Introduction
Effective weed control is essential to optimize pod yield and economic return of peanut (Wilcut et al., 1995). A range of herbicides can be applied PPI, PRE, and POST to control grasses, broadleaf weeds, and sedges (Wilcut et al., 1995). Diclosulam and flumioxazin are applied PRE on significant peanut hectares to control broadleaf weeds. Acifluorfen and paraquat are applied POST alone or with bentazon to control emerged weeds, and imazapic is applied POST to control broadleaf weeds, sedges, and escaped grasses. Imazapic also provides residual weed control.
Southern corn rootworm is one of the most important insect pests in peanut (Brandenburg, 2007). Clorpyrifos is often applied as a granular treatment over the peanut row at pegging to control this pest (Brandenburg, 2007). Although chlorpyrifos is effective in controlling southern corn rootworm, under relatively dry conditions chlorpyrifos applied in this manner can elevate populations of two-spotted spider mites (Tetranycychus urticae Koch) (Barbour and Brandenburg, 1995). A risk advisory was developed to assist practitioners in selecting fields that are at risk of damage from southern corn rootworm (Herbert et al., 1997; 2004). This approach can minimize two-spotted spider mite outbreaks by defining the likelihood of damage from southern corn rootworm so that practitioners do not apply chlorpyrifos in fields that are not conducive for southern corn rootworm development and are prone to two-spotted spider mites outbreaks. A more convenient application method would be preferable if efficacy is not reduced compared with application at pegging.
Herbert and Malone (2005) reported that chlorpyrifos incorporated prior to planting controls southern corn rootworm as effectively as application at pegging. Most growers apply pendimethalin prior to planting in conventional tillage systems, and applying chlorpyrifos with pendimethalin would be more convenient than making pegging applications of chlorpyrifos. Although not substantiated, it is plausible that application of chlorpyrifos prior to planting may minimize late-season outbreaks of two-spotted spider mites that are often attributed to pegging applications of chlorpyrifos (Barbour and Brandenburg, 1995).
Peanut response and weed control with herbicides can be affected by a variety of environmental and edaphic factors (Wilcut et al., 1995). Interactions of herbicides with other agrichemicals can affect crop response and efficacy of herbicides (Lancaster et al., 2005). Defining these interactions is important in optimizing pesticide performance. Corn (Zea mays L.) injury from nicosulfuron increased when chlorpyrifos was applied in the seed furrow at planting in some but not all experiments (Bailey and Kapusta, 1994; Kapusta, 1994; Morton et al., 1994; Rahman and James, 1993). Corn injury by nicosulfuron was exacerbated more by terbufos than chlorpyrifos (Bailey and Kapusta, 1994; Morton et al., 1994). While diclosulam, imazapic, and nicosulfuron are from different herbicide families, these herbicides affect acetolactate synthase in susceptible plants. Allen and Snipes (1995) reported that cotton (Gossypium hirsutum L.) injury was greater when pyrithiobac, an herbicide that inhibits acetolactate synthase in sensitive plants, was co-applied with chlorpyrifos.
Determining if chlorpyrifos increases herbicide injury potential is important in determining if this application method is an effective alternative to current recommendations of application of chlorpyrifos at pegging. Therefore, the objectives of this research were to 1) determine if visual peanut injury, pod yield, and market grade characteristics from PRE and POST herbicides is affected by chlorpyrifos application with pendimethalin prior to planting and 2) determine if PPI application of chlorpyrifos is as effective in controlling southern corn rootworm as application at pegging.
Materials and Methods
General Procedures
Two separate experiments were conducted in North Carolina during 2004, 2005, and 2006 at the Upper Coastal Plain Research Station located near Rocky Mount on a Goldsboro loamy sand soil (fine-loamy, siliceous, thermic, Typic Paleudults) under non-irrigated conditions. Organic matter ranged from 1.2 to 1.9% and soil pH ranged from 5.9 to 6.1. Experiments were conducted in conventionally prepared, raised seedbeds. Plot size was four rows spaced 91 cm apart by 12 m. Seeds of the cultivar VA 98R were placed 5 to 8 cm deep depending on soil moisture, and granular aldicarb (Temik, Bayer Crop Science, Research Triangle Park, NC) at 1.1 kg ai/ha was applied in the seed furrow prior to seed drop. The in-row plant population was 13 plants/m. Planting dates were 15 May 2004, 25 May 2005, and 12 May 2006.
With the exception of herbicide treatments and application of chlorpyrifos for southern corn rootworm control, production and pest management practices were held constant over the entire experiment and were based on Cooperative Extension recommendations for the region (Brandenburg, 2007; Jordan, 2007a, 2007b; Shew, 2007). Herbicides were applied using a CO2-pressurized backpack sprayer calibrated to deliver 140 L/ha using regular flat fan nozzles (8002 regular flat fan nozzles, Spraying Systems Company, Wheaton, IL).
Pendimethalin at 1.1 kg ai/ha was applied over the entire test area after the field was disked twice and then incorporated with two passes of a field cultivar traveling in opposite directions to a depth of 5 to 8 cm. The field was then bedded with an in-row subsoiler and approximately 8 cm of the top of the bed was leveled to allow planting. Escaped broadleaf weeds were removed by hand. Clethodim (Select herbicide, Valent USA Corp., Walnut Creek, CA) at 140 g ai/ha and crop oil concentrate (Agri-Dex crop oil concentrate, Helena Chemical Company, Memphis, TN) at 1.0% (v/v) were applied POST in late July of all years to control escaped grasses.
Visual estimates of percent peanut injury were recorded 3 weeks after planting (WAP) (PRE herbicide experiment) or 2 weeks after POST herbicide application (WAT) (POST herbicide experiment) using a scale of 0 to 100% where 0 = no injury and 100 = plant death. Foliar chlorosis, necrosis, and plant stunting were considered when making the visual ratings. Peanut pods were dug and vines inverted based on pod mesocarp color from a composite sample of four plants collected from the no-PRE herbicide or no-POST herbicide control treated with chlorpyrifos PPI (Sholar et al., 1995; Williams and Drexler, 1981). Peanut was harvested 4 to 7 days after digging and dried to final moisture of 8%. One hundred pods from each plot were removed at harvest to determine scarification caused by southern corn rootworm feeding. Percentages of ELK, FP, and TSMK were determined using Federal-State Inspection Service criteria (USDA, 1998).
Preemergence Herbicides
Treatments included a factorial arrangement of two levels of chlorpyrifos application method (PPI or pegging) and three levels of PRE herbicide (S-metalochlor, S-metalochlor plus diclosulam, S-metalochlor plus flumioxazin). Chlorpyrifos (Lorsban 4EC insecticide, Dow ArgroSciences, Indianapolis, IN) was applied at 2.2 kg ai/ha mixed with pendimethalin prior to planting as described previously or applied at pegging at 2.2 kg/ha on a 29 cm band over the row (Lorsban 15G insecticide, Dow AgroSciences, Indianapolis, IN). Diclosulam (Strongarm herbicide, Dow AgroSciences, Indianapolis, IN), flumioxazin (Valor SX herbicide, Valent USA Corp., Walnut Creek, CA), and S-metalochlor (Dual Magnum herbicide, Syngenta Crop Protection, Greensboro, NC) were applied at 24, 70, and 1100 g ai/ha, respectively. A no-chlorpyrifos and a no-PRE herbicide control was included.
Postemergence Herbicides
Treatments included a factorial arrangement of two levels of chlorpyrifos application method (PPI or pegging) and four levels of POST herbicide (acifluorfen, acifluorfen plus bentazon, imazapic, paraquat plus bentazon). Chlorpyrifos was applied PPI or at pegging as described previously. Acifluorfen (Ultra Blazer herbicide, BASF Corp., Research Triangle Park, NC), acifluorfen plus bentazon (Storm herbicide, BASF Corp., Research Triangle Park, NC), imazapic, (Cadre herbicide, BASF Corp., Research Park, NC), and paraquat (Gramoxone MAX herbicide, Syngenta Crop Protection, Greensboro, NC) plus bentazon (Basagran herbicide, BASF Corp., Research Triangle Park, NC) were applied at 560, 320 plus 1100, 70, and 140 plus 560 g ai/ha, respectively. A nonionic surfactant (Induce nonionic surfactant, Helena Chemical Company, Memphis, TN) was applied at 0.25% (v/v) with acifluorfen, acifluorfen plus bentazon, and imazapic or at 0.125% (v/v) with paraquat plus bentazon. A no-chlorpyrifos and a no-POST herbicide control was included.
Experimental Design and Statistical Analyses
The design for both PRE and POST herbicide experiments was a randomized complete block with four replications. Data for visual estimates of peanut injury, pod yield, the percentage of pods with scarring caused by southern corn rootworm feeding, %ELK, %TSMK, and %FP were subjected to analyses of variance for each experiment using appropriate error terms for fixed and random effects (Tables 1 and 2, Carmer et al., 1989). Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
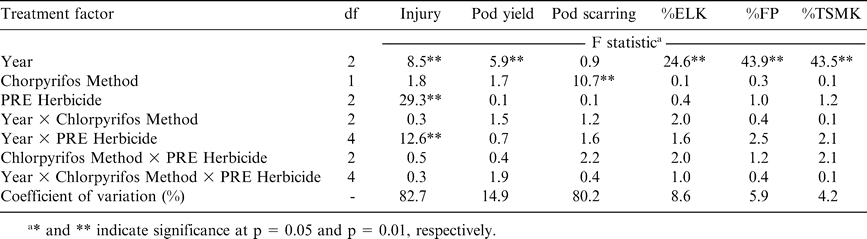
Analysis of variance for peanut injury 3 weeks after planting, pod yield, the percentage of pods with scarring caused by southern corn rootworm feeding, and percentages of extra large kernels (%ELK), fancy pods (%FP), and total sound mature kernels (%TSMK) following application of chlorpyrifos and selected preemergence (PRE) herbicides.
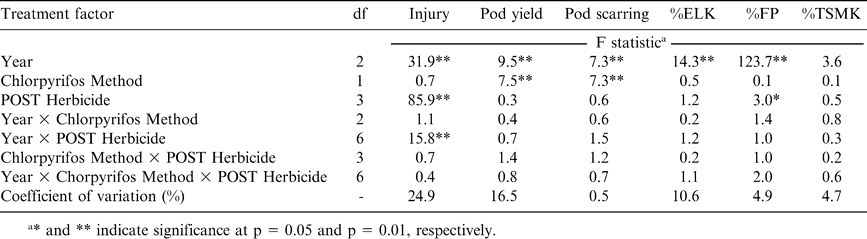
Analysis of variance for peanut injury 2 weeks after application, pod yield, and the percentage of pods with scarring caused by southern corn rootworm feeding, and percentages of extra large kernels (%ELK), fancy pods (%FP), and total sound mature kernels (%TSMK) following application of chlorpyrifos and selected postemergence (POST) herbicides.
Results and Discussion
The main affect of PRE herbicide and the interaction of year by PRE herbicide were significant for peanut injury 3 WAP (Table 1). Similarly, the main effect of POST herbicide and the interaction of POST herbicide with year were also significant for peanut injury 2 WAT (Table 2). In both experiments, chlorpyrifos application method did not affect peanut injury and there was no significant interaction between chlorpyrifos application method and PRE or POST herbicide treatment (Tables 1 and 2). The pegging application of chlorpyrifos was not applied until after PRE and POST herbicides were applied and visual estimates of peanut injury were recorded, and therefore serves as a no-chorpyrifos control for visual injury for each herbicide when compared with chlorpyrifos applied before planting.
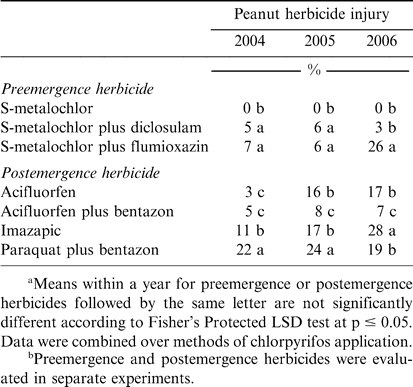
When pooled over chlorpyrifos method of application, PRE application of diclosulam and flumioxazin injured peanut similarly during 2004 and 2005 (Table 3). In contrast, peanut was injured 26% when flumioxazin was applied compared to only 3% when diclosulam was applied during 2006. Flumioxazin can be more injurious to peanut than diclosulam (Jordan, 2007b). Excessive injury from flumiozaxin in 2006 could not be explained by environmental conditions or application variables. Although excessive rainfall and below-normal temperatures at the time of peanut emergence have been suspected but not documented as causes of increased peanut injury from flumioxazin, these conditions were not present in 2006. Johnson et al. (2006) and Robinson et al. (2006) reported variation in peanut injury from flumioxazin applied according to the manufacturer's recommendations for use in peanut.
Peanut herbicide injury following application of acifluorfen was the same as injury observed for acifluorfen plus bentazon during 2004 but higher than acifluorfen plus bentazon during 2005 and 2006 (Table 3). Injury by imazapic exceeded that of acifluorfen during 2004 and 2006; injury by these herbicides was similar in 2005. Paraquat plus bentazon was more injurious than all treatments during 2004 and 2005 but was lower than injury caused by imazapic during 2006. Injury symptoms were typical for herbicides representing these modes of action (Senseman, 2007). Paraquat plus bentazon is often more injurious than acifluorfen or imazapic (Wilcut et al., 1995). However, in some instances imazapic can injure peanut significantly by stunting plants (Wilcut et al., 1995). Variation in visual injury across years could not be easily explained. However, lack of a significant interaction between chlorpyrifos and POST herbicides under conditions where variation in peanut response to POST herbicides was noted suggests that chlorpyrifos and POST herbicides will not interact under a wide range of environmental and edaphic conditions that may occur in peanut production systems.
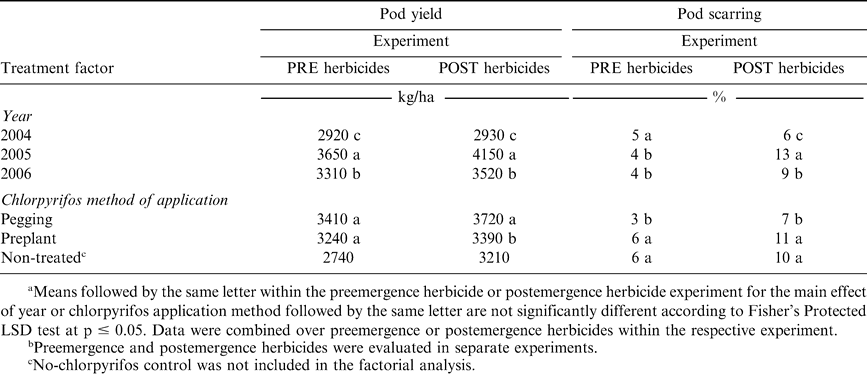
Pod yield was not affected by PRE or POST herbicides (Tables 1 and 2) even though herbicides injured peanut significantly early in the season (Table 3). The herbicides evaluated in these experiments are often applied to peanut and seldom adversely affect pod yield when applied based on the manufacturer's suggested use rate. Pod yield was affected by year in both PRE and POST herbicide experiments (Tables 1 and 2). However, year and chlorpyrifos method of application did not interact. When pooled over herbicide treatments and chlorpyrifos method of application, pod yield was the lowest during 2004, the highest during 2005, and intermediate during 2006 (Table 4). These experiments were conducted in the same field each year with the same cultivar, and therefore yield for the two experiments would have been expected to be similar. Surprisingly, method of application was not significant in the experiment evaluating PRE herbicides but was significant in the experiment evaluating POST herbicides (Tables 1 and 2). In the latter experiment, pod yield ranged from 3410 and 3240 kg/ha when chlorpyrifos was applied at pegging or PPI, respectively, with the no-chlorpyrifos control yielding 2740 kg/ha (Table 4). Pod yield was 3720 kg/ha when chlorpyrifos was applied at pegging compared with a lower yield of 3390 kg/ha when chlorpyrifos was applied before planting (Table 4). The no-chlorpyrifos control yielded only 3210 kg/ha in this experiment.
Percentages of ELK, FP, and TSMK were not affected by method of chlorpyrifos application, PRE herbicide, or interactions of these treatment factors (Table 1). Additionally, %ELK and %TSMK were not affected by chlorpyrifos method of application, POST herbicide, or interactions of these treatment factors (Table 2). Although not affected by chlorpyrifos method of application, %FP was affected by POST herbicide (Table 2). When pooled over years and methods of chlorpyrifos application, %FP was similar when acifluorfen, acifluorfen plus bentazon, or paraquat plus bentazon were applied (74 to 75%, data not shown). The %FP was 72% when imazapic was applied which was lower than the %FP for other herbicide treatments. The no-POST herbicide control produced 73% FP (data not shown). Although a lower %FP suggests a possible delay in pod development when comparing %FP following application of imazapic to application with the other herbicides, %FP from imazapic-treated peanut did not differ from the no-POST herbicide control. Additionally, %ELK and %TSMK were not affected by POST herbicides, and these market grade characteristics influence gross economic value of peanut while %FP does not affect gross economic value (USDA, 1998).
Pod scarring varied by year in the experiment with POST herbicides but not in the PRE herbicide experiment (Table 4). The highest amount of scarring in the POST experiment was noted during 2005 (13%) and exceeded scarring during 2004 (6%) or 2006 (9%) (Table 4). Planting date may have contributed to the higher amount of pod scarring during 2005 (25 May) compared with 2004 (15 May) and 2006 (12 May) (Table 4). The current advisory index for southern corn rootworm management (Brandenburg, 2007) indicates that peanut pods are at greater risk of damage when peanut is planted after 15 May than earlier plantings. A later planting date can result in pods being less developed and more susceptible to scarring and puncturing from southern corn rootworm feeding.
Pod scarring was lower when chlorpyrifos was applied at pegging rather than PPI, and this response was consistent across years and in both PRE and POST herbicide experiments within the same year (Tables 1, 2, and 4). In fact, pod scarring was the same when chlorpyrifos was applied before planting compared with the no-chlorpyrifos control (6%) in the PRE herbicide experiment (Table 4). Similarly, in the experiments with POST herbicides, scarring following chlorpyrifos applied prior to planting and the non-treated control was 11 and 10%, respectively (Table 4).
These data indicate that southern corn rootworm control, which is reflected in pod scarring, most likely will be lower when chlorpyrifos is applied prior to planting and incorporated. These results are in contrast to those of Brandenburg and Herbert (1991) and Herbert and Malone (2005) who reported similar control of southern corn rootworm by chlorpyrifos applied PPI versus pegging application in experiments where southern corn rootworm damaged peanut and reduced yield when chlorpyrifos was not applied. In our experiments, chlorpyrifos was applied to the soil surface and incorporated twice in the opposite direction to a depth approximately 8 cm using a field cultivator. The field was then ripped and bedded, and the top of the bed leveled prior to planting. Although uniformity and dilution of pesticides is a concern when fields are bedded, the majority of farmers in the Virginia-Carolina region apply PPI herbicides in conventional tillage systems in this manner. Less control of southern corn rootworm and subsequently greater scarring may have been associated with non-uniform distribution and possibly less chlorpyrifos in the pegging zone where southern corn rootworm develop after hatching and begin feeding on pegs and pods later in the season. Brandenburg and Herbert (1991) applied chlorpyrifos directly to the row bed after planting and incorporated in the pegging zone. In contrast, Herbert and Malone reported similar southern corn rootworm control when chlorpyrifos was applied as a granular at pegging or incorporated prior to begging in a manner similar to the method used in our experiment. Additional research is needed to determine the most feasible method of incorporation of chlorpyrifos at planting to obtain control that is both effective and consistent.
Results from these experiments also indicate peanut response to commonly used herbicides most likely will not vary when chlorpyrifos is applied to the soil prior to planting. However, the quantity of chlorpyrifos absorbed by emerging peanut shoots and roots is not known with respect to PPI application. Less corn injury from POST application of nicosulfuron was reported when organophosphate insecticides were applied in a band after planting rather than in the seed furrow (Bailey and Kapusta, 1994). Chlorpyrifos controls southern corn rootworm through contact between larvae and insecticide (Brandenburg and Herbert, 1991; Brandenburg, 2007) and is considered very insoluble in water and immobile in soil (Brandenburg and Herbert, 1991; Racke, 1992; Wauchope et al., 1992). Incorporation of chlorpyrifos at a more shallow depth after bed establishment or as a PRE application may result in more uniform distribution in the pegging zone where southern corn rootworm feeds on pods. Additional research is needed to define interactions of chlorpyrifos and herbicides when chlorpyrifos is applied in this manner.
Acknowledgements
This research was supported by the North Carolina Peanut Growers and the Peanut CRSP, USAID (LAG-G-0096-90013-00). Appreciation is expressed to staff at the Upper Coastal Plain Research Station and to Carl Murphey for assistance with these experiments.
Literature Cited
Allen R. L. and Snipes C. E. 1995 Interactions of foliar insecticides applied with pyrithiobac. Weed Technol 9 : 512 – 517 .
Bailey J. A. and Kapusta G. 1994 Soil insecticide and placement influence corn (Zea mays) tolerance to nicosulfuron. Weed Technol 8 : 598 – 606 .
Barbour J. D. and Brandenburg R. L. 1995 Impact of type and timing of southern corn rootworm treatments on predaceous arthropods in peanut. J. Entomol. Sci 30 / 4 : 447 – 462 .
Brandenburg R. L. 2007 Peanut insect management. 75 93 in 2007 Peanut Information North Carolina Coop. Ext. Serv. Ser. AG-331 132 pp.
Brandenburg R. L. and Herbert D. A. 1991 Effect of timing on prophylactic treatments for southern corn rootworm (Coleoptera: Chrysomelidae) in peanut. J. Econ. Entom 80 : 1894 – 1898 .
Carmer S. G. , Nyquist W. E. , and Walker W. M. 1989 Least significant differences for combined analyses of experiments with two- or three- factor treatment designs. Agron. J 81 : 665 – 672 .
Herbert D. A. and Malone S. 2005 Peanut soil insect pest studies and evaluation of chlorpyrifos management options. Proc. Am. Peanut Res. and Educ. Soc 37 : 82 .
Herbert D. A. , Malone S. , Brandenburg R. L. , and Royals B. M. 2004 Evaluation of the peanut southern corn rootworm advisory. Peanut Sci 31 : 28 – 32 .
Herbert D. A. , Petka W. J. , and Brandenburg R. L. 1997 A risk index for determining insecticide treatment for southern corn rootworm in peanut. Peanut Sci 24 : 128 – 134 .
Johnson W. C. , Prostko E. P. , and Mullinix B. G. 2006 Phytotoxicity of delayed applications of flumioxazin on peanut (Arachis hypogaea). Weed Technol 20 : 157 – 163 .
Jordan D. L. 2007a Peanut production practices. 23 – 46 in 2007 Peanut Information North Carolina Coop. Ext. Ser. AG-331 132 pp.
Jordan D. L. 2007b Peanut weed management:. 47 – 74 in 2007 Peanut Information North Carolina Coop. Ext. Ser. AG-331 132 pp.
Lancaster S. , Jordan D. , Brandenburg R. , Royals B. , Shew B. , Bailey J. , Curtis V. , York A. , Wilcut J. , Beam J. , Prostko E. , Culpepper S. , Grey T. , Johnson C. , Kemerait R. , Grichar J. , Baughman T. , Dotray P. , Brecke B. , MacDonald G. , Tredaway-Ducar J. , and Walls B. 2005 Tank mixing chemicals applied to peanut: are the chemicals compatible North Carolina Coop. Ext. Ser. Pub. AG-W-653R .
Morton C. A. , Harvey R. G. , Wedberg J. L. , Kells J. J. , Landis D. A. , and Lueschen W. E. 1994 Influence of corn rootworm insecticide on the response of field corn (Zea mays) to nicosulfuron. Weed Technol 8 : 289 – 295 .
Rahman A. and James T. K. 1993 Enhanced activity of nicosulfuron in combination with soil-applied insecticides in corn (Zea mays). Weed Technol 7 : 824 – 829 .
Racke K. D. 1992 The environmental fate of chlorpyrifos. Rev. Environ. Contam. Toxicol 131 : 1 – 151 .
Robinson B. L. , Clewis S. B. , and Wilcut J. W. 2006 Weed management with flumioxazin in strip-tillage peanut (Arachis hypogaea). Peanut Sci 33 : 41 – 46 .
Senseman S. A. 2007 Herbicide handbook. 9th edition Lawrence, KS Weed Science Society of America (in press) .
Shew B. B. 2007 Peanut disease management:. 2007 Peanut Information North Carolina Coop. Ext. Ser. AG-331 94 – 118 132 pp. in .
Sholar J. R. , Mozingo R. W. , and Beasley J. P. Pattee H. E. and Stalker H. T. Peanut cultural practices. 354 – 382 in Advances in Peanut Science 1995 Stillwater, OK Am. Peanut Res. Educ. Soc., Inc 614 pp.
[USDA] U.S. Department of Agriculture 1998 Farmers' stock peanuts inspection instructions Washington, DC USDA .
Wauchope R. D. , Buttler T. M. , Hornsby A. G. , Augustijn-Beckers P. W. M. , and Burt J. P. 1992 SCS/ARS/CES Pesticide properties database for environmental decision making. Rev. Environ. Contam. Toxicol 123 : 1 – 157 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification. Peanut Sci 8 : 134 – 141 .
Wilcut J. W. , York A. C. , Grichar W. J. , and Wehtjie G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea). 207 – 224 in Pattee H. E. and Stalker H. T. Advances in Peanut Science Stillwater: Am. Peanut Res. and Ed. Soc .
Notes
- Professor, Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695; William Neal Reynolds Distinguished Professor, Department of Entomology, North Carolina Sate University, Box 7613, Raleigh, NC 27695; Research Technician, Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695; Research Technician and Technician Assistant, Department of Entomology, North Carolina State University, Box 7613, Raleigh, NC 27695, respectively. [^] * Corresponding author's e-mail: david_jordan@ncsu.edu.
Author Affiliations