Organic production has been the fastest growing agricultural sector in the U.S. for many years (Dimitri and Greene, 2002). Although organic peanut production in the U.S. is currently concentrated in the Southwest (Ruark and Shew, 2010), significant efforts to develop organic peanut production practices that are viable in the Southeast have recently been employed (Lamb et al., 2007; Cantonwine et al., 2008b; Johnson and Mullinix, 2008; Ruark and Shew, 2010). Through these efforts, stand establishment has been identified as one of the most important challenges to organic peanut production in the Southeast because the synthetic seed treatments that are ubiquitous in conventional peanut production to reduce seed and seedling diseases are not allowed in organic production. Untreated peanut seed can result in 50% fewer established plants than seed treated with synthetic seed treatments even when seed quality is high (Melouk and Backman, 1995). Poor seedling emergence can not only directly limit yield potential, but can also have an indirect impact on yield due to increased incidence of Tomato spotted wilt tospovirus (TSWV) (Branch et al., 2003), and increased weed competition (Place et al., 2010).
There are a number of biological, botanical, and mineral seed treatments that are allowable in organic production. One of the most studied is a microbial-based treatment of Bacillus subtilis, a gram-positive, endospore-forming bacterium. Strain GB03 is marketed as the seed treatment Kodiak (Bayer CropScience, Research Triangle Park, NC). The effect of B. subtilis GB03 and related strains on improving stand establishment has been variable (Elliott et al., 2001; Turner and Backman, 1991; Ruark and Shew, 2010). In a recent study, Ruark and Shew (2010) found that stands were nearly identical between seed treated with a high rate of Kodiak (2.5 g/kg seed) and untreated seed in one year, yet two times greater than untreated seed in another year. Turner and Backman (1991) found that peanut seed treated with a synthetic seed treatment and B. subtilis AF 1 improved emergence when seed were planted early and soil temperatures were low (21°C), but did not affect emergence when planting conditions were optimal.
Factors that affect peanut seed quality, such as maturity, drying and storage conditions, and soundness of seed after shelling, can have a great impact on germination rates (McLean and Sullivan, 1981; Sullivan, 1995). Bell (1969) showed that seed damaged by mechanical shelling can significantly reduce seedling emergence compared to seed that were shelled by hand. However, not all seed become damaged during the mechanical shelling process. Using a standard laboratory assay, Dey et al. (1999), demonstrated that undamaged mechanically shelled peanut seed had comparable germination as hand-shelled seed, while the damaged mechanically shelled seed where the testa had slipped or been removed had lower germination. More recently, Tarekegan et al. (2007) compared performance of cleaned mechanically shelled seed, where discolored, shriveled, and split seed were removed, to uncleaned seed under field conditions in Ethiopia. They found that in most cases, the plots planted with cleaned seed had greater seedling emergence rates and reduced post-emergence plant mortality than those with uncleaned seed.
Our objectives were to evaluate how hand-shelled or mechanically shelled seed, with or without B. subtilis seed treatment affected stand establishment, seedling emergence rates, and incidence of Aspergillus crown rot, caused by Aspergillus niger, for 15 peanut genotypes. Genotypic differences of these variables were evaluated when possible. The effect of genotype on seedling biomass, which is relevant to early season weed suppression in organic systems, was also evaluated.
Materials and Methods
Field experiments were conducted in 2007 and 2009 on organically managed research plots certified for organic production in 2007 at the Horticulture Hill Farm, part of the University of Georgia, Coastal Plain Experiment Station in Tifton, GA. The 2007 field was planted to peanut the previous year, and the 2009 field was fallow cultivated the previous year to control weeds. The fields were prepared by harrowing, and plots were prepared with a bedder in 2007 and tractor tire indentions to differentiate beds in 2009. No pesticides or irrigation were applied during the experiments, which lasted 21 days in 2007 and 24 days in 2009. Weeds were removed within the plant row by hand. The field received 11.6 cm of rain across 10 rain events in 2007 and 4.9 cm of rain from 8 rain events in 2009.
The experimental design was a three factor factorial arranged in a randomized complete block with 5 replications. There were 60 experimental treatments consisting of 15 peanut genotypes, two shelling techniques (mechanically shelled or hand-shelled), and two seed treatments [untreated or treated with B. subtilis (2.5 × 10−6 kg Kodiak per g of seed)]. The 15 runner-type genotypes included seven cultivars: Georgia Green (Branch, 1996), Georganic (Holbrook and Culbreath, 2008), AP-3 (Gorbet, 2007), C-99R (Gorbet and Shokes, 2002), Georgia-01R (Branch, 2002), Tifguard (Holbrook et al., 2008), and Tifrunner (Holbrook and Culbreath, 2007); and eight breeding lines: C11-186, C11-154-61, C12-3-114-58, C34-24-38, C34-24-69, C34-24-124, C689-6-6, and C724-19-25. Each seed lot came from seed that had been harvested from plants grown for seed production under identical conditions the previous year, and harvested at maturity. Seed were stored in the shell through the winter and shelled (mechanically or by hand) in the spring. Mechanically shelled seed were shelled using standard presizer and box shellers that are commonly used in grading procedures. Seed from both shelling treatments were cleaned to remove discolored, shriveled, or split seed. Hand shelled seed with entire or partial testa losses were also discarded, and mechanically shelled seed with entire testa losses or significant damage to cotyledons were also discarded. Treatment plots were single rows, 3.0 m long, separated by 1.0 m alleys. Thirty seed were planted by hand, 3.8 cm deep and 10 cm apart on 29 May 2007 and 2 June 2009. Wide seed spacing was used to minimize the chance of pathogen movement from seed-to-seed and plant-to-plant.
Seedling emergence was estimated as the percentage of seed resulting in an established seedling 21 days after planting (DAP) in 2007 and 17 DAP in 2009. Incidence of Aspergillus crown rot, expressed as the percentage of established seedlings exhibiting symptoms or signs, was recorded 21 DAP in 2007 and 24 DAP in 2009. Establishment rate was estimated as the percentage of the stand at the final evaluation that was emerged at 10 DAP. Seedling biomass was estimated 21 DAP in 2007 and 24 DAP in 2009 by harvesting the aerial parts of ten consecutive plants, avoiding plants with obvious Aspergillus crown rot. If there were less than ten plants in a plot, all healthy plants were harvested. Harvested plants were dried for 48 hr at 100°C, allowed to cool to room temperature in plastic bags, and weighed.
General linear model univariate analyses were performed using the statistical package SPSS version 17. When interactions were not significant, genotype, shelling treatment, and seed treatment were classified as fixed factors, and year and replication were classified as random factors. In cases where shelling treatment × seed treatment interactions were significant, all four seed treatment combinations [mechanically shelled untreated (MU), mechanically shelled B. subtilis treated (MT), hand shelled untreated (HU), and hand shelled B. subtilis treated (HT)] were classified as a fixed factor (combined seed treatment). Factor effects on percent stand were evaluated by genotype because of the inability to standardize seed lots for germination variability. Stand count was evaluated as a covariate of biomass using linear regression by year. Studentized residuals were computed to analyze treatment effects on biomass minus influences of stand differences. When the effect of genotype could be evaluated, Tukey's honestly significant difference tests (P0.05) were conducted to distinguish level differences within the factor.
Results and Discussion
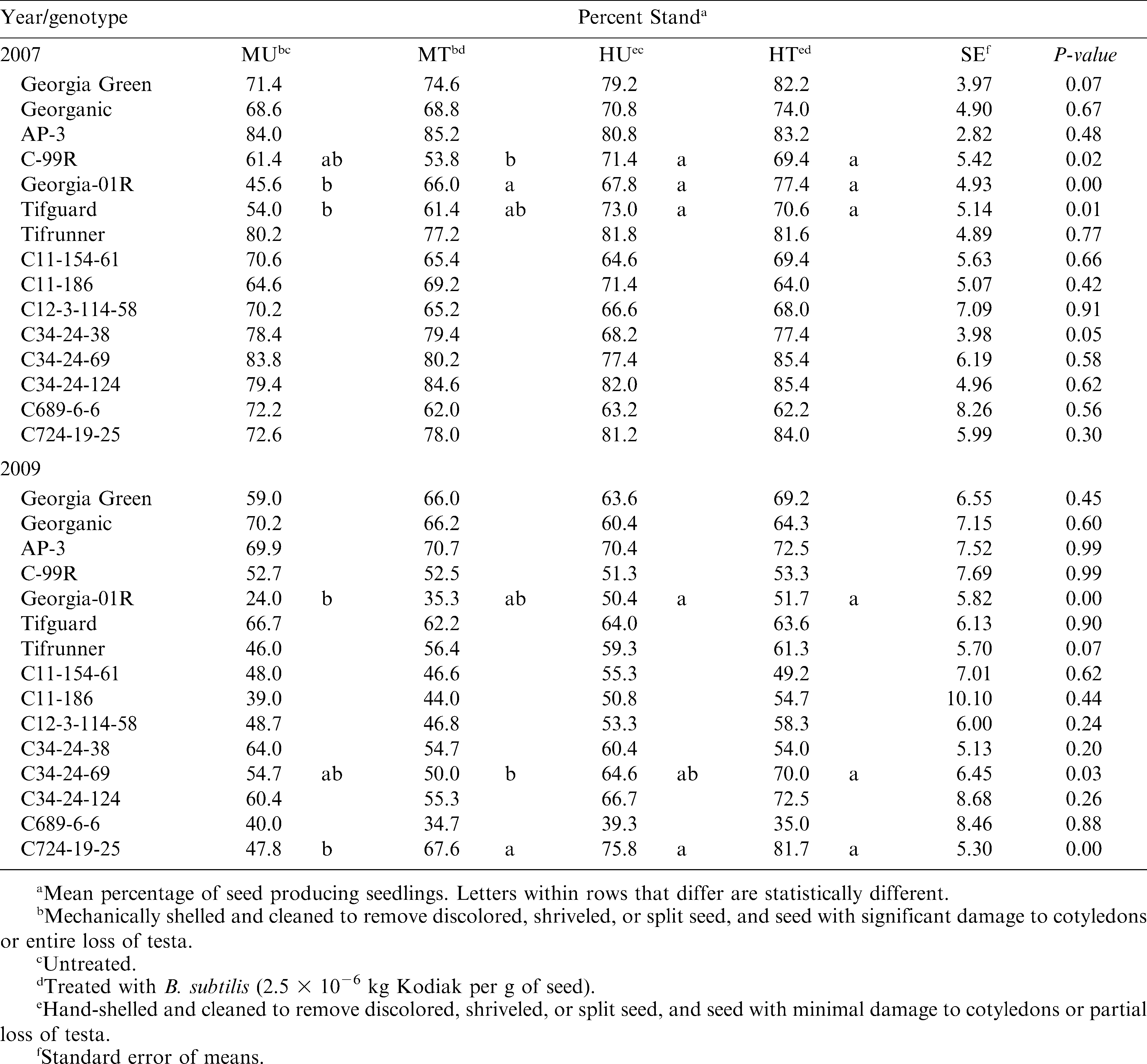
Due to significant 3- and 2-way interactions (P0.01), percent stand was evaluated using year, genotype, and combined seed treatment as fixed factors. Combined seed treatment significantly affected three genotypes each year, Georgia-01R both years, C-99R and Tifguard in 2007, and C34-24-69 and C724-19-25 in 2009 (Table 1). In all of these cases, the hand-shelled plots had significantly greater stand counts than the mechanically shelled plots for at least one of the B. subtilis seed treatment factors (untreated or treated). The application of B. subtilis as a seed treatment significantly improved stand counts for mechanically shelled Georgia-01R in 2007 and mechanically shelled C724-19-25 in 2009, but did not affect stand counts for any of the hand-shelled treatments. In both years, the genotypes that responded to hand shelling and/or B. subtilis seed treatment had relatively low stand counts in the mechanically shelled untreated plots ( 62% emergence). Of the 6 seed lots with stand counts 62% that were not significantly affected by combined seed treatment, 4 showed a numerical increase in stand count with hand-shelling and 3 showed a numerical increase with B. subtilis seed treatment (Table 1). These trends are worth noting as the power of our analysis was relatively low due to multiple factor interactions.
Where significant, reduced stand counts of mechanically shelled plots may have been due to more pre-emergence seed and seedling disease since seed with minor damage to cotyledons or partial testa loss were included in seed lots of these plots. Because the fields received rain shortly after planting each year, Rhizopus spp. and Pythium spp. are the most likely pathogen candidates for seed mortality (Melouk and Backman, 1995). The quantity and quality of seed damage to cotyledons and testa occurring in the seed lots of the mechanical shelling treatment were not evaluated, but it is possible that the gentoypes that did not respond to hand-shelling had less damage in their mechanically shelled seed than those genotypes where hand-shelling improved stands. The significant response to B. subtilis by mechanically shelled Georgia-01R in 2007 and C724-19-25 in 2009, which was not detected in the respective hand-shelled treatment, supports the possibility that differences in seed damage correlate with response to shelling treatment. Turner and Backman (1991) suggested that B. subtilis seed treatments were of greatest value to improve peanut emergence under stressful field conditions, such as low temperature or moisture. Our data suggests that this may also be true for seed stress, such as that caused by mechanical shelling. The lack of response to B. subtilis observed for the majority of the genotypes in this study might also be a result of the optimal (warm, moist) planting conditions each year.
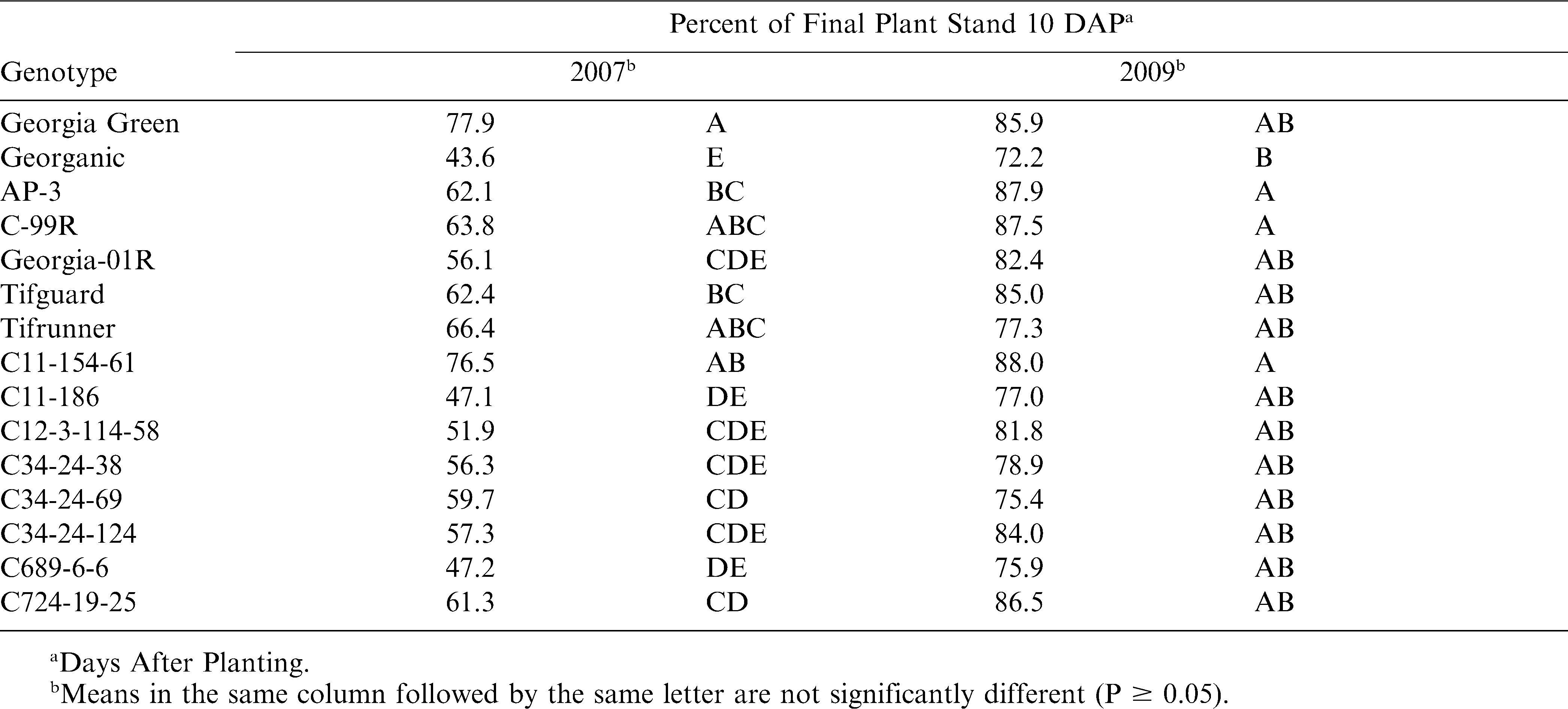
Although the seed in this study were grown and processed the same, we were not able to standardize seed lots for germination variability, and are therefore unable to make conclusions about the cause of differences observed for stand establishment across genotypes. We can however make conclusions about how the genotypes differed in their establishment rates, resistance to post-emergence crown rot, and seedling biomass. Establishment rates, the percentages of established seedlings at 17–21 DAP that had emerged by 10 DAP, were significantly affected by genotype (P0.01), and are presented by year due to a significant genotype-year interaction (P0.01) (Table 2). In 2007, Georgia Green, C11-154-61, Tifrunner, and C-99R were among the genotypes with the highest establishment rates, while Georganic was among the genotypes with the lowest establishment rates. Georganic had the lowest numerical rate again in 2009, but only C11-154-61, AP-3 and C-99R had significantly higher rates than Georganic that year. Because speed of emergence is negatively related to the length of time seed and pre-emergence seedlings are susceptible to pathogens (Melouk and Backman, 1995), emergence rates should be an important consideration for cultivar selection in organic systems. Shelling procedure and seed treatment did not significantly affect establishment rates (P0.51) or interact with genotype (P0.05).
Despite relatively low disease pressure, incidence of Aspergillus crown rot was significantly lower for seedlings from the hand shelled plots than mechanically shelled plots (P0.01). Across years, plots with mechanically shelled seed lost 3.1% of established plants to crown rot compared to 2.1% of established plants in the hand shelled treatment plots. Many of the plants affected by crown rot presented signs of A. niger on cotyledons, and perhaps the increase in incidence in the mechanically shelled plots were due to additional infection courts of damaged cotyledons during the shelling process. Crown rot was not affected by seed treatment (P = 0.97), and few significant differences were observed among genotypes each year (Table 3). C11-154-61 was among the genotypes with the greatest incidence of crown rot both years.
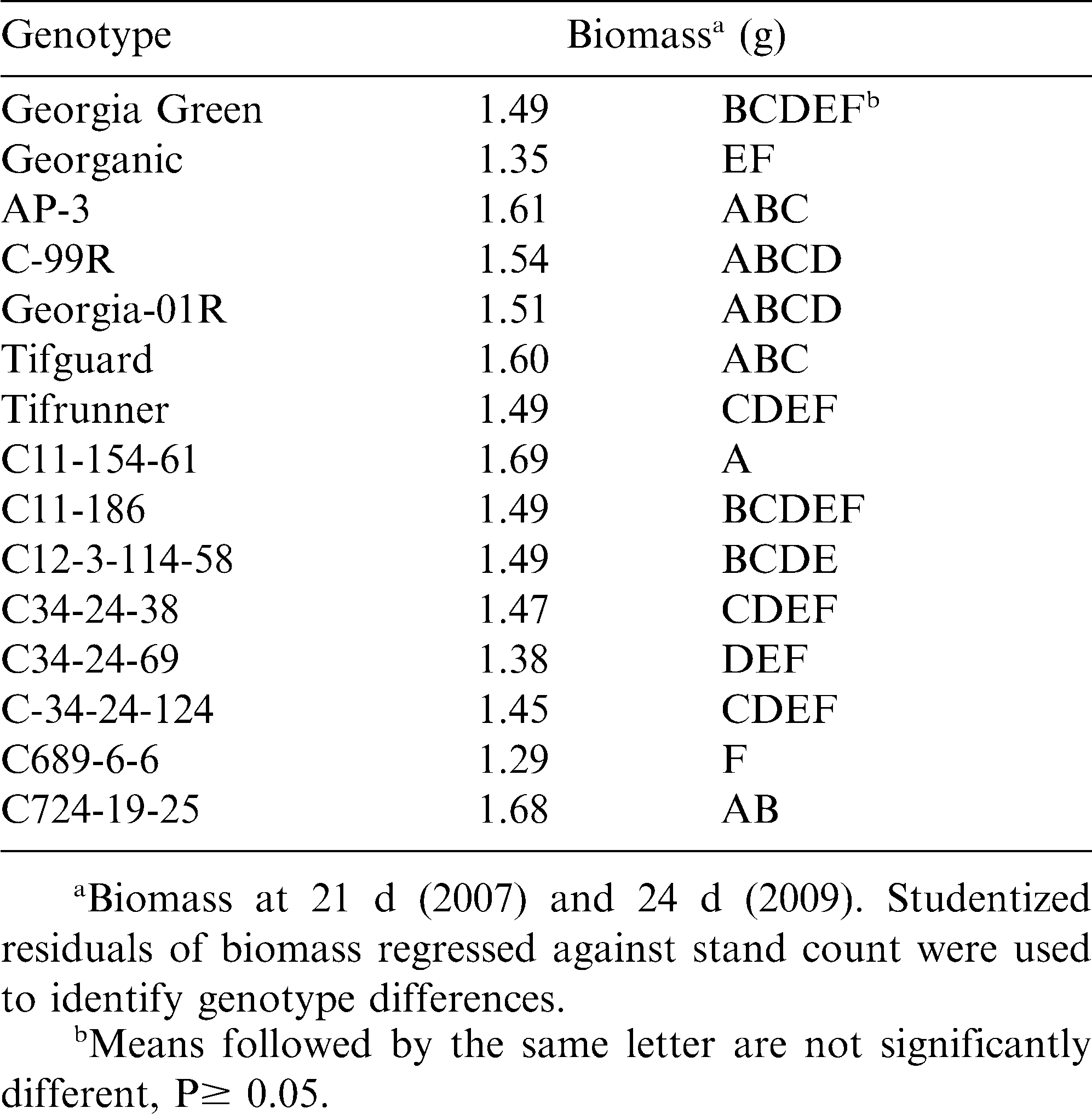
Seedling biomass was measured to better understand genotype variation in early season growth for the potential of cultivar development to help with weed management. If plants grow faster, they have the potential to shade out emerging weeds and may help reduce weed pressures. Neither B. subtilis seed treatment nor shelling procedure had an effect on biomass (P0.60), but genotype significantly affected biomass across years (P0.01). C11-154-61, C724-19-25, Tifguard, AP-3, C99R, and Georgia-01R had significantly greater biomass than Georganic, and C689-6-6 (Table 4). The relatively low biomass observed for Georganic and C689-6-6 may be related to the slow rates of establishment observed for these cultivars (Table 2).
The cultivar Georganic has many excellent qualities suitable for organic production, including high yield potential, and enhanced disease resistance to foliar pathogens, such as Cercospora arachidicola, Cercospora personatum, and TSWV (Holbrook and Culbreath, 2008; Cantonwine et al., 2006; Cantonwine et al., 2008a). As such, Georganic has been planted to organic peanut systems in Georgia in recent years. However, the slow rates of establishment and slow seedling growth, as observed in this study, have been seen in other studies (R.S. Tubbs, unpubl. data, 2009–2010) and in on-farm trials (M.A. Boudreau, unpubl. data, 2006–2008). Planting the highest quality seed, such as seed from cold storage, with a low level of damage, and high germination rate, may improve establishment rates. Cultivar evaluations for organic production must not only strive to enhance post emergence disease resistance and maximize yield potential (Branch and Culbreath, 2008), but also improve traits related to the rate and consistency of establishing a stand and resisting seedling diseases.
Summary and Conclusions
Hand shelling did not affect the rate that seedlings emerged or above ground biomass, but did reduce incidence of Aspergillus crown rot compared to the mechanical shelling treatment, and increased the percentage of seed that emerged for some of the genotypes with poor emergence rates. Although hand-shelling seed for production is not practical for most organic production fields, these results show the importance of planting seed from well-sorted seed lots where damaged seed are removed. Seed lots intended for organic production should be sorted using a lower damage threshold than those for conventional production, where seed with minor damage are of little concern due to the synthetic seed treatments. There were no negative effects observed with the use of B. subtilis, but in most cases B. subtilis did not improve the variables observed in this study, and when it did, the effect was not as great as that of hand shelling. As we saw in this study, without effective seed treatment stands have the potential to be so poor that profitable production is unrealistic. Additional research is needed to evaluate alternative seed treatments and to enhance cultivar traits involved in seedling establishment and growth.
Acknowledgements
The authors wish to thank David Edens, 2007 University of Georgia Young Scholar, and Cory Kendrick, Valdosta State University student for their assistance with this project. We would also like to thank Brad Buchanan, and technicians from the laboratories of Dr. Holbrook and Dr. Tubbs, especially Betty Tyler, Jason Golden, Katie Davis, Dylan Wann, and Corey Thompson, for their help shelling, sorting, and planting seed. This research was supported by a USDA-SARE Southern Region, Research and Education grant.
Literature Cited
Bell D. K Effects of mechanical injury, fungi, and soil temperature on peanut seed decay in soil Phytopathology 1969 64 : 241 – 243 .
Branch W. D Registration of ‘Georgia Green’ peanut Crop Sci. 1996 36 : 806 .
Branch W. D Registration of ‘Georgia-01R’ peanut Crop Sci. 2002 42 : 1750 – 1751 .
Branch W. D Baldwin J. A and Culbreath A. K Genotype × seeding rate interaction among TSWV-resistant, runner-type peanut cultivars Peanut Sci. 2003 30 : 108 – 111 .
Branch W. D and Culbreath A. K Disease and insect assessment of candidate cultivars for potential use in organic peanut production Peanut Sci. 2008 35 : 61 – 66 .
Cantonwine E. G Culbreath A. K Stevenson K. L Kemerait R. C Brenneman T. B Smith N. B and Mullinix B. G Integrated disease management of leafspot and spotted wilt of peanut Plant Dis. 2006 90 : 493 – 500 .
Cantonwine E. G Culbreath A. K Holbrook C. C and Gorbet D. W Disease Progress of Early Leaf Spot and Components of Resistance to Cercospora arachidicola and Cercospora personatum in Runner-Type Peanut Cultivars Peanut Sci. 2008a 35 : 1 – 10 .
Cantonwine E. G Culbreath A. K Shew B. B and Boudreau M. A Efficacy of organically acceptable fungicides for management of early and late leaf spot diseases on partially resistant peanut cultivars 2008b Online. Plant Health Progress doi:10.1094/PHP-2008-0317-03-RS .
Dimitri C and Greene C Recent Growth Patterns in the U.S. Organic Foods Market 2002 U.S. Department of Agriculture, Economic Research Services, Market and Trade Economics Division and Resource Economics Division. Agriculture Information Bulletin Number 77.
Dey G Mukherjee R. K and Bal S Influence of harvest and post-harvest conditions on the physiology and germination of peanut kernels Peanut Sci. 1999 26 : 64 – 68 .
Elliott M. L des Jardin E. A Batson W. E Caceres J Brannen P. M Howell C. R Benson D. M Conway K. E Rothrock C. S Schneider R. W Ownley B. H Canaday C. H Keinath A. P Huber D. M Sumner D. R Motsenbocker C. E Thaxton P. M Cubeta M. A Adams P. D Backman P. S Fajardo J Newman M. S and Pereira R. M Viability and stability of biological control agents on cotton and snap bean seeds Pest Manag. Sci. 2001 57 : 695 – 706 .
Gorbet D. W Registration of ‘AP-3’ peanut J. Plant Registrations 2007 1 : 126 – 127 .
Gorbet D. W and Shokes F. M Registration of ‘C-99R’ peanut Crop Sci. 2002 42 : 2207 .
Holbrook C. C Timper P Culbreath A. K and Kvien C. K Registration of ‘Tifguard’ peanut J. Plant Registrations 2008 2 : 92 – 94 .
Holbrook C. C and Culbreath A. K Registration of ‘Tifrunner’ peanut J. Plant Registrations 2007 1 : 124 .
Holbrook C. C and Culbreath A. K Registration of ‘Georganic’ peanut J. Plant Registrations 2008 2 : 17 .
Johnson W. C and Mullinix B. G Potential weed management systems for organic peanut production Peanut Sci. 2008 35 : 67 – 72 .
Lamb M. C Cantonwine E Sorensen R. B Nuti R. C Tillman G and Smith N. B Economics of organic versus conventional peanut and cotton Proc. Am. Peanut Res. Educ. Soc. 2007 39 : 73.
McLean D. E and Sullivan G. A Influence of cultural and harvest practices on peanut seed quality Peanut Sci. 1981 8 : 145 – 148 .
Melouk H. A and Backman P. A Management of soilborne fungal pathogens 75 – 85 In Melouk H. A and Shokes F. M editors Peanut Health Management American Phytopathological Society , St. Paul, MN 1995 .
Place G. T Reberg-Horton S. C and Jordan D. L Interaction of Cultivar, Planting Pattern, and Weed Management Tactics in Peanut Weed Science: October-December 2010, Vol. 58, No. 4, 2010 442 – 448 .
Ruark S. J and Shew B. B Evaluation of microbial, botanical, and organic treatments for control of peanut seedling diseases Plant Dis. 2010 94 : 445 – 454 .
Sullivan T. H Harvesting, storage, and quality of peanuts 23 – 31 In Melouk H. A and Shokes F. M editors Peanut Health Management American Phytopathological Society , St. Paul, MN 1995 .
Tarekegan G Sakhuja P. S Swart W. J and Tamado T Integrated management of groundnut root rot using seed quality and fungicide seed treatment Int. J. of Pest Management 2007 53 : 53 – 57 .
Turner J. T and Backman P. A Factors relating to peanut yield increases after seed treatment with Bacillus subtilis Plant Dis. 1991 75 : 347 – 353 .
Notes
- Department of Biology, Valdosta State University, Valdosta, GA 31698
- USDA-ARS, Tifton, GA 31793
- Department of Plant Pathology, University of Georgia, Tifton, GA 31793
- Department of Crop and Soil Sciences, University of Georgia, Tifton, GA 31793
- Department of Biological and Agricultural Engineering, University of Georgia, Griffin, GA 30223 *Corresponding author: Emily Cantonwine (E-mail:egcantonwine@valdosta.edu).
Author Affiliations