Introduction
Annual grasses are often present in peanut (Arachis hypogaea L.) in North Carolina and can reduce yield through interference and reduced harvest efficiency (Jordan, 2011; Wilcut et al., 1995). The relatively poor competitive ability of peanut and the requirement of digging and inversion for crop harvest require season-long weed control to optimize yield (Wilcut et al., 1995). In addition to monocotyledonous weeds, including annual and perennial grasses and sedges, dicotyledonous weeds are also prevalent in peanut grown in the United States (Webster, 2009; Wilcut et al., 1995). Comprehensive herbicide programs, in combination with appropriate cultural practices, are employed to manage weeds and minimize interference and subsequent yield loss (Wilcut et al., 1995). Large crabgrass [Digitaria sanguinalis (L.) Scop.] and goosegrass (Eleusine indica L.) are among the most common weeds found in peanut grown in North Carolina (Webster, 2009). The requirement of digging and inversion as part of the peanut harvesting procedure necessitates effective season-long control of weeds, especially grasses, to optimize peanut yield (Wilcut et al., 1995). Chloroacetamide herbicides are applied primarily at planting to control annual grasses, small-seeded broadleaf weeds and sedges (Wilcut et al., 1995). These herbicides can also be applied later in the season to minimize late-season emergence of annual grasses (Jordan, 2011).
Diseases, caused by viruses, bacteria, or fungi, can reduce peanut yield when not controlled. Fungicides are applied routinely to peanut to control foliar diseases including early leaf spot (caused by Cercospora arachidicola Hori), late leaf spot (caused by Cercosporidium personatum Berk. and Curtis), and web blotch (caused by Phoma arachidicola Marasas, Pauer, and Boerema) (Brenneman et al., 1994; Culbreath et al., 2008). Fungicides are also applied to control the soil-borne diseases stem rot (caused by Sclerotium rolfsii Sacc.) and Sclerotinia blight (caused by Sclerotinia minor Jagger) (Smith et al., 1992). Although variation is noted among geographical regions, years, and environmental conditions, during a typical growing season fungicides are applied either singly or in combination beginning approximately 45 days after peanut emergence and continuing throughout the remainder of the growing season up to several weeks prior to digging and vine inversion (Sherwood et al., 1995; Shew, 2011; Smith and Littrell, 1980).
Presence of the biotic and abiotic stresses mentioned previously often occurs simultaneously during the peanut growing season, and timing of application for many agrochemicals overlap (Brandenburg, 2011; Jordan, 2011; Shew, 2011). Therefore, many growers apply herbicides and fungicides simultaneously in peanut production systems. This approach is preferable because of convenience, savings in time, reduced application costs, and freeing labor for other operations. In spite of above mentioned benefits, incompatibility can be associated with some of these mixtures of agrochemicals. Co-applying pesticides can negatively influence agrochemical efficacy and increase crop phytotoxicity (Green, 1989; Hatzios and Penner, 1985). Identifying interactions of agrochemicals is important when considering applying agrochemicals simultaneously (Green, 1989; Hatzios and Penner, 1985; Nash, 1981).
Incompatibility can occur through physicochemical interactions in the spray tank, while biological incompatibility occurs on plant surfaces or by affecting physiological processes associated with absorption, translocation, and metabolism (Houghton, 1982; Johanson and Kaldon, 1972). Various types of interactions can occur in the spray tank, on the plant surface, and/or inside the plant when agrochemicals are applied simultaneously. These interactions may occur either due to change in solution pH or due to differential absorption, translocation, or metabolism of agrochemicals in target pests (Burke and Wilcut, 2003; Culpepper et al., 1999b; Hatzios and Penner, 1985; Nalewaja et al., 1994; Rhodes and Coble, 1984). To overcome these adverse interactions, more efficacious formulations or spray adjuvants can be used (Hazen, 2000; Jordan, 1995; McMullan, 2000; Stock and Briggs, 2000). However, in some instances, these interactions can increase crop phytotoxicity and decrease pest control (Byrd and York, 1988; Franzen et al., 2003; Jordan et al., 2003; Pankey et al., 2004). Research has been conducted to define interactions of herbicides (Askew et al., 1999; Burke et al., 2004; Culpepper et al., 1998; Culpepper et al., 1999a, 1999b; Flint and Barrett, 1989; Jordan and York, 1989; Wehtje et al., 1992) and herbicides and fungicides (Jordan et al., 2003; Lancaster et al., 2005a, 2005b, 2008).
Clethodim and sethoxydim, referred to as graminicides, can be applied postemergence to control annual grasses in peanut throughout the majority of the growing season (Jordan, 2011). Efficacy of clethodim or sethoxydim has been evaluated when applied with azoxystrobin, chlorothalonil, pyraclostrobin, tebuconazole, and other standard fungicides (Jordan et al., 2003; Lancaster et al., 2005b). Previous research indicates that annual grass control may be reduced when these graminicides are applied with other pesticides (Byrd and York, 1988; Grichar et al., 2002; Holshouser and Coble, 1990; Jordan et al., 2003). Most of these studies were focused only on the biological compatibility of graminicides with other pesticides in two- or three-way mixtures (Lancaster et al., 2005b). Studies showing biological and physicochemical responses of graminicides in four or five-way mixtures are limited.
Defining interactions among agrochemicals is important in assisting growers and their advisors as they make decisions on co-application of these products. Therefore, the objectives of this research were to compare grass control by clethodim or sethoxydim when applied alone or with combinations of dimethenamid-P, S-metolachlor, 2,4-DB, and pyraclostrobin and to define changes in spray solution characteristics with these combinations.
Materials and Methods
Large crabgrass and goosegrass control with graminicides applied with other pesticides.
Research was conducted in North Carolina from 2007 through 2010 at the Central Crops Research Station located near Clayton and the Upper Coastal Plain Research Station located near Rocky Mount. Soils were a Johns sandy loam (fine-loamy over sandy, siliceous, semiactive, thermic Aquic Hapludults) at Clayton and a Goldsboro fine sandy loam (fine-loamy, siliceous, subactive, thermic Aquic Paleudults) at Rocky Mount. Efficacy of graminicides was evaluated in separate experiments in non-crop areas. Plot size was 2.4 by 5.0 m.
Treatments included all possible combinations of clethodim (Select 2EC herbicide, Valent U.S.A. Corporation, Walnut Creek, CA 94596) or sethoxydim (Poast herbicide, BASF Corporation Agricultural Products, Research Triangle Park, NC 27709) with no chloroacetamide herbicide, dimethenamid-P (Outlook, BASF Corporation, Research Triangle Park, NC 27709); or S-metolachlor (Dual Magnum, Syngenta Crop Protection, Inc., Greensboro, NC 27419); with or without 2,4-DB (Butyrac 200, Albaugh Inc., Ankeny, IA 50021); and with or without pyraclostrobin (Headline, BASF Corporation, Research Triangle Park, NC 27709). Clethodim, dimethenamid-P, pyraclostrobin, sethoxydim, S-metolachlor, and 2,4-DB were applied at 0.14, 0.84, 0.17, 0.21, 1.1, and 0.28 kg ai ha−1, respectively. Crop oil concentrate (Agri-Dex nonionic spray adjuvant, Helena Chemical Company, Collierville, TN 38137) at 1.0% (v/v) was included in all treatments. Treatments were applied in 140 L ha−1 aqueous solution from a municipal water source using CO2-pressurized backpack sprayer with flat-fan nozzles (TeeJet TP8002 flat-fan spray nozzles, Spraying Systems Co., Wheaton, IL 60189) at 275 kPa.
Large crabgrass control was evaluated during 2007, 2008, and 2009 while goosegrass control was evaluated during 2009 and 2010. Goosegrass and large crabgrass were 15 to 20 cm in height at the time of application. Visual estimates of percent weed control were recorded 21 d after treatment using a scale of 0 to 100% where 0 = no weed control and 100 = complete weed control (Frans et al., 1986). Foliar chlorosis, necrosis, and plant stunting were considered when making the visual estimates.
Physicochemical compatibility of graminicides with other pesticides.
Laboratory experiments were conducted to compare physicochemical compatibility of the graminicide combinations at rates and spray volumes used in field experiments. Deionized water at pH 6.3 was used in the laboratory experiments. Agrochemicals were mixed in the following order: emulsifiable concentrates (clethodim, pyraclostrobin, sethoxydim, S-metolachlor, and 2,4-DB), and soluble liquids (crop oil concentrate and dimethenamid-P). Graminicides solutions were prepared in a final volume of 80 ml in sterilized plastic specimen cups (Specimen cup120ml-53 ST ORG CAP, Fisher Scientific, NJ 07410) of 120 ml capacity. After mixing different agrochemicals, solution was vortexed (Vortex Genie 2, Fisher Scientific, Fairlawn, NJ 07410) immediately and examined for precipitates followed by determining pH using a portable pH meter (Oakton portable pH meter, Fisher Scientific, Fairlawn, NJ 07410). Solutions were allowed to settle for 6 h after the time of mixing, examined for precipitates, vortexed, and re-examined for precipitates followed by pH determination. Similarly, mixtures were allowed to settle for 24 h after the initial solution preparation using the same procedure. Presence or absence of precipitates were determined visually and described as Yes or No, respectively. Any visual depositions on the bottom of the specimen cup or the presence of suspended solids in the solution were considered as precipitates. Two types of precipitates, temporary and permanent, were noticed when herbicides were mixed with other agrochemicals. Temporary precipitates went back into solution upon vortexing while permanent precipitates did not go into solution after vortexing.
Statistical analysis.
The experimental design in the field was a randomized complete block and treatments were replicated four times. In the laboratory, the experimental design was a completely randomized design and the experiment was repeated with two replicates in each experiment. Data for visual estimates of percent grass control and solution pH were subjected to ANOVA using the PROC MIXED procedure of SAS (Statistical Analysis Systems version 9.2, SAS Institute Inc., Cary, NC 27513) with year and sampling intervals as random variables for field and laboratory studies, respectively. Data for visual estimates of percent weed control were transformed to the arcsine square root prior to analysis. However, non-transformed means are presented because transformation did not affect data interpretation. Means of significant main effects and interactions were separated using Tukey's pair-wise comparison tests at P = 0.05.
Results and Discussion
Large crabgrass and goosegrass control with graminicides applied with other pesticides.
Interactions of graminicide by 2,4-DB by pyraclostrobin and graminicide by chloroacetamide herbicide by pyraclostrobin were significant for goosegrass control. Large crabgrass control was affected by the interaction of graminicide by pyraclostrobin by 2,4-DB. Significant interaction of chloroacetamide herbicide by 2,4-DB was noted for both goosegrass and large crabgrass.
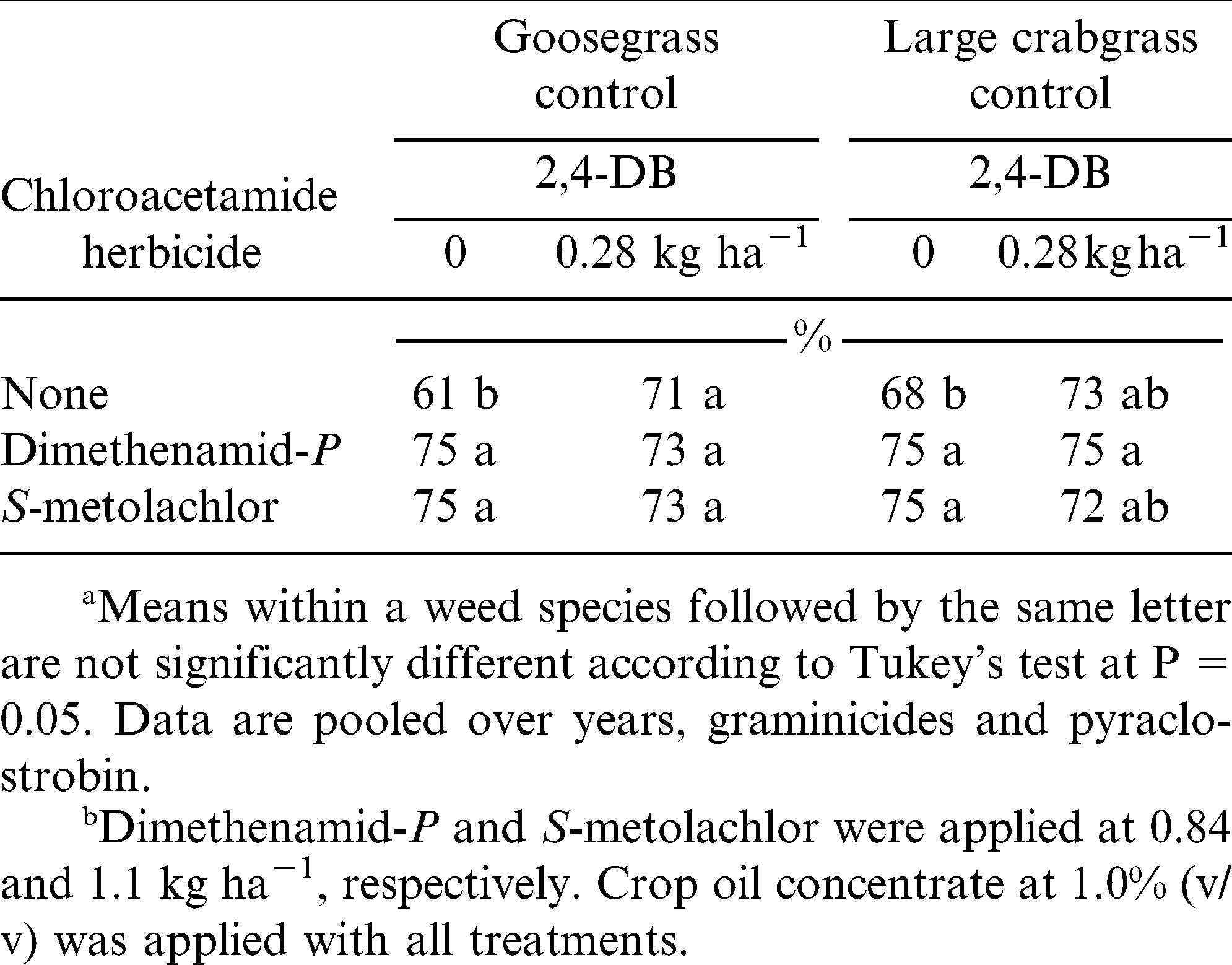
When pooled over years and chloroacetamide herbicide treatments, goosegrass control with clethodim was greater than control with sethoxydim when pyraclostrobin or 2,4-DB was not included in the mixture (Table 1). Clethodim plus pyraclostrobin controlled goosegrass less than clethodim alone when 2,4-DB was not included in the mixture. Regardless of pyraclostrobin, control by clethodim applied with 2,4-DB was similar to that by clethodim alone. Goosegrass control with sethoxydim was not negatively affected by pyraclostrobin but only when applied with 2,4-DB; in absence of 2,4-DB, pyraclostrobin did not affect goosegrass control with sethoxydim (Table 1).
When pooled over years and 2,4-DB treatment, clethodim plus pyraclostrobin controlled goosegrass less than clethodim alone when dimethenamid-P or S-metolachlor was not included in this mixture (Table 2). When compared with clethodim alone, chloroacetamide herbicides did not affect goosegrass control irrespective of pyraclostrobin treatment. In the absence of chloroacetamide herbicides, control of goosegrass with sethoxydim plus pyraclostrobin was similar to sethoxydim alone. When dimethenamid-P or S-metolachlor was applied with sethoxydim, goosegrass control was similar regardless of pyraclostrobin treatment when comparing within a chloroacetamide herbicide or with sethoxydim alone.
Combinations of graminicides with dimethenamid-P or S-metolachlor controlled goosegrass more effectively than graminicides alone irrespective of 2,4-DB (Table 3). On the other hand, addition of 2,4-DB to graminicides plus S-metolachlor provided similar control of large crabgrass as compared to graminicides alone. However, the control of large crabgrass with graminicides plus chloroacetamide herbicides was greater than graminicides alone.
Results for large crabgrass control by clethodim and sethoxydim were surprising in that pyraclostrobin reduced control by clethodim but not sethoxydim when 2,4-DB was not included (Table 4). Control of large crabgrass was greater with clethodim than with sethoxydim. In contrast, applying 2,4-DB with graminicides alone or in mixture with pyraclostrobin resulted in similar control.
Previous research indicates that clethodim was more effective in controlling broadleaf signalgrass [Brachiaria platyphylla (Griseb.) Nash] and large crabgrass than sethoxydim (Jordan et al., 1993; Lancaster et al., 2005a). As noted for goosegrass control by clethodim, co-application of clethodim with pyraclostrobin reduced large crabgrass control compared with clethodim alone. Lancaster et al. (2005a) reported that large crabgrass control was reduced in three of four experiments when sethoxydim was applied with pyraclostrobin.
Results suggesting that chloroacetamide herbicides can mitigate adverse interactions of graminicides with other pesticides or increase control over the graminicide alone were surprising. Data in the peer-reviewed literature are limited with respect to defining interactions of graminicides and chloroacetamide herbicides. The inconsistent impact of 2,4-DB on interactions of graminicides with pyraclostrobin also was surprising. In previous research (Chahal et al., 2012d), pyraclostrobin and 2,4-DB influenced efficacy of graminicides independently.
Although interactions were noted in these experiments among pesticide combinations, the magnitude of negative impact did not exceed 19% when comparing within a graminicide (Tables 1, 2, and 4). Also, the improved control in presence of chloroacetamide herbicides did not exceed 14% (Table 3). The biological significance of these interactions is difficult to quantify. None-the-less, these data do indicate that in some instances control by clethodim and sethoxydim can be negatively or positively impacted by pesticide combinations included in this study but at a relatively low level.
Physicochemical compatibility of graminicides with other pesticides.
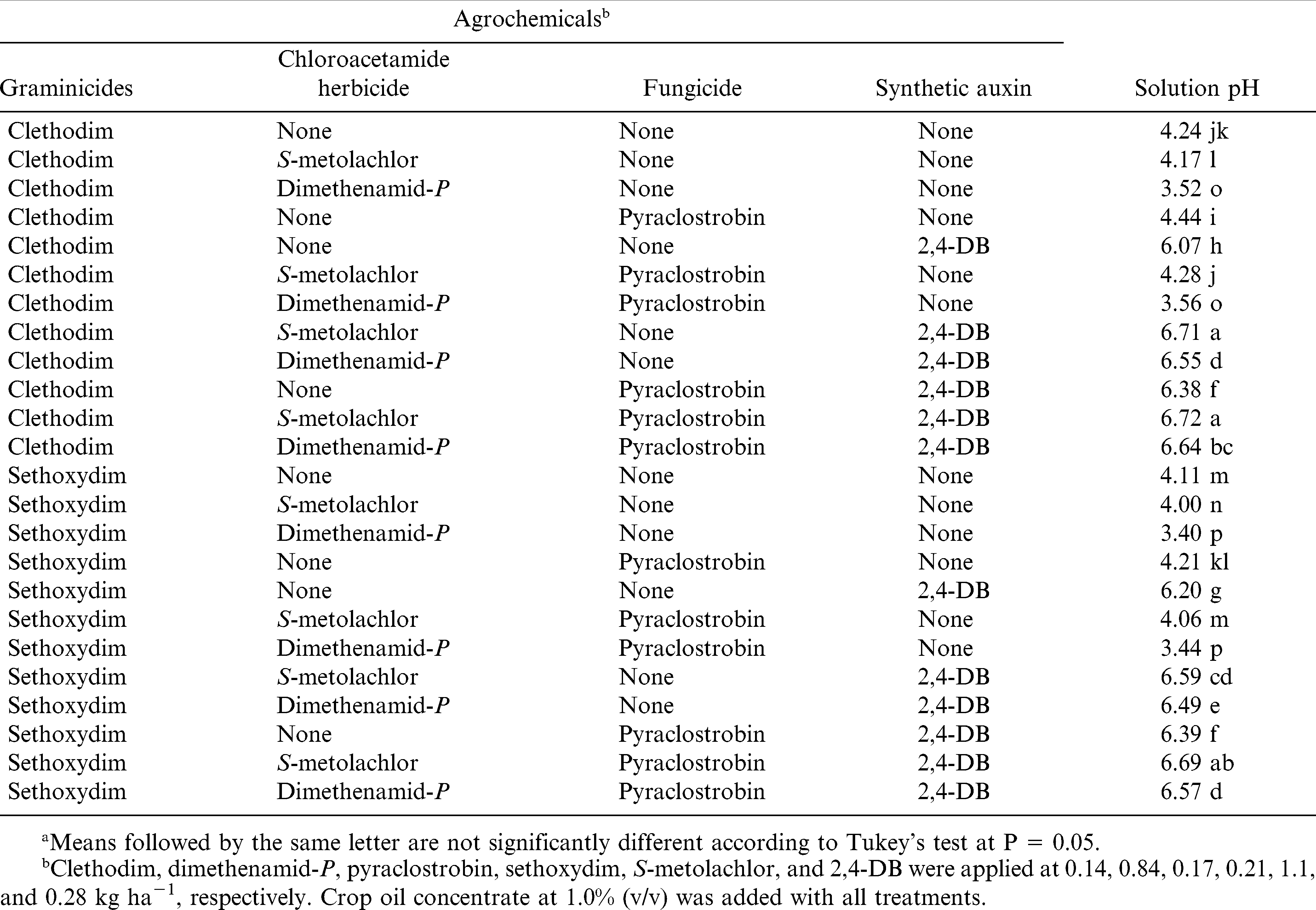
The interaction of graminicide by chloroacetamide herbicide by 2,4-DB by pyraclostrobin was significant for solution pH determined for graminicide combinations. Solution with clethodim or sethoxydim alone had pH 4.24 or 4.11, respectively, over the sampling intervals (Table 5). In the absence of 2,4-DB and pyraclostrobin, addition of dimethenamid-P and S-metolachlor to clethodim or sethoxydim solutions slightly decreased solution pH compared with solution containing clethodim or sethoxydim alone. Regardless of chloroacetamide herbicides, fungicide, and graminicides, 2,4-DB added to graminicide solution increased pH greater than 6.0. Regardless of graminicides, fungicide, and 2,4-DB, solutions containing dimethenamid-P had pH lower than solutions with S-metolachlor (Table 5).
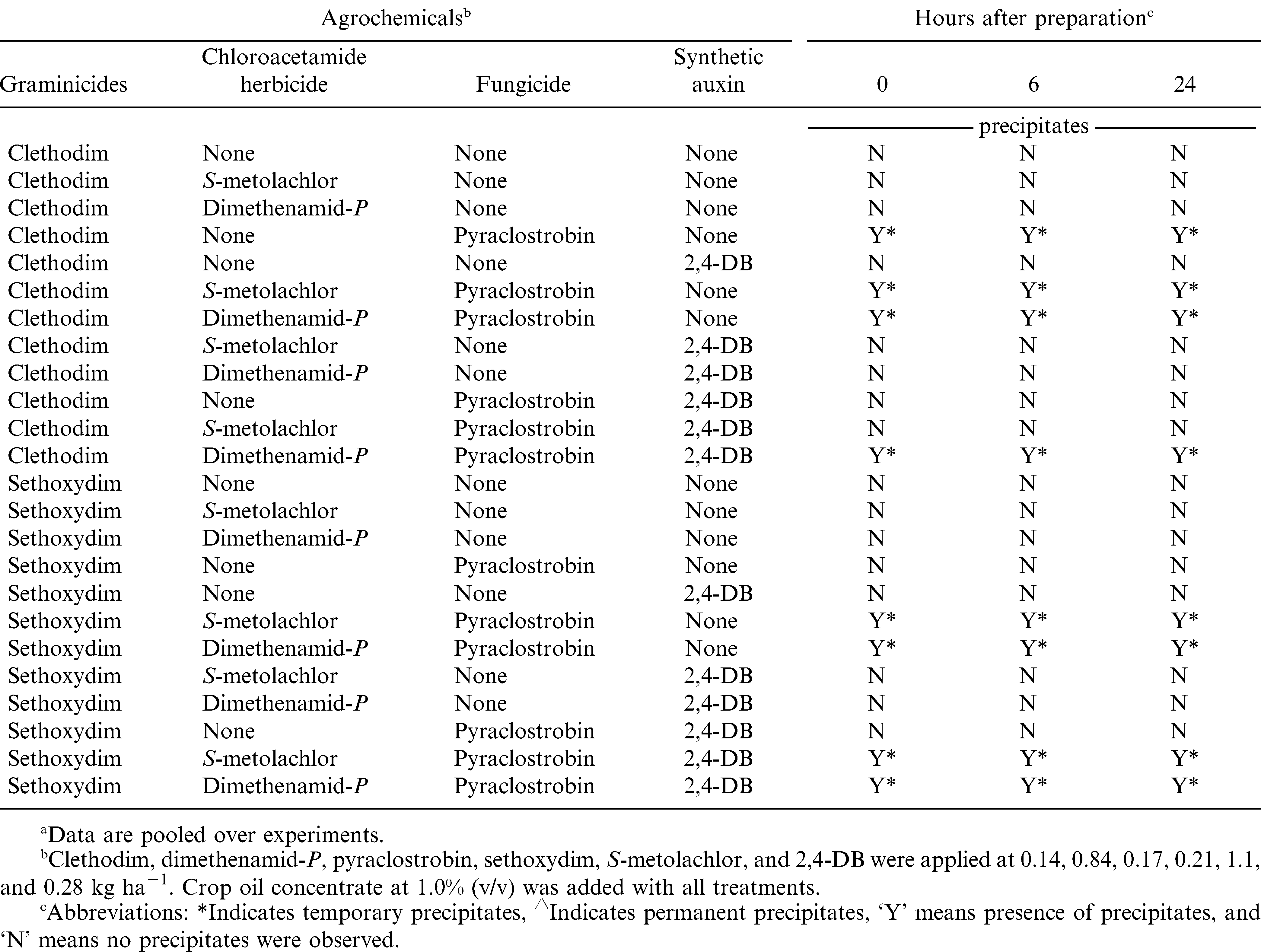
In a few instances, solution with graminicides combinations formed temporary precipitates at 0, 6, and 24 h sampling times (Table 6). For instance, a combination of clethodim plus crop oil concentrate plus pyraclostrobin with or without S-metolachlor formed temporary precipitates across sampling times but solution was reestablished after vortexing. In contrast, combination of sethoxydim plus crop oil concentrate plus pyraclostrobin did not form any precipitates regardless of sampling time.
The application of results from the pH component of this research is unknown. Although pH can impact herbicide efficacy, the impact of mixtures was relatively minor, either positively or negatively on graminicide performance. Results from the precipitate observations suggest that these pesticides can be mixed with minimal concern of permanent precipitates forming which could cause major issues with application and sprayer cleanout.
Previous research (Chahal et al., 2012a 2012b 2012c; Lancaster et al., 2005a 2005b) with three or more components in tank mixtures often used in peanut have been evaluated but did not include chloroacetamide herbicides. Results from the current research suggest that the chloroacetamide herbicides dimethenamid-P and S-metolachlor will not adversely affect control by graminicides or the previously defined interactions of graminicides with 2,4-DB or pyraclostrobin. These experiments demonstrated that pyraclostrobin consistently reduced goosegrass and large crabgrass control by clethodim. When applied alone, clethodim controlled goosegrass and large crabgrass more effectively than sethoxydim. Lack of a consistent reduction of sethoxydim activity by pyraclostrobin may have been a reflection of relatively poor control by sethoxydim regardless of co-applied pesticides. Of the herbicides used in these experiments, 2,4-DB had the least effect on graminicides. Results of physicochemical compatibility indicated that addition of clethodim, sethoxydim, dimethenamid-P, S-metolachlor and 2,4-DB changed the carrier pH dramatically. Although several clethodim and sethoxydim combinations with chloroacetamide herbicides, 2,4-DB, and fungicide formed temporary precipitates at different time intervals, pesticides and crop oil concentrate could be brought back into solution after agitation. The impact of changes in pH on efficacy was not determined in this experiment. Future research measuring efficacy and pH affects with solutions used in the field would be more informative than results reported here. Lack of permanent precipitate formation from these mixtures is important in eliminating one possible negative impact of co-application of these pesticide combinations.
Acknowledgements
This research was supported financially by the North Carolina Peanut Growers Association and the USAID Peanut CRSP (grant LAG-G-00-96-0013-00). Appreciation is expressed to Rick Seagroves, Jamie Hinton, Gary Little, Peter Eure, Bart Clewis, and staff at the Central Crops and Upper Coastal Plain Research Stations for technical support.
Literature Cited
Askew S.D Wilcut J.W and Cranmer J.R 1999 Weed management in peanut (Arachis hypogaea) with flumioxazin preemergence Weed Technol. 13 : 594 – 598 .
Brandenburg R.L 2011 Peanut insect management . pp. 88 – 100 In: 2011 Peanut Information. North Carolina Coop Ext. Ser. Publication AG-331 .
Brenneman T.B Sumner H.R Chandler L.R Hammond J.M and Culbreath A.K 1994 Effect of application techniques on performance of propiconazole for peanut disease control Peanut Sci. 21 : 134 – 138 .
Burke I.C Price A.J Wilcut J.W Jordan D.L Culpepper A.S and Ducar J.T 2004 Annual grass control in peanut (Arachis hypogaea) with clethodim and imazapic Weed Technol. 18 : 88 – 92 .
Burke I.C and Wilcut J.W 2003 Physiological basis for antagonism of clethodim by imazapic on goosegrass [Eleusine indica (L.) Gaertn.] Pestic. Biochem. Physiol. 76 : 37 – 45 .
Byrd J.D and York A.C 1988 Interactions of carbaryl and dimethoate with sethoxydim Weed Sci. 2 : 433 – 436 .
Chahal G.S Jordan D.L Shew B.B Brandenburg R.L York A.C Burton J.D and Danehower D 2012a Interactions of agrochemicals applied to peanut; Part 1: Effects on herbicides Crop Prot. 41 : 134 – 142 doi:10.1016/j.cropro.2012.05.014 .
Chahal G.S Jordan D.L Shew B.B Brandenburg R.L Burton J.D Danehower D and York A.C 2012b Interactions of agrochemicals applied to peanut; Part 2: Effects on fungicides Crop Prot. 41 : 143 – 149 doi:10.1016/j.cropro.2012.05.008 .
Chahal G.S Jordan D.L Brandenburg R.L Shew B.B Burton J.D Danehower D and York A.C 2012c Interactions of agrochemicals applied to peanut; Part 3: Effects on insecticides and prohexadione calcium Crop Prot. 41 : 150 – 157 doi:10.1016/j.cropro.2012.05.006 .
Chahal G.S Jordan D.L Shew B.S Brandenburg R.L Burton J.D Danehower D and Eure P.M 2012d Influence of selected fungicides on efficacy of clethodim and 2,4-DB Peanut Sci. 39 : 121 – 126 .
Culbreath A.K Kemerait R.C and Brenneman T. B 2008 Management of leaf spot diseases of peanut with prothioconazole applied alone or in combination with tebuconazole or trifloxystrobin Peanut Sci. 35 : 149 – 158 .
Culpepper A.S York A.C Jennings K.M and Batts R.B 1998 Interaction of bromoxynil and postemergence graminicides on large crabgrass (Digitaria sanguinalis) Weed Technol. 12 : 554 – 559 .
Culpepper A.S Jordan D.L York A.C Corbin F.T and Sheldon Y.S 1999a Influence of adjuvants and bromoxynil on absorption of clethodim Weed Technol. 13 : 536 – 541 .
Culpepper A.S York A.C Jordan D.L Corbin F.T and Sheldon Y.S 1999b Basis for antagonism in mixtures of bromoxynil plus quizalofop-P applied to yellow foxtail (Setaria glauca) Weed Technol. 13 : 515 – 519 .
Flint J.L and Barrett M.B 1989 Effects of glyphosate combinations with 2,4-D or dicamba on field bindweed (Convolvulus arvensis) Weed Sci. 37 : 12 – 18 .
Frans R.E Talbert R Marx D and Crowley H 1986 Experimental design and techniques for measuring and analyzing plant responses to weed control practices, In: Camper N. D (ed.) Research Methods in Weed Science. South. Weed Sci. Soc Champaign, IL . pp. 29 – 46 .
Franzen D.W O'Barr J.H and Zollinger R.K 2003 Interaction of a foliar application of iron HEDTA and three postemergence broadleaf herbicides with soybeans stressed from chlorosis J. Plant Nutr. 26 : 2365 – 2374 .
Green M.J 1989 Herbicide antagonism at the whole plant level Weed Technol. 3 : 217 – 226 .
Grichar W.J Besler B.A Brewer K.D and Baughman T.A 2002 Grass control in peanut (Arachis hypogaea) with clethodim and selected broadleaf herbicide combinations Peanut Sci. 29 : 85 – 88 .
Hatzios K.K and Penner D 1985 Interaction of herbicides with other agricultural chemicals in higher plants Rev. Weed Sci. 1 : 1 – 64 .
Hazen J.L 2000 Adjuvants - Terminology, classification, and chemistry Weed Technol. 14 : 773 – 784 .
Holshouser D.L and Coble H.D 1990 Compatibility of sethoxydim with five postemergence broadleaf herbicides Weed Technol. 4 : 128 – 133 .
Houghton R.D 1982 Pesticide compatibility: an overview from technical services. Pesticide Tank Mix Applications: First conference, ASTM STp 764 Wright J. F Lindsay A. D and Sawyer E (eds.) American Society for Testing and Materials. pp. 3 – 10 .
Johanson H.F and Kaldon H.E 1972 Compatibility of pesticide tank mixtures, In: Tahori A.S (ed.) Proceedings, Second International IUPAC Congress of Pesticide Chemistry. Vol. 5 Gordon and Breach : New York . pp. 485 – 522 .
Jordan D.L and York A.C 1989 Effects of ammonium micronutrients and BCH 81508 S on antagonism with sethoxydim plus bentazon mixtures Weed Technol. 3 : 450 – 454 .
Jordan D.L Frans R.E and McClelland M.R 1993 Interactions of DPX-PE350 with fluazifop-P, sethoxydim, clethodim, and quizalofop-P Weed Technol. 7 : 605 – 610 .
Jordan D.L 1995 Influence of adjuvants on the antagonism of graminicides by broadleaf herbicides Weed Technol. 9 : 741 – 747 .
Jordan D.L Culpepper A.S Grichar W.J Ducar J.T Brecke B.J and York A.C 2003 Weed control with combinations of selected fungicides and herbicides applied postemergence to peanut (Arachis hypogaea L.) Peanut Sci. 30 : 1 – 7 .
Jordan D.L 2011 Weed management in peanuts . pp. 56 – 62 In: 2011 Peanut Information. North Carolina Coop. Ext. Ser Publication AG-331 .
Lancaster S.H Jordan D.L and Johnson P.D 2008 Influence of graminicides formulation on compatibility with other pesticides Weed Technol. 22 : 580 – 583 .
Lancaster S.H Jordan D.L York A.C Burke I.C Corbin F.T Sheldon Y.S Wilcut J.W and Monks D.W 2005a Influence of selected fungicides on efficacy of clethodim and sethoxydim Weed Technol. 19 : 397 – 403 .
Lancaster S.H Jordan D.L York A.C Wilcut J.W Monks D.W and Brandenburg R.L 2005b Interactions of clethodim and sethoxydim with selected agrichemicals applied to peanut Weed Technol. 19 : 456 – 461 .
McMullan P.M 2000 Utility adjuvants Weed Technol. 14 : 792 – 797 .
Nalewaja J.D Matysiak R and Szelezniak E 1994 Sethoxydim response to spray carrier chemical properties and environment Weed Technol. 8 : 591 – 597 .
Nash R.G 1981 Phytotoxic interaction studies - techniques for evaluation and presentation of results Weed Sci. 29 : 147 – 155 .
Pankey J.H Griffin J.L Leonard B.R Miller D.K Downer R.G and Costello R.W 2004 Glyphosate-insecticide combinations effects on weed and insect control in cotton Weed Technol. 18 : 698 – 703 .
Rhodes G.N and Coble H. D 1984 Influence of bentazon on absorption and translocation of sethoxydim in goosegrass Weed Sci. 32 : 595 – 597 .
Sherwood J.L Beute M.K Dickson D.W Elliot V.J Nelson R.S Opperman C.H and Shew B.B 1995 Biological and biotechnical control in Arachis diseases, In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science Raleigh: Am. Peanut Res. and Educ. Soc . pp. 160 – 206 .
Shew B.B 2011 Peanut disease management . pp. 103 – 119 In: 2011 Peanut Information. North Carolina Coop. Ext. Ser Publication AG-331 .
Smith F.D Phipps P.M and Stipes R.J 1992 Fluazinam: A new fungicide for control of Sclerotinia blight and other soilborne pathogens of peanut Peanut Sci. 19 : 115 – 120 .
Smith D.H and Littrell R.H 1980 Management of peanut foliar diseases with fungicides Plant Dis. 64 : 356 – 361 .
Stock D and Briggs G 2000 Physicochemical properties of adjuvants: values and applications Weed Technol. 14 : 798 – 806 .
Webster T.M 2009 Weed survey- southern states Proc. South. Weed Sci. Soc. 62 : 509 – 524 .
Wehtje G Wilcut J.W and McGuire J.A 1992 Paraquat behavior as influenced by 2, 4-DB in peanut (Arachis hypogaea L.) and selected weeds Peanut Sci. 19 : 51 – 55 .
Wilcut J.W York A.C Grichar W.J and Wehtje G.R 1995 The biology and management of weeds in peanut (Arachis hypogaea L.), In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science. Stillwater: Am. Peanut Res. and Educ. Soc . pp. 207 – 224 .
Notes
- Graduate Research Assistant, Professor, and William Neal Reynolds Distinguished Professor Emeritus, respectively, Department of Crop Science, Box 7620, North Carolina State University, Raleigh, NC 27695
- William Neal Reynolds Distinguished Professor, Department of Entomology, Box 7613, North Carolina State University, Raleigh, NC 27695
- Research Assistant Professor, Department of Plant Pathology, Box 2510, North Carolina State University, Raleigh, NC 27695
- Associate Professor, Department of Horticultural Sciences, Box 7609, North Carolina State University, Raleigh, NC 27695
- Associate Professor, Department of Crop Science, Box 7620, North Carolina State University, Raleigh, NC 27695 * Corresponding author E-mail: david_jordan@ncsu.edu
Author Affiliations