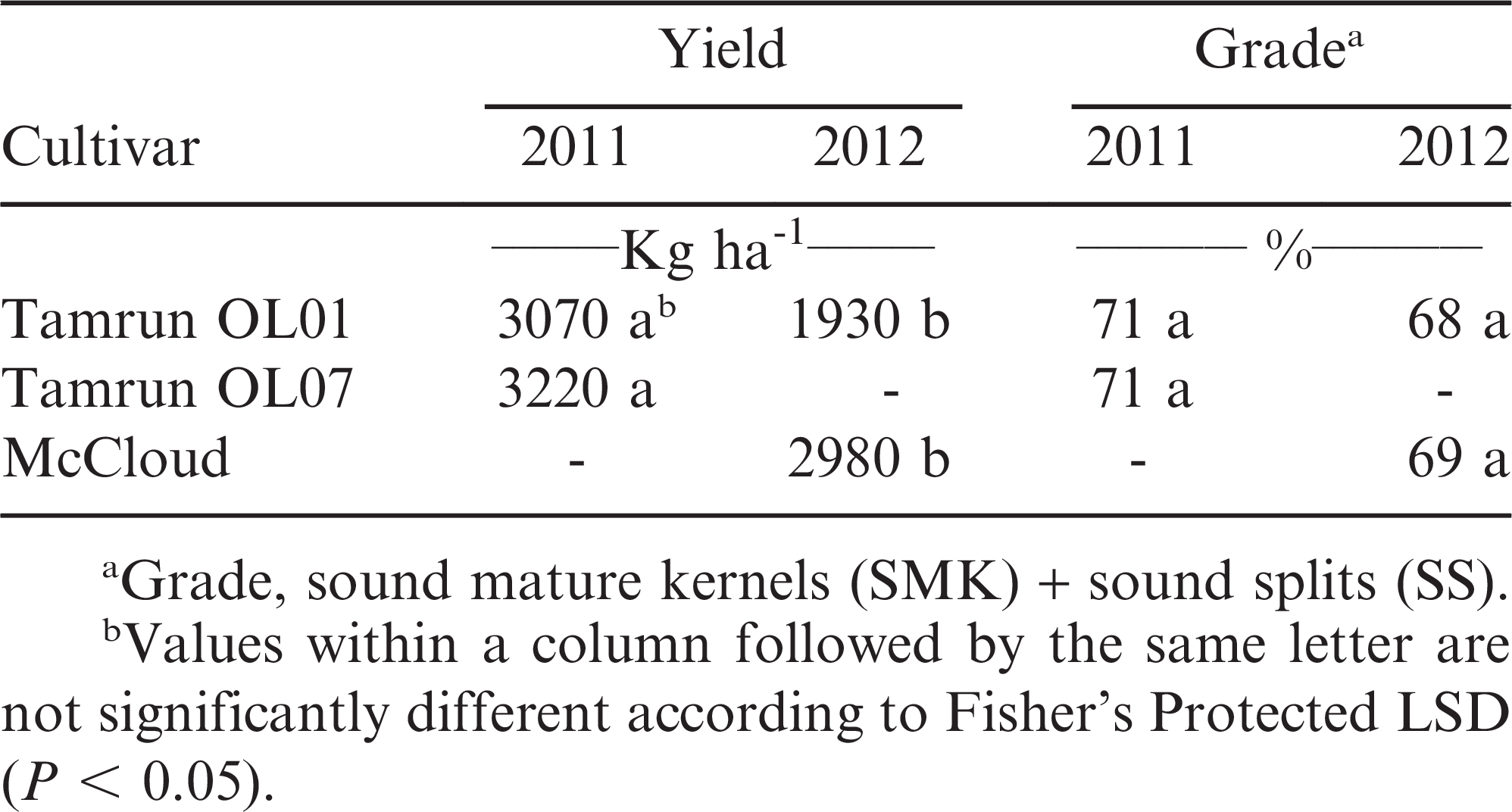
Study 3
At Yoakum, only a cultivar response was noted in one of two years. No differences were noted in 2011; however, in 2012, McCloud produced a higher yield that Tamrun OL01 (Table 4). No difference in grade of sound mature kernels plus sound splits (SMK+SS) between cultivars was noted at the Yoakum location. At Halfway, no effect of any herbicide treatment or application timing was noted on peanut yield or grade (data not shown). With the Olin variety, yields in 2011 ranged from 2500 kg/ha for the untreated to 2950 kg/ha for acetolachlor at 1.26 kg/ha applied PRE. Grades ranged from 64.2% for the non-treated control to 67.9% for acetochlor at 2.52 kg/ha applied PPI (data not shown).
Conclusions
Results from this research demonstrated that acetochlor controlled weeds similar to several herbicides currently used in peanut production without any phytotoxicity to peanut. In most instances, acetochlor is not a stand-alone herbicide and should be included in a systems approach for the most effective weed control. This herbicide will provide growers with another option in their arsenal against hard-to-control weeds. In the past, some growers in the southwest suggested that a total POST program using only imazapic or imazethapyr would be sufficient; however, a soil-applied herbicide is important in order to maintain season-long weed control, increase net returns, and avoid herbicide resistant issues (Grichar, 2008; Grichar and Dotray, 2011). Monoculture production systems and the repeated use of herbicides with similar modes of action have led to herbicide resistance in weeds (Culpepper et al., 2006; Peterson, 1999; VanGessel, 2001; Lovell et al., 1999). Since Amaranthus spp. are sensitive to ALS-inhibiting herbicides and possess characteristics that predispose them to have herbicide resistant biotypes, the use of soil-applied and POST herbicides with alternative sites of action are necessary to reduce the rate of development of herbicide-resistant weed populations (Shaner et al., 1997).
Literature Cited
Anderson R. J Norris A. E. and Hess F. D. 1994 Synthetic organic chemicals that act through the prophyrin pathway . In Duke S. O. Rebeiz C. A. eds. Porphyric Pesticides: Chemistry, Toxicity, and Pharmaceutical Applications. ACS Symposium Series 559 Washington, DC Amer. Chem. Soc . pp. 18–33 .
Anonymous 2010 Warrant herbicide; there's a new sheriff in town Monsanto Corp., St. Louis , MO, USA , 2 p .
Anonymous 2012 Degree® Herbicide Label Monsanto Corp., St. Louis , MO , 8 p.
Anonymous 2014a ‘McCloud’ Florida Foundation Seed Producers, Inc www.ffsp.net/varieties/peanut/mccloud, accessed 8 April, 2014 .
Anonymous 2014b Warrant herbicide label, Preplant, At-Planting, Preemergence and Postemergence Applications in Peanut Monsanto Corp. , St. Louis, MO 2 p.
Armel G. R Wilson H. P. Richardson R. J. and Hines T. E. 2003 Mesotrione, acetochlor, and atrazine for weed management in corn (Zea mays) Weed Technol. 17 : 284 – 290 .
Arregui M Sanchez D. Althaus R. Scotta R. and Bertolaccini I. 2010 Assessing the risk of pesticide environmental impact in several Argentinian cropping systems with a fuzzy expert indicator Pest Manage. Sci. 66 : 736 – 740 .
Askew S. D Wilcut J. W. and Cranmer J. R. 1999 Weed management in peanut (Arachis hypogaea) with flumioxazin preemergence Weed Technol. 13 : 594 – 598 .
Baring M. R Simpson C. E. Burow M. D. Black M. C. Cason J. M. Ayers J. Lopez Y. and Melouk H. A. 2006 Registration of ‘Tamrun OL07’ peanut Crop Sci. 46 : 2720 – 2721 .
Beasley J. and Baldwin J. 2009 Peanut cultivar options and descriptions http://www.uga/commodities/fieldcrops/peanuts/production/cultivardescription.html. Accessed March 21, 2014 .
Brecke B. J. and Colvin D. L. 1991 Weed management in peanuts . In Pimentel D. (ed.). CRC Handbook of Pest Management in Agriculture , 2nd ed. Boca Raton, FL CRC Press , pp. 239–251 .
Buchanan G. A Murray D. S. and Hauser E. W. 1982 Weeds and their control in peanuts , pp. 206–249 . In Pattee H. E. Young C. T. (eds.) Peanut Science and Technology Amer. Peanut Res. Educ. Soc., Inc. , Yoakum, TX .
Cardina J. and Swann C. W. 1988 Metolachlor effects on peanut growth and development Peanut Sci. 15 : 57 – 60 .
Chamblee R. W Thompson L. and Bunn T. M. 1982 Management of broadleaf signalgrass (Brachiaria platyphylla) in peanuts (Arachis hypogaea) Weed Sci. 30 : 40 – 44 .
Craigmyle B. D Ellis J. M. and Bradley K. W. 2013 Influence of herbicide programs on weed management in soybean with resistance to glufosinate and 2,4-D Weed Technol. 27 : 78 – 84 .
Culpepper A. S Grey T. L. Vencill W. K. Kichler J. M. Webster T. M. Brown S. M. York A. C. Davis J. W. and Hanna W. W. 2006 Glyphosate-resistant Palmer amaranth (Amaranthus palmeri) confirmed in Georgia Weed Sci. 54 : 620 – 626 .
Ellis J. M. and Griffin J. L. 2002 Benefits of soil-applied herbicides in glyphosate-resistant soybean (Glycine max) Weed Technol. 16 : 541 – 547 .
Frans R. R Talbert R. Marx D. and Crowley H. 1986 Experimental design and techniques for measuring and analyzing plant responses to weed control practices In: Camper N. D. (ed) Research Methods in Weed Science , 3rd edition South.Weed Sci. Soc Champaign, IL . pp. 29–46 .
Geier P. W Stahlman P. W. Regehr D. L. and Olson B. L. 2009 Preemergence herbicide efficacy and phytotoxicity in grain sorghum Weed Technol. 23 : 197 – 201 .
Grichar W. J Colburn A. E. and Baumann P. A. 1996 Yellow nutesedge (Cyperus esculentus) control in peanut (Arachis hypogaea) as influenced by method of metolachlor application Weed Technol. 10 : 278 – 281 .
Grichar W. J Besler B. A. Dotray P. A. Johnson W. C. , and Prostko E. P. 2004 Interaction of flumioxazin with dimethenamid or metolachlor in peanut (Arachis hypogaea L.) Peanut Sci. 31 : 12 – 16 .
Grichar W. J. 2007 Horse purslane (Trianthema portulacastrum), smellmelon (Cucumis melo), and Palmer amaranth (Amaranthus palmeri) control in peanut with postemergence herbicides Weed Technol. 21 : 688 – 691 .
Grichar W. J. 2008 Herbicide systems for control of horse purslane (Trianthema portulacastrum L.), smellemlon (Cucumis melo L.) and Palmer amaranth (Amaranthus palmeri S. Wats) in peanut Peanut Sci. 35 : 38 – 42 .
Grichar W. J. and Dotray P. A. 2011 Controlling weeds found in peanut with lactofen Crop Management doi: http://doi.org/10.1094/CM-2011-0912-01-RS .
Grichar W. J. and Dotray P. A. 2013 Smellmelon control and peanut response to flumioxazin and paraquat alone and in combination Peanut Sci. 40 : 135 – 141 .
Henning R. J Allison A. H. and Tripp L. D. 1982 Cultural practices In: Pattee H. E. and Young C. T. (eds.) Peanut Science and Technology Yoakum, TX ., Am. Peanut Res. and Educ. Soc , pp. 123–138 .
Johnson W. C , Prostko E. P. and Mullinix B. G. 2006 Phytotoxicity of delayed applications of flumioxazin on peanut (Arachis hypogaea) Weed Technol. 20 : 157 – 163 .
Jordan D. L Lancaster S. H. Lanier J. E. Lassiter B. R. and Johnson P. D. 2009 Peanut and eclipta (Eclipta prostrate) response to flumioxazin Weed Technol. 23 : 231 – 235 .
Lovell T S. Wax L. M. Horak M. J. and Peterson D. E. 1999 Imidazolinone and sulfonylurea resistance in a biotype of common waterhemp (Amaranthus rudis) Weed Sci. 44 : 789 – 794.
Mueller T. C Shaw D. R. and Witt W. W. 1999 Relative dissipation of acetochlor, alachlor, metolachlor, and SAN 582 from three surface soils Weed Technol. 13 : 341 – 346 .
Osborne B. T Shaw D. R. and Ratliff R. L. 1995 Response of selected soybean (Glycine max) cultivars to dimethenamid and metolachlor in hydroponic conditions Weed Technol 9 : 178–181 .
Parker D. C Simmons F. W. and Wax L. M. 2005 Fall and early preplant application timing effects on persistence and efficacy of acetamide herbicides Weed Technol. 19 : 6 – 13 .
Peterson D. E. 1999 The impact of herbicide-resistant weeds on Kansas agriculture Weed Technol. 13 : 632 – 635 .
Ross M. A. and Childs D. J. 1996 Herbicide mode of action summary Cooperative Extension Service Purdue Univ West Lafayette, IN WS-23-W. http://www.extension.purdue.edu/extmedia/ws/ws-23-w.html. Accessed March 24, 2014 .
SAS Institute Incorporated 2007 SAS/STAT User’s Guide: Statistics, Version 9.1 SAS Institute , Cary, IN .
Scott G. H Askew S. D. and Wilcut J. W. 2001 Economic evaluation of diclosulam and flumioxazin systems in peanut (Arachis hypogaea) Weed Technol. 15 : 360 – 364 .
Shaner D. L Feist D. A. and Retzinger E. J. 1997 SAMOA: one company’s approach to herbicide-resistant weed management Pesticide Sci. 51 : 367 – 370 .
Simpson C. E Baring M. R. Schubert A. M. Melouk H. A. Lopez Y. and Kirby J. S. 2003a Registration of ‘Olin’ peanut,” Crop Sci 43 : 1880 – 1881 .
Simpson C. E Baring M. R. Schubert A. M. Melouk H. A. Black M. C. Lopez Y. and Keim K. A. 2003b Registration of ‘Tamrun OL01’ peanut Crop Sci. 43 : 2298 .
Steckel L. E Sprague C. L. and Hager A. G. 2002 Common waterhemp (Amaranthus rudis) control in corn (Zea mays) with single preemergence and sequential applications of residual herbicides Weed Technol. 16 : 755 – 761 .
Tredaway-Ducar J Clewis S. B. Wilcut J. W. Jordan D. L. Brecke B. J. Grichar W. J. Johnson W. C. and Wehtje G. R. 2009 Weed management using reduced rate combinations of diclosulam, flumioxazin, and imazapic in peanut Weed Technol. 23 : 236 – 242 .
U. S. Dept of Agriculture (USDA) 1998 Inspection Instructions for Farmers’ Stock Peanuts (Revised) Agricultural Marketing Service U. S. Dept of Agric. , Washington, D. C .
VanGessel M. J. 2001 Glyphosate-resistant horseweed from Delaware Weed Sci. 49 : 453 – 459 .
Walker R. H Wells L. W. and McGuire J. A. 1989 Bristly starbur (Acanthospermum hispidum) interference in peanuts (Arachis hypogaea) Weed Sc. 37 : 196 – 200 .
Wehtje G Wilcut J. W. Hicks T. V. and McGuire J. A. 1988 Relative tolerance of peanut to alachlor and metolachlor Peanut Sci. 15 : 53 – 56 .
Wilcut J. W Wehtje G. R. and Walker R. H. 1987a Economics of weed control in peanuts (Arachis hypogaea) with herbicides and cultivations Weed Sci. 35 : 711 – 715 .
Wilcut J. W Wehtje G. R. and Patterson M. G. 1987b Economic assessment of weed control systems for peanuts (Arachis hypogaea) Weed Sci. 35 : 433 – 437 .
Wilcut J. W York A. C. and Wehtje G. R. 1994 The control and interaction of weeds in peanut (Arachis hypogaea) Rev. Weed Sci. 6 : 177 – 205 .
Wilcut J. W York A. C. Grichar W. J. and Wehtje G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea) In: Pattee H. E. and Stalker H. T. (eds.) Advances in Peanut Science Stillwater, OK ., Am. Peanut Res. and Educ. Soc , pp. 207–244 .
Young J. H Person N. K. Donald J. O. and Mayfield W. H. 1982 Harvesting, curing, and energy utilization In: Pattee H. E. and Young C. T. (eds.) Peanut Science and Technology Yoakum, TX Am. Peanut Res. and Educ. Soc . pp. 458–487 .
Yoshida R. M Sakaki R. Sato R. Haga T. Nogano E. Oshio H. and Kamoshita K. 1991 S-53482-a new N-phenyl phthalimide herbicide Proc. Brighton Crop Protect. Conf. Weeds 1 : 69 – 75 .
Notes
- First author: Texas A&M AgriLife Research, Corpus Christi, TX 78102; 2nd author: Texas A&M AgriLife Research and Extension Service, Lubbock, TX 79403; 3rd author: Monsanto Company, Llano, TX 78643 *Corresponding author: W. James Grichar, w-grichar@tamu.edu
Author Affiliations