Introduction
Weed management in conventional peanut production is typically an integrated system of crop rotations, cultural practices, herbicides, and cultivation (Buchanan et al., 1982). There is still heavy dependence on herbicides, with up to seven different active ingredients applied during the growing season (E. P. Prostko, personal communication). Cultivation is a proven supplement to herbicides in peanut production (Bridges et al., 1984; Wilcut et al., 1987). However, approximately 23% of the Georgia peanut acreage uses a form of reduced tillage (N. B. Smith, personal communication) due to reduced labor costs and reduction in incidence of spotted wilt, a viral disease of peanut endemic to southeastern U. S. (Brown et al., 2003, 2005). There is now less dependence on cultivation for weed control in reduced tillage systems and greater herbicide use, compared to conventional tillage systems.
There is interest in growing certified organic peanut to support increasing demand for organic processed peanut foods. Currently, demand exceeds supply. In 2005, there was a supply shortage of nearly 5000 tons of organic peanut in the U. S. (Culbreath, 2005). If demand for organic peanut increases at rates similar to the demand for other organic foods, there will be a need for 125,000 tons of certified organic peanut in 15 years. Weed management is universally considered to be the major limiting factor in organic crop production (Organic Farming Research Foundation, 2001). Weed management in organic cropping systems is conceptually based on the same principles of conventional crop production; an integration of crop rotations, cultural practices, mechanical controls, and herbicides. However, the only herbicides that can be used in organic crop production are those approved by the Organic Materials Resource Institute (OMRI; Box 11558; Eugene, OR 97440). In general terms, OMRI certification ensures that growers can use the herbicide, without compromising their organic crop production certification. Herbicides approved by OMRI are derived from natural sources. While OMRI herbicides are approved to be used in certified organic crop production, such approval generally does not consider weed control efficacy or crop injury.
A form of cultural weed control that may be useful in organic peanut production is modified row patterns that enhance the competitiveness of peanut with weeds by achieving quicker canopy closure (Cardina et al., 1987; Hauser and Buchanan, 1982; Wehtje et al., 1984). Peanut seeded in narrowly spaced rows suppressed weed emergence better than peanut seeded in wide rows (Buchanan and Hauser, 1980; Hauser and Buchanan, 1981). While the concept of narrow row spacings improving weed management by quicker canopy closure is fundamentally sound, peanut yield increases over wide row plantings were not always attributed to improved weed control. It was the consensus that narrow row patterns created conditions favorable for crop growth that increased yield (Colvin et al., 1985; Cox and Reid, 1965). Furthermore, in recent years narrow row spacings were shown to reduce incidence of spotted wilt (Brown et al., 2005).
Cultivation is a form of weed control that is feasible in organic peanut production. While cultivation has been shown to be a useful component of integrated systems in conventional production (Hauser et al., 1973; Hauser and Parham, 1969), the utility of cultivation is limited by the tendency of cultivation to predispose peanut to incidence of soil borne diseases (Boyle, 1952, 1956). Cultivation that moves soil onto the peanut crown also moves sclerotia from Sclerotium rolfsii Sacc. onto the peanut crown, increasing stem rot incidence. For this reason, peanut are cultivated at a slow ground speed with sweeps angled to cut weeds and not displace soil.
Another form of weed control compatible with certified organic crop production is the use of propane flaming. Flaming is a proven means of layby weed control in cotton (Byrd et al., 1994; Byrd and Snipes, 1994; Matthews and Smith, 1971; Snipes, 1996). In this case, flaming is directed to the woody stalk of cotton and targeted to mid-season weeds that escaped earlier control efforts. Flaming does not incinerate weeds. Rather, the heat generated by flaming ruptures plant cells causing dessication of the whole plant (Snipes, 1996). Despite the proven utility of flaming for layby weed control in cotton, there is no information on using flaming for weed control in peanut. Arachis spp. are plants native to South America. The ecosystem of this region is prone to cyclic drought and heavily grazed, and Arachis spp. have evolved to thrive under these conditions (Hammons, 1973). It is plausible that peanut may be tolerant to flaming early in the growing season and this technique may be a useful form of early-season weed control in organic peanut production.
Field trials were initiated in Georgia to systematically evaluate integrated systems of cultural practices, OMRI-approved herbicides, flaming, and cultivation for weed control in organic peanut production.
Materials and Methods
Irrigated field trials were conducted at the Coastal Plain Experiment Station Ponder Farm near Tifton, GA in 2004 and 2005. The soil was a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults) with 88% sand, 6% silt, and 6% clay and 0.2% organic matter. The soil at this location is representative of soils in the southeastern U. S. peanut producing region. These sites had been managed for weed science research the preceding 18 years, which featured crop plantings alternating yearly with weedy fallow.
The experimental design was a factorial arrangement of treatments in a split, split-plot experimental design with four replications. Treatments were row patterns (two levels), remedial weed control inputs (four levels), and cultivation (three levels).
Main plots of the experimental design were peanut seeded in either wide or narrow row patterns. Wide rows were two rows spaced 91 cm apart on a 1.8 m raised seedbed. Narrow rows were four rows spaced 30 cm apart centered on a 1.8 m wide raised seedbed. All plots were seeded using vacuum planters (ATI, 17135 West 116th St., Lenexa, KS 66285) that placed seeds at a depth of 3.8 cm. Peanut were seeded in wide rows at a density of 20 seed/m in each row and peanut seeded in narrow rows were seeded at a density of 10 seed/m in each row. The total seeding rate for both row spacings was 120 kg/ha.
Sub-plots of the experimental design were four levels of early-season remedial weed control: clove oil (Matran®; 50% clove oil; EcoSMART Technologies; 318 Seaboard Lane, Suite 208; Franklin, TN 37067), citric plus acetic acid (Ground Force®; Abby Laboratories, Inc.; 14000 Sunfish Lake Blvd., NW, Suite 100; Ramsey, MN 55303), broadcast flaming, and a nontreated control. Clove oil and citric plus acetic acid are marketed for weed control in organic cropping systems and were applied at 70 and 65 L/ha, respectively. All remedial treatments were applied to peanut 2-wk after emergence, with weeds present from the cotyledon through one true leaf stage of growth. Sprayable treatments were applied with a tractor-mounted, CO2 pressurized, plot sprayer calibrated to deliver 234 L/ha using low-drift spray tips (Turbo TeeJet®; Spraying Systems Co., P. O. Box 7900; Wheaton, IL 60189) treating a swath 1.8 m wide. No adjuvants were added to the sprayable treatments. Flaming treatments were applied with a tractor mounted flamer (Red Dragon Row Crop Flamer®, Flame Engineering, Inc.; P. O. Box 577; LaCrosse, KS 67548) fueled by liquidified propane, featuring eight burners spaced 15 cm apart, with each burner directing flame in a 18 cm band, resulting in broadcast flaming swath 1.3 m wide. The portions of the seedbed not flamed were the outside edges, which were cultivated with a single sweep mounted on the flamer frame, centered in each wheel track. Based on the amount of fuel consumed and area treated, we estimated that broadcast flaming consumed liquid propane at 280 L/ha.
Sub, sub-plots of the experimental design were frequencies of cultivation; 1X, 2X, and a noncultivated control. Sweeps 23 cm wide were arrayed to cultivate peanut seeded in narrow row patterns, while sweeps 41 cm wide were used to cultivate peanut seeded in wide row patterns. The initial cultivation was 2-wk after peanut emergence. The second cultivation was one week after the initial cultivation.
The DP-1 peanut cultivar was used in these trials since it has excellent host plant resistance to early and late leafspot (Cercospora arachidicola and Cercosporidium personatum, respectively), southern stem rot, and tomato spotted wilt virus (A. K. Culbreath, personal communication). Plots were not sprayed with any fungicide, insecticide, or synthetic herbicide during the growing season. However, these plots were not able to be certified organic due to the use of seed treated with non-approved protectants and lack of suitable isolation from adjacent areas that are routinely treated with pesticides.
Parameters measured were visual estimates of mid-season weed control and peanut yield. Peanut yields were measured by mowing to reduce weed biomass and improve the digging operation, digging, inverting, air-curing, and combining peanut from the entire plot using commercial two-row equipment. Yield samples were mechanically cleaned to remove foreign material, particularly weed biomass, with yields reported as cleaned farmer stock peanut.
Data were analyzed using a mixed-model analysis. Degrees of freedom were partitioned to test singularly and in combination the effects of row spacing, remedial herbicides, and cultivation on visual estimates of weed control and peanut yield. Means were separated using Fisher's LSD (P≤0.05).
Results and Discussion
In 2004, the experimental site had heavy natural infestations of Texas millet (Urochloa texana (Buckl.) R. Webster) at 10/m2, southern crabgrass [Digitaria sanguinalis (L.) Scop.] at 10/m2, and pitted morningglory (Ipomoea lacunosa L.) at 5/m2. In 2005, there were heavy infestations of crowfootgrass [Dactyloctenium aegyptium (L.) Richt./P. Beauv] at 10/m2, smallflower morningglory [Jacquemontia tamnifolia (L.) Griseb.] at 5/m2, and Florida beggarweed [Desmodium tortuosum (Sw) DC] at 3/m2.
In early June 2005, the experimental site had excessive rainfall, due in part to Tropical Storm Arlene. As a result of the rainfall, all field operations were suspended for two weeks which coincided with scheduled cultivation treatments. Once the experimental site dried sufficiently for field operations, peanut plants were too large to cultivate. Therefore, none of the cultivation treatments were conducted in 2005 and all data for 2005 were pooled across cultivation treatments. This also precluded pooling of data between years. Therefore, all data are presented by year.
The flamer evaluated in this study was troublesome. Individual burners were prone to 'flame-out' due to rust or flaked paint clogging the burner orifice. Furthermore, the flame was nearly colorless in daylight hours making it impossible to notice when burners were no longer lit. This was noted in practice flaming events in border areas. Flaming treatments were conducted pre-dawn in 2004 in total darkness to facilitate monitoring the burners. While we were able to easily monitor burner operations in the pre-dawn darkness, heavy dew on weed seedlings cooled the leaf surface to the extent that efficacy was likely affected. In 2005, we altered the protocol by flaming at pre-dusk. It was dark enough to easily monitor the burners, but there was no dew on weed leaves and efficacy seemed to be better compared to pre-dawn flaming (data not shown).
Annual Grass Control
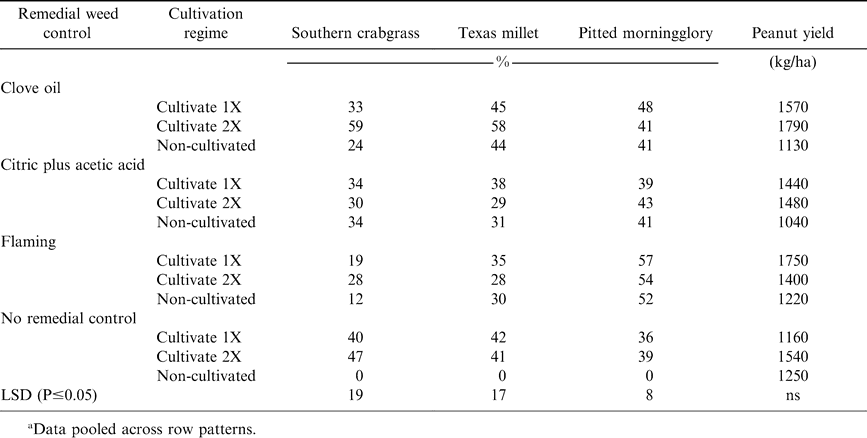
There was no significant effect of row pattern on annual grass control in 2004 (data not shown). However, mid-season annual grass control was affected by the interactive effects of remedial weed control and cultivation (Table 1). Overall weed control was unacceptable, regardless of the treatment combination. Southern crabgrass and Texas millet were not effectively controlled, with flaming and citric plus acetic acid being the least effective of all remedial weed control treatments. Furthermore, both treatment combinations offered no improvement over cultivation alone. Three days after treatment, there was minimal evidence of citric plus acetic acid phytotoxicity on treated grasses. Grasses treated with flaming showed some foliar necrosis, however the growing point was not affected. Clove oil improved control of both grass species compared to other remedial treatments, but the maximum control of both grasses occurred when clove oil was combined with cultivation 2X, which was still unacceptable (58 to 59%). Annual grass control with cultivation alone was similar to efficacy from clove oil combined with cultivation. The level of annual grass control with cultivation alone was also unacceptable (≤ 47%).
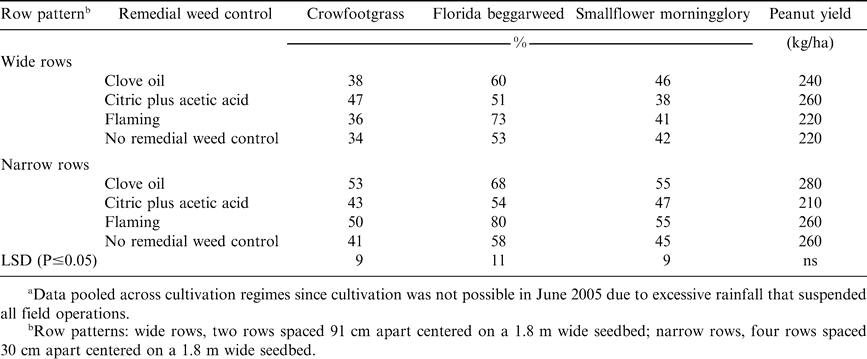
In 2005, the interactive effects of row patterns and remedial weed control affected annual grass control (Table 2). Crowfootgrass was the predominant weed present and none of the treatment combinations effectively controlled crowfootgrass at mid-season.
Dicot Weed Control
Pitted morningglory was present in 2004 and no combination of remedial herbicides and cultivation effectively controlled the weed when rated at mid-season (Table 1). Observation of pitted morningglory three days after treatment indicated that the weed was particularly sensitive to clove oil and flaming (data not shown). There was little evidence of citric plus acetic acid efficacy on pitted morningglory at the same time of observation. These observations, along with the mid-season ratings, suggest there is little or no residual control of dicot weeds by any of the remedial weed controls used in this study. Cultivation used in conjunction with the remedial weed controls did not improve mid-season pitted morningglory control over the remedial controls alone (Table 1).
Florida beggarweed and smallflower morningglory were present in 2005 (Table 2). In general, the remedial weed controls in this study were more effective in controlling smallflower morningglory in narrow row plantings than in wide rows, while Florida beggarweed control was not improved by narrow rows. Flaming was the most effective remedial treatment evaluated in controlling Florida beggarweed when rated mid-season, with visual ratings ranging from 73 to 80% control with flaming for wide vs narrow rows, respectively. None of the remedial weed controls evaluated effectively controlled smallflower morningglory, with the greatest level of mid-season control being 55% for the clove oil and flaming, both with the narrow row system.
Peanut Yield
Peanut yields were poor each year of the study, with row pattern, remedial weed control, and cultivation having no effect on peanut yield (Tables 1 and 2). In 2004, peanut yields averaged 1400 kg/ha, compared to the average yield in Georgia of 3340 kg/ha (Georgia Farm Report; USDA-NASS; Athens, GA 30601. In 2005, peanut yields in our trials averaged 210 kg/ha, compared the Georgia yield average of to 3360 kg/ha. Clove oil caused moderate injury on peanut, but symptoms were transitory and peanut quickly recovered (data not shown). Citric plus acetic acid produced no symptoms of phytotoxicity on peanut. Broadcast flaming caused moderate, but short-lived, injury symptoms on peanut. Therefore, the poor yields were due solely to ineffective weed control by all treatment combinations evaluated in these trials. The better yields in 2004 compared to 2005 were likely due to the ability to cultivate for weed control. Annual grasses are among the most competitive weeds of peanut (Johnson and Mullinix, 2005) and no treatment combination effectively controlled any of the three annual grass species present in these trials.
In conclusion, the systems of weed management evaluated for use in organic peanut production were not effective under the conditions present in these trials. The main reason for the poor yields, particularly in 2005, was ineffective grass control. It should also be noted that these sites had a history of severe weed infestations that led to continual emergence of weeds. One of the principles of weed management in organic cropping systems is intense weed management and not allowing weeds to propagate. The previous practice of allowing these sites to be weedy fallow is contrary to these principles. Peanut growers in the southeastern U. S. who are interested in diversifying into certified organic production tend to initiate organic production on long-term fallow fields as a means to expedite the three-year transition period prior to organic certification. Typically, these fields were weedy during the previous fallow period. It is likely that weed control will be extraordinarily difficult in their initial attempts at certified organic peanut production, like it was in our trials.
Poor efficacy aside, these weed management systems are cost prohibitive and dependent on ideal edaphic and environmental conditions for maximum efficacy. At the use rates evaluated in this study, broadcast applications of clove oil, citric plus acetic acid, and flaming cost $1112, $968, and $195/ha, respectively. In contrast, the total cost of weed management in irrigated conventional peanut is estimated at $104/ha (N. B. Smith, personal communication). Since these remedial treatments are marginally efficacious at best, conditions at the time of treatment must be ideal for any reasonable chance of success. It is also clear that successful weed management in organic peanut will be costlier and riskier than weed management in conventional peanut production systems.
Acknowledgements
We acknowledge the technical contributions of Daniel R. Evarts and Andy M. Hornbuckle in this study. This research was partially supported by a grant from the Southern Regional Sustainable Agricultural Research and Education Program, through Hebert Green Agroecology, Inc., Dr. Mark A. Boudreau, President.
Literature Cited
Notes
- Res. Agron., USDA-ARS and Agric. Res. Statistician, Coastal Plain Exp. Sta., Tifton, GA 31793-0748 [^] * Corresponding author's Email: Carroll.Johnson@ars.usda.gov.
Author Affiliations