Introduction
Peanut is an important cash crop in Texas with production estimated at 0.4 million metric tons during 2005 and an average statewide yield at 3,850 kg/ha (Texas Agricultural Statistics Annual Bulletin, 2005). Peanut production is concentrated in the Southern High Plains of Texas (West Texas), which contributes about 70% of the state's total yield (Texas Agricultural Statistics Annual Bulletin, 2005).
Pod rot is an important disease problem on peanuts in West Texas (Wheeler et al., 2006). The disease is widespread among the peanut growing areas of the world with yield losses ranging from 5–50% (Frank, 1968; Kolte, 1984; Mercer, 1977). In the United States, pod rot is a problem in peanut growing states such as North Carolina, Oklahoma, Virginia and Texas (Filonow et al, 1985; Garren, 1964a; Hollowell et al., 1998; Wheeler et al., 2006 ). As early as in 1961, substantial losses in yield and market quality of peanuts due to pod rot was reported in Virginia (Garren, 1964a). In Oklahoma. 43% of 37 peanut fields sampled during the early 1980's had pod rot with the disease incidence ranging from 5–36.7% (Filonow et al., 1985). In North Carolina, a farm survey estimated mean incidence of pod rot at 6.6% in 1995 and 5.9% in 1996 (Hollowell et al.,1998). Most recently, pod rot was reported in 58% of the 107 sampled peanut fields in Texas Southern High Plains (Wheeler et al., 2006).
Various biotic and abiotic factors are involved in incidence and severity of pod rot. Species of Pythium and Rhizoctonia are often the most important causal organisms of pod rot (Frank, 1968; Garren, 1963; Hollowell et al., 1998; Wheeler et al., 2006). Other fungi isolated from diseased pods include Aspergillus spp., Cylindrocladium parasiticum, Fusarium spp., Macrophomina phaseoli, Rhizopus spp., Sclerotinia minor and Sclerotium rolfsii (Csinos et al., 1984; Hollowell et al., 1998; Mercer, 1977; Porter et al., 1982). Soilborne mites and plant parasitic nematodes were also been reported to play a significant role in pod rot incidence (Garcia and Mitchell, 1975; Shew and Beute, 1979). However, plant pathogens may not always be the primary cause of pod rot. Nutrient imbalance such as deficiency of calcium and excess of other competitive cations in the pods was shown to predispose the pods to pathogen infection (Csinos et al., 1984). Many studies have underlined the importance of calcium in controlling pod rot (Csinos and Gaines, 1986; Garren, 1964; Hallock and Garren,1968). However, arguments to the positive effects of calcium in controlling pod rot were also found (Filonow et al, 1988).
Resistance to Pythium pod rot has been reported in some peanut genotypes (Frank and Krikun, 1969; Garren, 1970; Godoy et al, 1984; Godoy et al., 1985; Smith et al., 1989). However, most of the current high oleic runner market types that comprise the largest percentage of acreage in West Texas are susceptible to Pythium pod rot infection (Texas Peanut Production Guide, 2001). Even though cultivars such as Tamrun 96 and Tamspan 90 have some resistance to Pythium pod rot, they are not widely cultivated in West Texas ((Texas Peanut Production Guide, 2001). Application of a calcium source such as gypsum at pegging may be helpful for pod rot control. However, West Texas soils are usually high in calcium and any additional application in the form of gypsum to control pod rot can be expensive for the grower due to high transportation costs (Texas Peanut Production Guide, 2001). Fungicides used to control pod rot (azoxystrobin, mefenoxam, flutolanil) are applied approximately 50–70 d after planting when pegs or young pods are present. While leaf spot control recommendations are to apply fungicides on a 10–14 d interval, pod rot applications are more typically 30 d apart. In West Texas, disease occurs from mid-July through September and the crop is dug in late September and through October. According to the fungicide label, two fungicide applications provide less than 60 d of protection. However, the susceptible period lasts for at least 75 d if counting from the onset of pegging (Texas Peanut Production Guide, 2001). Protection during the entire period when pegs or pods are present requires at least three fungicide applications, thereby significantly increasing production costs. A major percentage of peanut acreage is scouted for disease in West Texas and the advice of a scout is a key factor in making a decision on control including use of a fungicide (Smith et al., 1998). Hence, many producers prefer to use scouts to recommend the timing of the applications, rather than spray by a calendar date. At $20/ha in Terry and Yoakum counties (pers. comm. Texas Pest Management Association), the cost of a crop consultant in this region is considerably less than a fungicide application for pod rot control. Consultants prefer to delay the first fungicide spray as long as possible and save an application for late August or early September when the risk of pod rot incidence is greatest.
Irrigation is important for profitable peanut production in West Texas, where average annual rainfall is < 51 cm and rainfall from June through August is < 19 cm. In West Texas, 61 to 71 cm of water is needed per growing season for optimum peanut yield and the months of highest water demand are June, July, and August (Texas Peanut Production Guide, 2001). However, irrigation can increase the development of diseases by its influence on moisture and temperature (Frank and Ashri, 1985; Frank, 1967; Porter et al., 1987). Seedling diseases caused by Pythium spp. and Rhizoctonia solani are more severe under cool, wet conditions (Bateman, 1961; Klisiwicz, 1968; Mendel et al., 1995; Wright, 1957). In irrigated peanuts, diseases such as pod rot are influenced by various factors which include structural make up of pods (Godoy et al., 1985; Pettit et al., 1975); the complex of pathogens present including multiple species of Pythium and Rhizoctonia solani (Garcia and Mitchell, 1975; Hollowell et al., 1998; Wheeler et al., 2006); and the effect of fungicides that may target only part of the pathogen complex, and do not eradicate the problem (Texas Peanut Production Guide, 2001). The logical view that wetter soil equals more disease may not be an adequate hypothesis while making recommendations for controlling the disease on irrigated peanuts.
The purpose of this study was to evaluate whether there was a consistent and positive association of pod rot incidence caused by Pythium spp. alone or in association with Rhizoctonia spp., with soil moisture in irrigated peanut fields in an environment where water is not significantly limiting infection or plant growth.
Materials and Methods
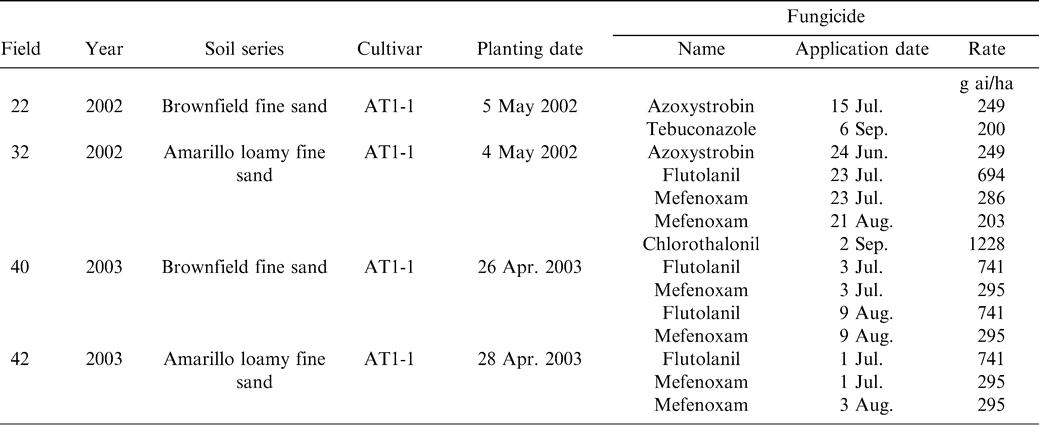
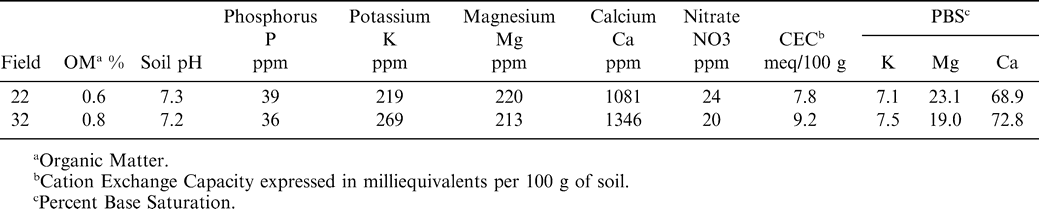
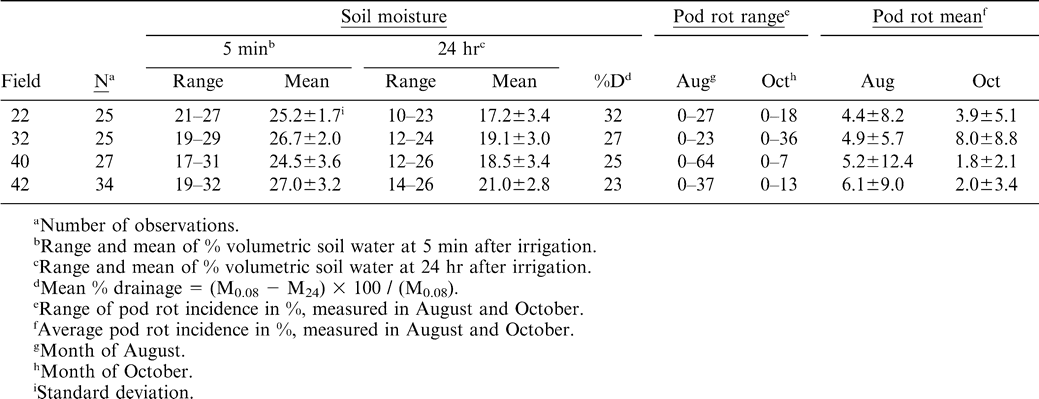
Studies were conducted in four commercial fields in Yoakum County, TX during 2002 and 2003 (Table 1). The producer practiced a four-year rotation program with wheat -watermelon-wheat-peanut. Fields were selected that had a history of pod rot in previous peanut crops. In addition, a general survey of these fields ascertained the presence of pod rot. In each year, the fields chosen were a Brownfield (Loamy, mixed, superactive, thermic Arenic Aridic Paleustalfs) fine sand or Amarillo (Fine-loamy, mixed, superactive, thermic Aridic Paleustalfs) loamy fine sand, (Table 1). The Brownfield type soils had a higher sand content and lower clay content than the Amarillo type soils. One of the four fields (field 22) appeared to have greater slope compared to other three fields. Soil analysis report from fields 22 and 32 indicated high concentrations of nutrients including calcium (Table 2). The soil pH was near neutral and organic matter content was very low (Table 2). All fields had approximately 25 ha planted to AT1-1, a runner type peanut and 25 ha that was planted to wheat in the fall and fallowed the following summer. The fields were irrigated by center-pivot, sprinkler irrigation systems.
Fungicide applications of mefenoxam (Ridomil Gold EC; Syngenta Crop Protection, Greensboro, NC) were applied by the producer to fields 32, 40, and 42 for Pythium pod rot (Table 1). Field 22, which had a Pythium incited early-season peg rot was treated with azoxystrobin (Abound FL; Syngenta Crop Protection, Greensboro, NC) (Table 1). All four fields were treated with either azoxystrobin (Abound FL; Syngenta Crop Protection, Greensboro, NC) or flutolanil (Moncut 70 DF; Gowan Company, Yuma, AZ) or both to control Rhizoctonia pod rot (Table 1).
Moisture readings were taken by following the sprinkler, rather than independent of the sprinkler position, since rainfall totals for July and August 2002 and 2003 were low. Soil moisture was measured in August using a theta probe (Model: ML2x, Delta-T Devices Ltd., Cambridge, UK) at 5 min and 24 hr after irrigating particular areas in the field. Theta probe uses a simplified impedance measuring system to determine the soil water content (Gaskin and Miller, 1996). The device consists of an oscillator, fixed impedance section of coaxial transmission line and a sensing probe whose impedance depend on the dielectric constant of the surrounding soil. An amplitude difference occurs when there is a difference in the impedance between the probe and coaxial transmission, following a signal propagation from the oscillator. Amplitude difference in turn provides the apparent dielectric constant value, which is directly proportional to the volumetric water content. The linear relationship is depicted as θv = (√ϵ − a0)/a1 where, θv = volumetric soil water content, expressed as a ratio (m3/m3), ϵ = apparent dielectric constant and a0 and a1 are constants that change with soil type. The method used in the theta probe compares well with results from the standard neutron probe under simulated field conditions, confirming its acceptance in general irrigation research (Gaskin and Miller, 1996).
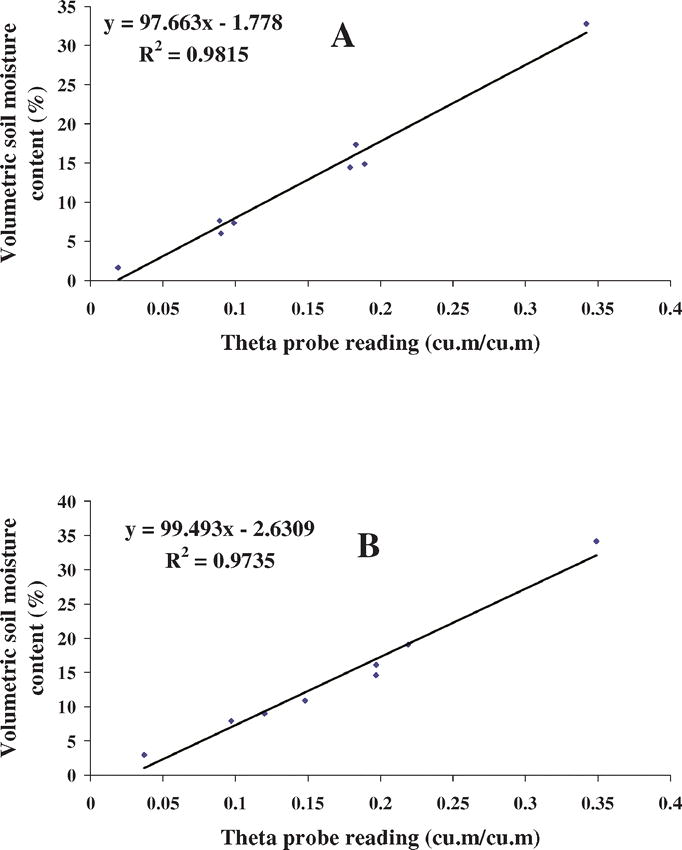
Soil-specific calibrations were conducted on eight undisturbed soil samples collected from each field (Fig. 1A and 1B). Theta probe measurements were taken by inserting the sensing head into the soil core sample. Soil wet weights were recorded and the sample was oven dried at 105 C for 24 hr. Gravimetric soil water content (%), dry bulk density (gms/cm3) and volumetric soil moisture content (%) were determined (Gardner, 1986; Hartge and Blake, 1986). Estimated volumetric soil moisture content (%) was regressed against the theta probe measurements (m3/m3) to derive the following linear model: Volumetric soil water content (%) = a × theta probe reading (m3/m3) + b, where a = slope and b = intercept.
The sensing head of the theta probe was inserted into the soil such that the sensing element measured volumetric moisture content (m3/m3) over a 6–12 cm depth. The typical peanut fruiting depth is 6–8 cm. The difference between the moisture readings taken at 5 min and 24 hr was calculated for each field and expressed as mean percentage drainage.
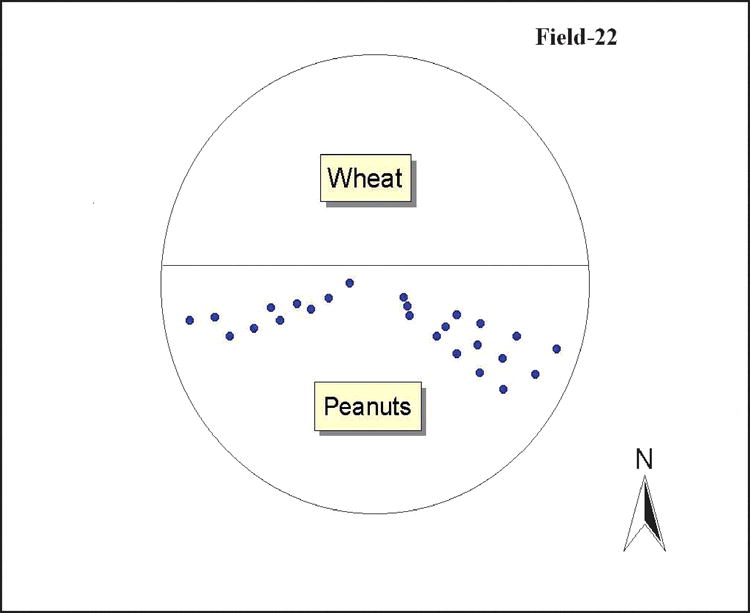
The total number of sampling sites varied for each field (Table 3). Sampling points were along the line (radius) of the sprinkler irrigation system with 3–4 hr intervals, to allow the sprinkler to proceed farther around the field (Fig. 2). Gaps in the field where no data were collected occurred when peanuts were irrigated during the night hours. After the 24 hr soil moisture reading, three plant samples were collected at each moisture measuring point and percent incidence of pod rot was estimated by weighing pods with symptoms of pod rot divided by total weight of pods from three plants. An additional disease assessment was made in October at the same locations as in August.
Eight pods with characteristic symptoms of pod rot were collected at harvest from each sampling point and were assayed for the presence of fungi. Pods were washed for several minutes and then dried for 2–3 hr at room temperature. Each pod lesion was split into two pieces, one plated out on water agar and the other on a Pythium selective medium (Lewis and Filonow, 1990). Mycelium was transferred to potato dextrose agar to obtain pure cultures for identification. Mean percentage of total pods that were positive for either Pythium or Rhizoctonia was calculated for each sample. Product-moment correlations were calculated between the soil moisture readings and disease incidence that occurred during August and October, using the CORR procedure from SAS (SAS Institute Inc., Cary, NC).
The Yoakum County weather station within the Texas Tech-West Texas mesonet system (latitude = 33.2281, longitude = −102.8394) was used to monitor soil temperature and rainfall events. Rainfall totals would not necessarily be accurate since the station was some distance from field sites, but provides a good indication of seasonal trends.
Results
The weather in Yoakum County was dry during July and August in 2002 and 2003 and hence water applied to the peanut fields through irrigation was the primary source of moisture during this period. Immediately after irrigation, soil moisture content differences within a field were as low as 6% (field 22) to as great as 14% (field 40) (Table 3). Overall, the incidence of pod rot was higher in August compared to the end of the growing season except in field 32, where increased disease incidence was observed at harvest (Table 3).
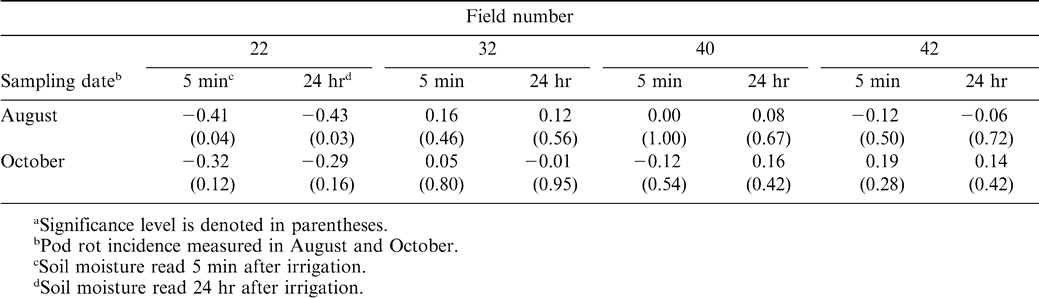
Pod rot incidence in August was negatively correlated with soil moisture at 5 min (r = −0.41, P = 0.04, n = 25) and 24 hr (r = −0.43, P = 0.03, n = 25) in field 22 (Table 3). For the same field, pod rot incidence in October was not significantly correlated with soil moisture readings (Table 4). This field had substantial topographical changes, and the highest incidence of pod rot was found on the steepest slope of a hill, rather than in the low area of the field. No significant correlations were found between pod rot incidence and soil moisture in other fields (Table 4).
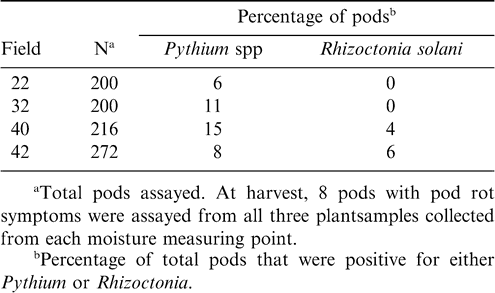
Infected pods showed symptoms of either black watery rot or brown decay and seeds inside severely rotted pods were completely decayed. In all four fields, Pythium spp. was the most common pathogen isolated from rotted pods, followed by Rhizoctonia spp. In fields 22 and 32, only Pythium spp. were found whereas both Pythium spp. and Rhizoctonia solani were found in fields 40 and 42 (Table 5). Sclerotium rolfsii was isolated from two sampling points in field 22 (Data not presented).
Discussion
The negative correlation between soil moisture and pod rot incidence observed in field 22 is contrary to that predicted by controlled experiments on the effect of Pythium spp. and soil matric potential, where the severity of infection was greater under wet soil conditions (Bhatti and Kraft, 1992; Hering et al., 1987; Pankhurst et al., 1995; Pieczarka and Abawi, 1978; Stanghellini et al., 1983). High soil moisture content favors the development of oospores and prevented the formation of sporangia by Pythium ultimum (Bainbridge, 1970). However, it is important to note that soil moisture and temperature optimal for infection can vary among different species of Pythium (Biesbrock and Hendrix, 1970). Root necrosis of peach caused by P. vexans was severe at periodically saturated soil water regimes, but that P. irregulare was unaffected by soil water conditions (Biesbrock and Hendrix, 1970). Klisiwicz (1968) found that seedling disease of sunflower caused by Pythium ultimum and P. irregulare was worse at cool temperatures, but that P. aphanidermatum was more damaging at higher temperatures. Pythium myriotylum is also more of a warm temperature species (Gay, 1969). A recent survey has shown that Pythium myriotylum, P. ultimum, and P. irregulare were the most common species isolated from peanut pods in West Texas (Wheeler et al., 2006). However, field 22, which had such an unusual relationship between soil moisture and disease, had an unknown species of Pythium. Isolates were collected and tested from this field on two occasions for species identification, but no sexual structures formed.
Soil moisture was measured at two intervals (5 min and 24 hr after irrigation) to assess the drainage condition of the fields. Soil moisture immediately after irrigation is subject to loss by gravitational forces and moisture retention is heavily dependent on soil texture, nature of the soil horizonation, recency of irrigation event, and landscape differences (Cassel and Nielsen, 1986). Field elevation changes, irrigation nozzle spacing, nozzle types, and proximity to the center pivot of the irrigation system may also affect the quantity of water reaching an area of the field (Kranz, 1988). The percentage drainage was relatively high in field 22 compared to the other three fields. Field 22 had obvious slopes and low-lying areas that were not seen in other fields, which could have resulted in a high runoff rate, surface movement of applied water within the field and reduced infiltration.
Even though Pythium spp. were isolated more frequently than Rhizoctonia in all the fields, the overall percentage of positive isolations was low. It is not known whether fungicide applications made during the season for pod rot control or soil factors such as pH have affected the mean percent recovery of Pythium spp. and Rhizoctonia spp. The frequency of Pythium spp. and Rhizoctonia spp. and the percentage of disease caused by each of these pathogens may change from year to year (Hollowell et al., 1998; Wheeler et al., 2006). Pod rot was not associated with any above ground symptoms or root infection. In all the fields except field 32, estimated pod rot incidence was higher during August compared to the estimates made near the end of the growing season. It is not known why pod rot levels were high in field 32, since it was treated both in July and August with mefenoxam (Table 1).
Pod rot was aggregated in the sampled fields. An equation developed based on mean disease incidence and variance has estimated that the number of samples required to adequately estimate pod rot (coefficient of variation = 0.5) was 42, 12 and 7 when fields had 1, 5 or 10% pod rot, respectively (Wheeler et al., unpubl.data, 2006). Results from the current study suggest that samples taken from the wettest areas of a field were not more likely to have pod rot than samples taken from drier areas. Therefore, scouting program for pod rot should not be based on site-specific soil moisture content. Instead, scouting should be done throughout the field, without bias toward soil moisture. Inadequate sampling may result in wrong estimation of the disease. Site-specific treatment of wet areas with fungicides for management of pod rot is also not suggested. When fields are sampled with inadequate intensity, recommendations for controlling pod rot including fungicide applications should be based on other factors such as field history and plant maturity rather than based on field scouting.
Acknowledgements
We sincerely thank the Western Peanut Growers Association for funding this project; and Mr. Jerry Hartman, owner of JA Farms, Plains, TX for his assistance throughout our study in his peanut fields.
Literature Cited
Bainbridge A. 1970 Sporulation by Pythium ultimum at various soil moisture tensions. Trans. Br. Mycol. Soc 55 : 485 – 488 .
Bateman D. F. 1961 The effect of soil moisture upon development of poinsettia root rots. Phytopathology 51 : 445 – 451 .
Bhatti M. A. and Kraft J. M. 1992 Influence of soil moisture on root rot and wilt of chickpea. Plant Dis 76 : 1259 – 1262 .
Biesbrock J. A. and Hendrix F. F. 1970 Influence of soil water and temperature on root necrosis of peach caused by Pythium spp. Phytopathology 60 : 880 – 882 .
Cassel D. K. and Nielsen D. R. 1986 Field capacity and available water capacity. 901 – 926 In Methods of Soil Analysis: Physical and Mineralogical Methods. 2nd ed Soil Science Society of America, Inc Madison, WI .
Csinos A. S. and Gaines T. P. 1986 Peanut pod rot complex: a geocarposphere nutrient imbalance. Plant Dis 70 : 525 – 529 .
Csinos A. S. , Gaines T. P. , and Walker M. E. 1984 Involvement of nutrition and fungi in the peanut pod rot complex. Plant Dis 68 : 61 – 65 .
Filonow A. B. , Melouk H. A. , and Martin M. 1985 Effect of calcium sulfate on pod rot of peanut c.v ‘Early Bunch’ (Abstr.) Proc. Am. Peanut Res. Educ. Soc 17 : 44 .
Filonow A. B. , Melouk H. A. , Martin M. , and Sherwood J. 1988 Effect of calcium sulfate on pod rot of peanut. Plant Disease 72 : 589 – 593 .
Frank Z. R. 1967 Effect of irrigation procedure on Pythium rot of groundnut pods. Plant Dis. Rep 51 : 414 – 416 .
Frank Z. R. 1968 Pythium pod rot of peanut. Phytopathology 58 : 542 – 543 .
Frank Z. R. and Ashri A. 1985 Pythium pod rot reduction in peanuts, as affected by breeding for resistance and by disease level. Phytoparasitica 13 : 41 – 45 .
Frank Z. R. and Kirkun J. 1969 Evaluation of peanut (Arachis hypogaea) varieties for Verticillium resistance. Plant Dis. Rep 53 : 744 – 756 .
Garcia R. and Mitchell D. J. 1975 Synergistic interactions of Pythium myriotylum with Fusarium solani and Meloidogyne arenaria in pod rot of peanut. Phytopathology 65 : 832 – 833 .
Gardner W. H. 1986 Water Content. 493 – 544 In Methods of Soil Analysis: Physical and Mineralogical Methods. 2nd ed Soil Science Society of America, Inc Madison, WI .
Garren K. H. 1963 Evidence of two different pathogens of peanut pod rot. Phytopathology 53 : 746 .
Garren K. H. 1964 Landplaster and soil rot of peanut pods in Virginia. Plant Dis. Rep 48 : 707 – 709 .
Garren K. H. 1964a Recent developments in research on peanut pod rot. Proc. 3rd Nat'l. Peanut Research Conf. Auburn, AL 20 – 27 .
Garren K. H. 1970 Rhizoctonia solani vs. Pythium myriotylum as pathogens of peanut pod rot breakdown. Plant Dis. Rep 54 : 804 – 843 .
Gaskin G. J. and Miller J. D. 1996 Measurement of soil water content using a simplified impedance measuring technique. J. Agric. Eng. Res 63 : 153 – 160 .
Gay J. D. 1969 Effects of temperature and moisture on snap bean damping off caused by three isolates of Pythium myriotylum. Plant Dis. Rep 53 : 707 – 709 .
Godoy R. , Smith O. D. , and Boswell T. E. 1984 Evaluation of six peanut genotypes for pod rot resistance. Peanut Sci 11 : 49 – 52 .
Godoy R. , Smith O. D. , Taber R. A. , and Pettit R. E. 1985 Anatomical traits associated with pod rot resistance in peanut. Peanut Sci 12 : 77 – 82 .
Hallock D. L. and Garren K. H. 1968 Pod breakdown, yield, and grade of Virginia-type peanuts as affected by Ca, Mg, and K sulfates. Agron. J 60 : 253 – 257 .
Hartge K. H. and Blake G. R. 1986 Bulk Density. 363 – 375 In Methods of Soil Analysis: Physical and Mineralogical Methods. 2nd ed Soil Science Society of America, Inc Madison, WI .
Hering T. F. , Cook R. J. , and Tang W. 1987 Infection of wheat embryos by Pythium species during seed germination and the influence of seed age and soil matric potential. Phytopathology 77 : 1104 – 1108 .
Hollowell J. E. , Shew B. B. , Beute M. K. , and Abad Z. G. 1998 Occurrence of pod rot pathogens in peanuts grown in North Carolina. Plant Dis 82 : 1345 – 1349 .
Klisiwicz J. M. 1968 Relation of Pythium spp. to root rot and damping-off of safflower. Phytopathology 58 : 1384 – 1386 .
Kolte S. J. 1984 Peanut diseases. 53 – 56 In Diseases of annual edible oilseed crops, vol. 1 CRC Press, Inc Boca Raton, FL .
Kranz W. 1988 Flow Control Devices for Center Pivot Irrigation Systems. File G88-888-A Cooperative Extension, University of Nebraska Lincoln, NE .
Lewis P. I. and Filonow A. B. 1990 Reaction of peanut cultivars to Pythium pod rot and their influence on populations of Pythium spp. in soil. Peanut Sci 17 : 90 – 95 .
Mendel H. H. , Huang H. C. , Kozub G. C. , and Barr D. J. S. 1995 Effect of soil moisture and temperature on seedling emergence and incidence of Pythium damping-off in safflower (Carthamus tinctorius L.). Canad. J. Plant Sci 75 : 505 – 509.
Mercer P. C. 1977 A pod rot of peanuts in Malawi. Plant Dis. Rep 61 : 51 – 55 .
Pankhurst C. E. , McDonald H. J. , and Hawke B. G. 1995 Influence of soil water matric potential on the control of Pythium root infection of wheat with metalaxyl in two contrasting soils of South Australia. Aust. J. Exp. Agric 35 : 603 – 610 .
Porter D. M. , Smith D. H. , and Rodriguez-Kabana R. 1982 Peanut plant diseases. 326 – 410 In Patee H. E. and Young C. T. Peanut Science and Technology American Peanut Res. and Ed. Soc., Inc Yoakum, TX .
Porter D. M. , Wright F. S. , and Powell N. L. 1987 Effects of sprinkler irrigation on peanut diseases in Virginia. Plant Dis 71 : 512 – 515 .
Pieczarka D. J. and Abawi G. S. 1978 Influence of soil water potential and temperature on severity of Pythium root rot of snap beans. Phytopathology 68 : 766 – 772 .
Pettit R. E. , Taber R. A. , and Smith O. D. 1975 Structural features of peanut pods: Arachis hypogaea cultivars. Proc. Amer. Peanut Res. And Educ. Assoc 7 : 91 (abstr).
Shew H. D. and Beute M. K. 1979 Evidence for the involvement of soilborne mites in Pythium pod rot of peanut. Phytopathology 69 : 204 – 207 .
Smith D. T. , New M. G. , and Criswell J. T. 1998 Pests, Pesticide Use Management Practices in the Southwestern Peanut Industry. National Agricultural Pesticide Impact Assessment Program USDA/CSREES and the Southern Regional Office. Technical report 98-08, Texas AM University, College Station, and Oklahoma State University, Stillwater.
Smith O. D. , Boswell T. E. , Grichar W. J. , and Simpson C. E. 1989 Reaction of select peanut (Arachis hypogaea L.) lines to southern stem rot and Pythium pod rot under varied disease pressure. Peanut Sci 16 : 9 – 14 .
Stanghellini M. E. , Stowell L. J. , Kronland W. C. , and von Bretzel P. 1983 Distribution of Pythium aphanidermatum in rhizosphere soil and factors affecting expression of the absolute inoculum potential. Phytopathology 73 : 1463 – 1466 .
Texas Agricultural Statistics Annual Bulletin-2005 Texas Agricultural Statistics Service Austin, TX .
Texas Peanut Production Guide 2001 23 – 29 Texas Cooperative Extension Fact Sheet B-1514.
Wheeler T. A. , Howell C. R. , Cotton J. , and Porter D. 2006 Pythium species associated with pod rot on west Texas peanut and in vitro sensitivity of isolates to mefenoxam and azoxystrobin. Peanut Sci 30 : 9 – 13 .
Wright E. 1957 Influence of temperature and moisture on damping-off of American and Siberian elm, black locust, and desertwillow. Phytopathology 47 : 658 – 662 .
Notes
- First and second authors: Res. Agron. , USDA-ARS and Agric. Res. Statistician; Coastal Plain Exp. Sta., Tifton, GA 31793-0748. Third author: President and CEO, Hebert Green AgroEcology; Asheville, NC 28801. [^]
- This research was supported by the Texas State Precision Agriculture Initiative, Western Peanut Growers Association, Texas Agricultural Experiment Station, and West Texas AM University. [^]
- Texas Agricultural Experiment Station, Lubbock, TX 79403 [^]
- Division of Agriculture, WTAMU, Canyon, TX 79016 [^]
- Texas Cooperative Extension, Lubbock, TX 79403 [^] *Corresponding author: V. Choppakatla (email: VijayC@BioSafeSystems.com).
Author Affiliations