Introduction
Spotted wilt disease caused by TSWV is a serious disease in peanut in the United States (Black et al., 1986; Culbreath et al., 1990; Garcia et al., 2000; Hagan et al., 1990). In 1971, TSWV was first reported infecting peanut in Texas (Halliwel and Philley, 1974). During 1985, 1986, 1990, and 1991, several epidemics of TSWV occurred on peanut in southwest Texas, with yield reductions approaching 95% (Black 1987; Black et al., 1986). In 1985 and 1986, losses in Texas were estimated in the millions of dollars (Mitchell and Smith, 1991). From 1989 throughout the mid 1990's in Georgia, the incidence of spotted wilt increased greatly in peanut and other crops. (Culbreath et al., 1990; 1991; 1992). TSWV has a wide host range of more than 1,000 plant species in at least 92 families where both monocotyledons and dicotyledons have been reported to host the virus (Peters, 1998). Symptoms in peanut due to TSWV include yellowing, necrosis, chlorotic ring spots of various sizes and shapes, and stunting (Kokalis-Burelle et al., 1997; Sherwood and Melouk, 1995; Simpson et al., 2003). TSWV infection in peanut causes reduction in pod number and size. Kernels of infected plants may also be reduced in size or become malformed, and discolored (Sherwood and Melouk, 1995).
TSWV is transmitted by at least seven thrips species belonging to the insect Order Thysanoptera. The tobacco thrips, Frankliniella fusca (Hinds), western flower thrips, Frankliniella occidentalis (Pergande) are two predominant species in the peanut-producing states that transmit TSWV (Mitchell and Smith, 1991; Mulder et al., 1991; 2001; Todd et al., 1990). TSWV is acquired by immature thrips feeding on infected host plants (German et al., 1991; Goldback and Peters, 1996), and is retained in the gut of thrips throughout the molting, pupation, and adult stages in a persistent manner. TSWV replicates within the vector F. occidentalis, allowing the virus to be transmitted for long periods of time and increasing the vector's ability to infest many healthy peanut plants within the field (Wijkamp et al., 1993).
Controlling the vector chemically seems to have no or little impact in reducing disease incidence, and other alternative means of managing this disease are needed (Culbreath et al., 2003). Host resistance is the most effective means of reducing the incidence of spotted wilt disease in most peanut-producing states (Culbreath et al., 1996; 1999; 2000; Wells et al., 2002). Several peanut cultivars have been reported to have moderate resistance and/or tolerance to TSWV infection (Culbreath et al., 2003). Southern Runner was the first cultivar reported to possess partial resistance to multiple soilborne diseases and foliar diseases, including TSWV (Culbreath et al., 2003). Georgia Browne, Georgia Green, ViruGard, UF MDR 98, C-99R, AnduII, NC V-11, and Norden are peanut cultivars reported to have moderate resistance to TSWV (Culbreath et al., 1996; 1999; 2000; Wells et al., 2002). Tamrun-96 is a runner-type cultivar developed by Texas A&M University, reported to have moderate resistance to TSWV (Smith et al., 1998).
Recently, several breeding lines have shown greater field resistance to TSWV in the eastern U. S. (Lyerly et al., 2002; Mandal et al., 2001). Research on host resistance to TSWV in western peanut growing areas is needed. Therefore, the objectives of this research are: 1) to evaluate the reaction of eight peanut cultivars to TSWV under field conditions in the southwest, and 2) to evaluate the response of these cultivars to mechanical inoculation with TSWV under greenhouse conditions.
Materials and Methods
Reaction of Peanut Cultivars to TSWV in Field Plots
During the 2001, 2002, and 2003 growing seasons, peanut cultivars Tamrun-98, Tamrun-96, Southwest Runner, Georgia Green, Tamrun OL-01, Georgia Hi Oleic, Tamrun OL-02 and Okrun were planted in field plots near Pearsall, TX, where TSWV disease is endemic. In all growing seasons, field plots consisted of eight cultivars (treatments) arranged in a randomized complete block design with four replications. In each replicate, cultivars were planted in two rows 6 m long with row spacing of 0.91 m. Number of plants per plot was between 120–130 which was lower than commercial fields to promote higher incidence of spotted wilt disease (Culbreath et al., 1999), and to facilitate observation of symptoms in individual plant. Plants with symptoms on one or more leaflets were considered symptomatic (Culbreath et al., 1999). Positive diagnosis of TSWV was confirmed by sampling two symptomatic leaves and performing enzyme-liked immuneosorbant assay (ELISA) test (Clark and Adam, 1977) using a commercial kit obtained from Agdia, Inc. (Elkhart, IN). Reactions of ELISA were measured with an ELISA-plate reader (Microplate Automated Reader EL 310, Bio-Tek Instruments, Winooski, VT) at 490 nm. A sample was considered positive for TSWV if the A490 reading was greater than twice the average negative control.
Area under disease progress curve (AUDPC) was calculated for each plot as described by Shaner and Finney (1977) using time in days after planting and disease incidence. Chlorothalonil or other recommended fungicides were applied on a 14-day schedule for managing leaf spot fungal diseases.
All data were subjected to statistical analysis of variance (SAS Inst., 1985). Duncan's multiple-range test (MRT) was used for means comparison of peanut cultivars (Steel et al., 1997).
Response of Peanut Genotypes Mechanically Inoculated with TSWV Under Greenhouse Conditions
The greenhouse experiment was designed as an 8 × 3 × 2 factorial arrangement of treatments (8 genotypes × 3 inoculation times × 2 inoculation treatments) in a randomized complete block design (RCBD) with four replications. Seeds were germinated in trays lined with water-saturated- filter paper (Whatman #1). Each germinating seed was planted in a polyvinyl chloride (PVC) tube (10 cm inside diameter and 76 cm in length) filled with fritted clay as described by Ketring (1984). Twenty four hours prior to inoculation plants were covered with paper (Mandal et al., 2001). Plants at 5, 12, and 19 days post planting were dusted with 320 grit carborundum (Fisher Scientific, New Jersey USA) and mechanically inoculated with TSWV. Inoculum was prepared by grinding 5g of TSWV- infected leaves of red tasselflower (Emilia sochifolia L.) or jimsonweed (Datura stramonium L.) in 10 ml. of 0.05 M potassium phosphate buffer (pH 7.0) containing 0.2% of 2-mercaptoethanole and 1% Celite (Fisher Scientific, New Jersey USA). Plants inoculated with buffer containing 0.2% of 2-mercaptoethanole and 1% Celite served as controls. All plants were maintained in the greenhouse, watered daily, and fertilized weekly with a solution containing 2 g/L of a fertilizer (20-20-20). Plants were examined daily for symptoms. Three weeks after each inoculation, plants were lifted from the tubes, and root length (cm), root volume (cm3), plant height (cm), fresh weight (g), relative water content, and values from ELISA from leaves were determined. Values of ELISA were used to indicate the relative presence of TSWV, similar to research reported by Hunger et al (1991). Root volume was determined by water displacement method (Heinzman and Eilrich, 1977; Ketring, 1984). Relative water content (RWC) of foliage was determined following a standard method according to Erikson et al (1991). Disease severity index (DSI) of TSWV symptoms was scored for each plant using a 1–4 scale where 1 = no symptoms, 2 = only inoculated leaves displaying symptoms, 3 = systemic symptoms (mosaic and / or mottled), yellowing, top chlorosis, and / or leaf distortion, and 4 = severe necrosis, stunting, and / or dead plant.
Results and Discussion
Field Experiments
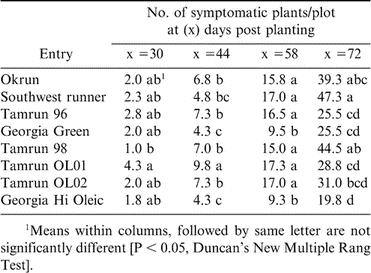
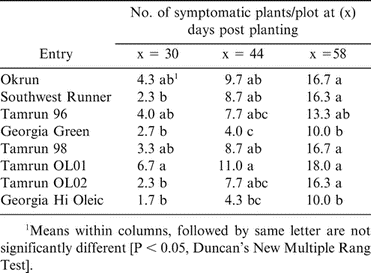
In the 2001 growing season, TSWV-like symptoms were first observed at 30 days post-planting (DPP) in all peanut genotypes tested. Leaves from symptomatic plants were collected and tested for the presence of TSWV by ELISA, where values ranged from 0.28 to 1.33. TSWV disease incidence in 2001, in all peanut cultivars increased with time (Table 1). Disease incidence of spotted wilt at 72 DPP was higher (P < 0.05) in Southwest Runner, Okrun, and Tamrun 98 than the other cultivars (Table 1). During the 2002 growing season, disease incidence of TSWV was low and thus only one reading was taken at 135 DPP where the cultivars Georgia Green and Georgia Hi Oleic exhibited the least TSWV incidence (Table 2). In the 2003 growing season, TSWV infection was observed at 30 DPP. At 58 DPP, Tamrun-96, Georgia Green, and Georgia Hi Oleic showed the lowest disease incidence compared with other peanut cultivars (Table 3).
During the 2001 growing season, AUDPC values of spotted wilt of Georgia Hi Oleic, Georgia Green, and Tamrun-96 were 162, 206 and 212, respectively, which were lower than those of Okrun, Tamrun-98, and Southwest Runner (310, 341, and 372, respectively). Similarly, in the 2003 growing season, the AUDPC of Georgia Hi Oleic, Georgia Green, and Tamrun-96 were 120, 125 and 187, respectively, which were lower than those of Southwest Runner, Tamrun-98, and Okrun (208, 215 and 230, respectively). During the three growing seasons, disease incidence of TSWV varied between the peanut cultivars tested. Georgia Green, Georgia Hi Oleic, and Tamrun-96 cultivars exhibited greater resistance to TSWV compared to the other cultivars tested.
The resistance of Georgia Green to TSWV observed in our plots in 2001, 2002, and 2003 corroborated with the partial resistance reported by other researchers (Culbreath et al., 1996). In addition to other moderate resistant cultivars, Georgia Green has been accepted as a cultivar with moderate level of resistance to TSWV by growers in Southeastern peanut producing areas (Culbreath et al., 2003). In field evaluations conducted in Georgia and Florida, Georgia Green exhibited on average 31 to 59% TSWV disease incidence during the 1997 and 1998 growing seasons, respectively (Culbreath et al., 2000). In a related study it was reported that Georgia Green had the lowest incidence of TSWV compared to other peanut genotypes evaluated (Culbreath et al., 1996). Tamrun-96, a peanut cultivar released by Texas A&M University, is reported as being moderately resistant to TSWV (Smith et al., 1998). However, it exhibited susceptibility to TSWV, when evaluated in field plots in Georgia and Florida (Culbreath et al., 1996). Georgia Hi Oleic cultivar was released recently and displayed some resistance to TSWV under field conditions. Tamrun OL-01 cultivar, recently released by Texas A&M University, was reported to have a moderate tolerance to TSWV, southern blight (Sclerotium rolfsii Sacc.), and Sclerotinia blight (Sclerotinia minor Jagger) (Simpson et al., 2003). In our study, Georgia Green, Georgia Hi Oleic, and Tamrun-96 exhibited moderate resistance to TSWV in field plots near Pearsall, TX. These results agree with results reported on moderate resistance of other cultivars to TSWV when evaluated under field conditions (Culbreath et al., 1996; 1999; 2000). The mechanisms that are responsible for differences in disease incidence of TSWV among peanut cultivars have not yet been characterized (Culbreath et al., 1996; Lyerly et al., 2002).
Response of Peanut Cultivars to Mechanical Inoculation by TSWV Under Greenhouse Conditions.
Disease Severity
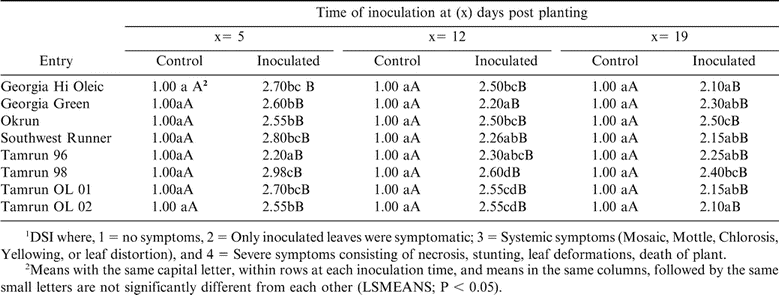
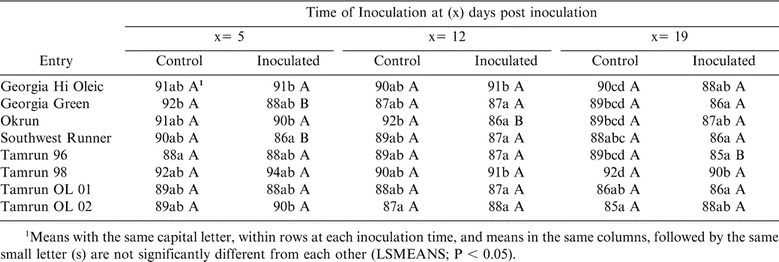
More than 90% of TSWV-inoculated peanut plants grown in the greenhouse, of all peanut cultivars expressed foliar symptoms. Plants inoculated with buffer only (Controls) did not exhibit TSWV symptoms. Expression of TSWV on inoculated peanut cultivars is shown in Table 4. The first inoculation at 5 DPP, revealed that Tamrun-96 had significantly lower DSI values compared to the other cultivars. However, Tamrun-98 was the most susceptible to TSWV compared to other cultivars. The second inoculation, at 12 DPP, showed that Georgia Green, Southwest Runner, and Tamrun-96 had the lowest DSI values when compared to other cultivars while Tamrun-98 was the most susceptible to TSWV (Table 4). When plants were inoculated with TSWV at 19 DPP, Okrun had significantly higher DSI value than other cultivars with the exception of Tamrun-98 (Table 4).
It was reported that Georgia Green did not express the observed level of field resistance to TSWV following mechanical inoculation. The resistance of Georgia Green in the field might be effective only against thrips-borne inoculum, and might have broken down against the relatively high dosage applied in a mechanical inoculation (Mandal et al., 2002). Tamrun-96 evaluated in field plots in Georgia and Florida has exhibited susceptibility to TSWV. In contrast, in our study Tamrun-96 grown in Texas plots exhibited a moderate level of resistance to TSWV. Differences between these studies might be due to variation between the isolates of TSWV in Southwest and in the Southeast.
Detection of TSWV
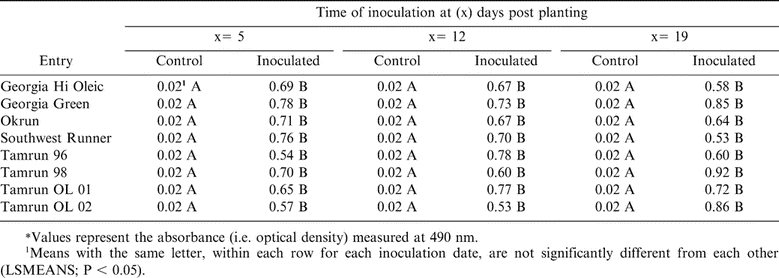
Significant differences were observed in all inoculated plants compared with non-inoculated controls at all three mechanical inoculation times under greenhouse conditions. Readings of ELISA in inoculated planted showed variations (Table 5). TSWV was not detected in non-inoculated plants (Table 5); however, no differences were observed in ELISA values among the various cultivars irrespective of time of inoculation.
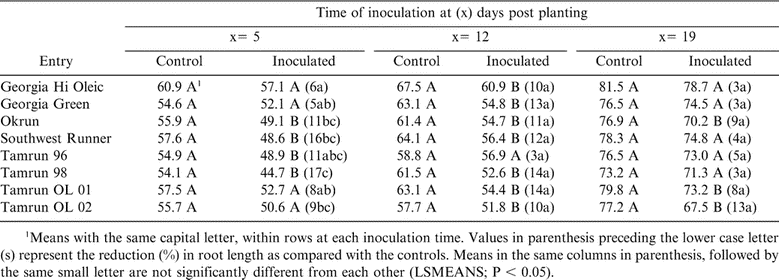
Root Length
Root lengths of inoculated plants and non-inoculated controls are presented in Table 6. Inoculation at 5 DPP, resulted in a 17% reduction in root length of Tamrun-98 which was significant (P < 0.05) from Georgia Hi Oleic, Georgia Green and Tamrun OL-01 (Table 6). At 12 DPP, the root length of Tamrun-96 was not affected, while other cultivars were reduced significantly (Table 6). At 19 DPP inoculation, the greatest reduction in root length compared to non-inoculated plants, was observed on Okrun, Tamrun OL-01, and Tamrun OL-02. Generally, at 12 DPP most inoculated plants compared to non-inoculated peanuts showed significant reductions in root length, but the percent reduction among cultivars was not significant at the P < 0.05 level (Table 6).
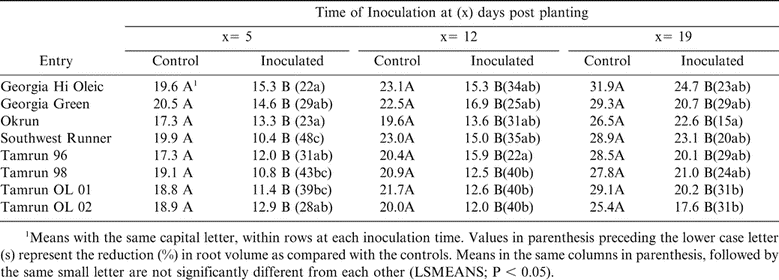
Root Volume
When non-inoculated controls and inoculated plants were compared for root volume, significant differences were revealed at all three inoculation times (Table 7). However, percentages of reduction in root volume varied among cultivars. At 5 DPP, inoculation of Southwest Runner with TSWV resulted in 48% reduction in root volume, which was significant (P < 0.05) from Georgia Hi Oleic, Georgia Green, Okrun, Tamrun-96, and Tamrun OL-02 (Table 7). When inoculation was performed at 12 DPP, Tamrun-96 showed the least reduction (22%) in root volume which was significant (P < 0.05) from Tamrun-98 and Tamrun OL-01 (Table 7). Least reduction (15%) in root volume was observed in Okrun at 19 DPP inoculation, which was significant (P < 0.05) from Tamrun OL 02 only (Table 7).
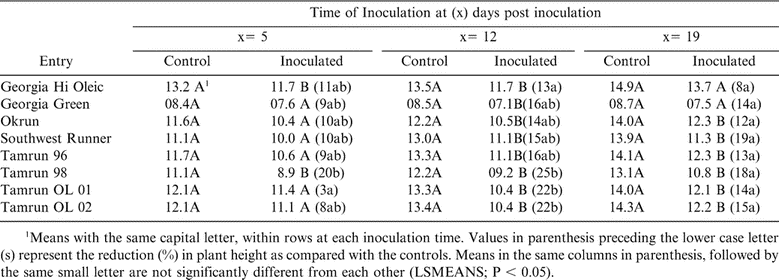
Plant Height
When comparing non-inoculated controls and inoculated plants at 5 DPP, plant height of Tamrun OL 01 was reduced the least (3%), which was significant (P < 0.05) from Tamrun-98 only (Table 8). At 12 DPP inoculation, plant height of Georgia Hi Oleic was least affected (13%) which was significant (P < 0.05) from Tamrun-98, Tamrun OL-01, and Tamrun OL-02 (Table 8). At 19 DPP inoculation, the percentage reduction of plant height was not significant (P < 0.05) among the cultivars (Table 8).
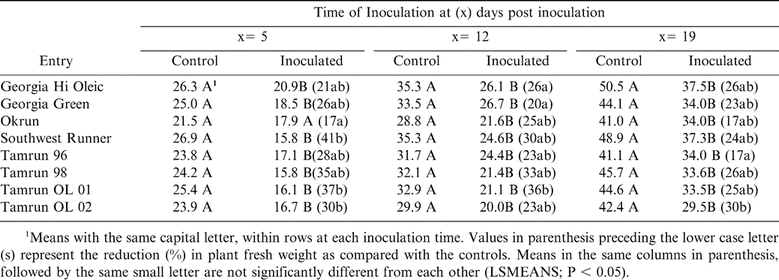
Plant Fresh Weight
Fresh weights of peanut cultivars of TSWV-mechanically-inoculated and non-inoculated controls were presented in Table 9. Inoculations at 5 DPP showed that Okrun had the least reduction (17%) of fresh weight which was significant (P < 0.05) from Southwest Runner. When plants were inoculated at 12 DPP, Tamrun OL-01 exhibited the most reduction (36%) in fresh weight, which was significant (P < 0.05) from Georgia Hi Oleic, Georgia Green, Okrun, and Tamrun-96 (Table 9). When plants inoculated at 19 DPP, the least reduction (17%) in fresh weight occurred on Tamrun-96 which was significant (P < 0.05) from the other cultivars (Table 9). Generally, across all three times of inoculation, significant reductions in fresh weight were observed in all cultivars except Okrun when inoculated at 5 DPP (Table 9).
Relative Water Content
When peanut cultivars were inoculated at 5 DPP, significant differences in relative water content (RWC) were observed in Georgia Green and Southwest Runner compared to non-inoculated plants (Table 10). At 12 and 19 DPP inoculation, no significant differences in RWC were observed between treated and untreated plants with the exception of Okrun at the12 DPP inoculation, and Tamrun-96 at the 19 DPP inoculation (Table 10).
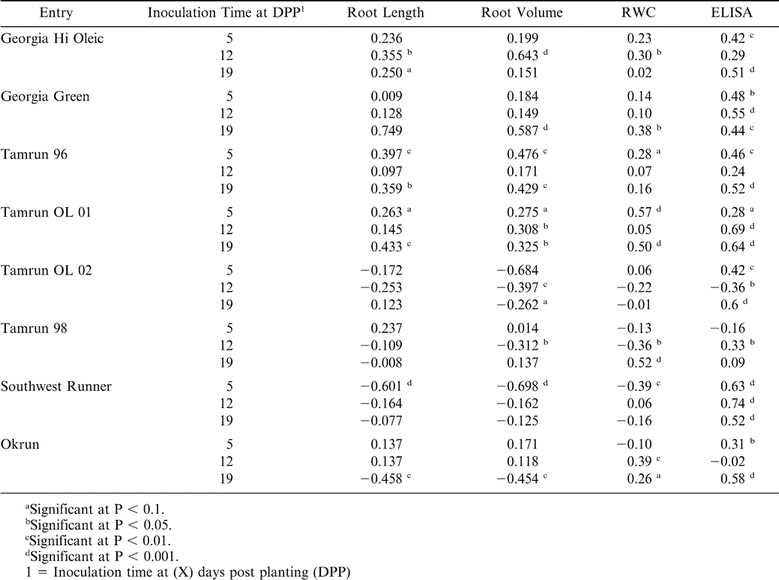
Positive correlations were obtained between DSI and ELISA values among most inoculated cultivars (Table 11). Correlation coefficient between DSI and root length, root volume, and RWC varied across cultivars and times of inoculation. The positive correlation coefficient values between DSI and root volume or root length in the cultivars Georgia Hi Oleic, Georgia Green, Tamrun 96, and Tamrun OL01 imply resistance or tolerance to mechanical inoculations with TSWV (Table 11). The negative correlation coefficients between DSI and root length or root volume in the cultivars Tamrun-98 and Tamrun OL-02, Southwest Runner, and Okrun imply lack of resistance or tolerance to mechanical inoculations with TSWV (Table 11). Also, the positive correlation coefficient values between DSI and RWC in the cultivars Georgia Hi Oleic, Georgia Green, Tamrun-96, and Tamrun OL-01, imply resistance or tolerance to mechanical inoculations with TSWV (Table 11).
Under controlled conditions, Pereira et al. (1995), reported the response of Florunner and Southern Runner against TSWV when seven quantitative growth parameters (e.g. main stem length, the two cotyledonary stem lengths, the two primary stem lengths, pod and peg number, canopy fresh weight, and root fresh weight) were determined to elucidate the impact of mechanical inoculation on these cultivars at two growth stages. No significant differences were observed among the two cultivars. Hoffmann et al. (1998) reported that mechanical inoculation of TSWV alone or co-inoculation with Peanut mottle potyvirus (PMV) to six peanut cultivars (GK7, NC7, VC1, Florunner, Southern Runner, and Spanco) did not reveal any significant differences in disease progress. Mandal et al. (2002) studied the reaction of three peanut cultivars and one breeding line against an isolate of TSWV. Their data were limited only to detection and distribution of the virus on these inoculated plants at two temperature regimes, based on ELISA. In this study, we obtained a detailed account of plant growth parameters that included; root length, root volume, plant height, fresh and dry weight. Disease severity index, relative water content, and virus titer were also evaluated on the eight cultivars inoculated mechanically at three time intervals.
Screening for plant resistance to viral agents via mechanical inoculation can provide useful information on genetic variations within the peanut germplasm. The identification of such genetic variations is important in breeding efforts to develop genotypes with resistance to TSWV. Mechanical inoculation, to screen for resistance under controlled conditions can be performed throughout the year, regardless of seasonal variations. However, it has the disadvantage of excluding the role of the vector in determining the observed reaction.
Screening peanut genotypes under field conditions has the advantage of identifying inherent viral resistance, and possibly vector preference and/or genotypes resistant to the thrips that can also provide indirect strategies for disease management to TSWV. However, the disadvantages of field screening include; limiting the evaluation to the growing season, variation of TSWV pressure from one location to another and yearly variation. In addition, the field environment with its complex biotic and abiotic agents may interfere either positively or negatively with identification of resistance. Our data of the field and greenhouse evaluations indicate the potential usefulness of the peanut cultivars Georgia Green, Tamrun-96, and Georgia Hi Oleic for managing TSWV in peanut production in Southwestern United States.
Literature Cited
Black M. C. 1987 Pathological aspect of TSWV in south Texas. Proc. Am. Peanut Res. Educ. Soc 19 : 66 .
Black M. C. , Lummus P. F. , Smith D. H. , and Demski J. W. 1986 An epidemic of spotted wilt disease in south Texas peanuts in 1985. Proc. Am. Peanut. Res. Educ. Soc 18 : 66 .
Clark M. F. and Adam A. N. 1977 Characterization of the microblate method of enzyme-linked immunosorbant assay for the detection of plant viruses. J. Gen. Virol 34 : 475 – 483 .
Culbreath A. K. , Csinos A. S. , Bertrand P. F. , and Demski J. W. 1991 Tomato spotted wilt virus epidemic in flue-cured tobacco in Georgia. Plant Dis 75 : 483 – 485 .
Culbreath A. K. , Todd J. W. , and Demski J. W. 1990 Epidemiology of TSWV on peanut. Proc. Am. Peanut Res. Educ. Soc 22 : 81 .
Culbreath A. K. , Todd J. W. , Demski J. W. , and Chamberlin J. R. 1992 Disease progress of spotted wilt in peanut cultivars Florunner and Southern Runner. Phytopathology 82 : 766 – 771 .
Culbreath A. K. , Todd J. W. , Gobret D. W. , Branch W. D. , and Shokes F. M. 1996 Beyond Southern Runner: The next generation of field resistance to Tomato spotted wilt virus. Proc. Am. Peanut Res. Educ. Soc 28 : 50 .
Culbreath A. K. , Todd J. W. , Gobret D. W. , Brown S. L. , Baldwin J. , Pappu H. , and Shokes F. M. 2000 Reaction of peanut cultivars to spotted wilt. Peanut Sci 27 : 35 – 39 .
Culbreath A. K. , Todd J. W. , Gobret D. W. , Brown S. L. , Baldwin J. , Pappu H. , Holbrook C. , and Shokes F. M. 1999 Response of early, medium, and late peanut breeding lines to field epidemic of tomato spotted wilt. Peanut Sci 26 : 100 – 106 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Branch W. D. , Sprenkel R. K. , Shokes F. M. , and Demski J. W. 1996 Disease progress of Tomato spotted wilt virus in selected peanut cultivars and advanced breeding lines. Plant Dis 80 : 70 – 73 .
Culbreath A. , Todd J. W. , and Brown S. L. 2003 Epidemiology and management of Tomato spotted wilt in peanut. Annu. Rev. Phytopathology 41 : 53 – 75 .
Erikson P. I. , Ketring D. , and Stone J. F. 1991 Response of internal tissue water balance of peanut to soil water. Agron. J 83 : 248 – 253 .
Garcia L. E. , Brandenburg R. L. , and Bailey J. E. 2000 Incidence of Tomato spotted wilt virus (Bunyaviridae) and tobacco thrips in Virginia-type peanuts in North Carolina. Plant Dis 84 : 459 – 464 .
German L. T. , Ulman D. E. , and Moyer J. E. 1992 Tospoviruses: Diagnosis, molecular biology, phylogency, and vector relationships. Annu. Rev. Phytopath 30 : 315 – 348 .
Goldback R. and Peters D. 1996 Molecular and biological aspects of Tospoviruses. In Elliott R. M. ed. The Bunyaviridae Plenum Press New York .
Hagan A. K. , Weeks J. R. , French J. C. , Gudauskas R. T. , Mullen J. M. , Gazaway W. S. , and Shelby R. 1990 Tomato spotted wilt virus in peanut in Alabama. Plant Dis 74 : 615 .
Halliwel R. S. and Philley G. 1974 Spotted wilt of peanut in Texas. Plant Dis. Reporter 58 : 23 – 25 .
Heinzman C. D. and Eilrich G. L. 1977 Technique for measuring vegetative volume and seed yield in soybeans. Can. J. Plant Sci 57 : 613 – 614 .
Hoffmann K. , Geske S. M. , and Moyer J. W. 1998 Pathogenesis of Tomato spotted wilt virus in peanut plants dually infected with peanut mottle virus. Plant Dis 82 : 610 – 614 .
Hunger R. M. , Sherwood J. L. , Smith E. L. , and Armitage C. R. 1991 Symptomatology and enzyme-linked immunosorbent assay used to facilitate breeding for resistance to wheat soilborne mosaic. Crop Sc 31 : 900 – 905 .
Ketring D. L. 1984 Root diversity among peanut genotypes. Crop Science 24 : 229 – 232 .
Kokalis-Burelle N. , Porter D. M. , Rodriguez-Kabana R. , Smith D. H. , and Subrahmanyam P. 1997 Compendium of peanut diseases. 2nd Ed. APS Press Inc St. Paul, MN .
Lyerly J. H. , Stalker H. T. , Moyer J. W. , and Hoffman K. 2002 Evaluation of Arachis species for resistance to tomato spotted wilt virus. Peanut Sci 29 : 79 – 84 .
Mandal B. , Pappu H. R. , Culbreath A. , Holbrook C. , and Gorbet D. 2002 Differential response of selected peanut (Arachis hypogaea) genotypes to mechanical I inoculation by Tomato spotted wilt virus. Plant Dis 86 : 939 – 944 .
Mandal B. , Pappu H. R. , and Culbreath A. K. 2001 Factors affecting mechanical transmission of Tomato spotted wilt virus to peanut (Arachis hypogaea). Plant Dis 85 : 1259 – 1263 .
Mitchell F. L. and Smith J. W. 1991 Epidemiology of Tomato spotted wilt virus relative to thrips populations. 46 – 52 In Hsu H. and Lawson R. H. eds. Virus-Thrips-Hosts- Interaction of Tomato spotted wilt virus Proceedings of USDA workshop, USDA-ARS, ARS-87.
Mulder P. G. , Cole C. L. , Karner M. A. , and Bolte J. R. 1991 Seasonal prevalence of the thysanoptera in an Oklahoma peanut ecosystem and potential for Tomato spotted wilt virus. Southwestern Entomolo 16 : 108 – 116 .
Mulder P. G. , Jackson K. E. , and Damicone J. P. 2001 Effects of peanut variety and insecticides on thrips population and transmission of Tomato spotted wilt virus. Proc. Am. Peanut Res. Educ. Soc 33 : 71 .
Pereira M. J. , Pfahler P. L. , Knauft D. A. , and Simone G. W. 1995 Cultivar differences in Tomato spotted wilt virus tolerance in peanut. Proc. Soil Crop Sci 54 : 12 – 16 .
Peters D. 1998 An updated list of plant species susceptible to tospoviruses. 107 – 110 In Proc. Int. Symp. Tospovirus Thrips flora vegetable crops, 4th Wageningen The Netherlands .
SAS Institute 1985 SAS User's Guide: Statistics, Vers. 5. SAS Inst Cary, NC .
Shaner G. and Finney R. E. 1977 The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology 67 : 1051 – 1056 .
Sherwood J. L. and Melouk H. A. 1995 Viral diseases and their management. 59 – 63 In Melouk H. A. and Shokes F. M. eds. Peanut Health Management APS Press St. Paul, MN .
Simpson C. E. , Baring M. R. , Schubert A. M. , Melouk H. A. , Black M. C. , Lopez Y. , and Keim K. A. 2003 Registration of ‘Tamrun OL-01’ peanut. Crop Sci 43 : 2298 .
Smith O. D. , Simpson C. E. , Black M. C. , and Besler B. A. 1998 Registration of Tamrun-96 peanut. Crop Sci 38 : 1403 .
Steel R. G. , Torrie J. H. , and Dickey D. A. 1997 Principles and procedures of statistics a biometrical approach. 3rd Ed. McGraw Hill, Inc New York .
Todd J. W. , Culbreath A. , Demski J. W. , and Beshear R. 1990 Thrips as vectors of TSWV. Proc. Am. Peanut Res. And Educ. Soc 22 : 81 .
Wells M. L. , Culbreath A. , Todd J. , Brown S. L. , and Gorbet D. W. 2002 A regression approach for comparing field resistance of peanut cultivars to Tomato spotted wilt tospovirus. Crop Prot 21 : 467 – 474 .
Wijkamp I. , van Lent J. , Kormelink R. , Goldbach R. , and Peters D. 1993 Multipication of Tomato spotted wilt virus in its vector Frankliniella occidentalis. J. of Gen. Virol 74 : 341 – 349 .
Notes
- 1Mohammed A. AL-Saleh, Former Graduate Student [^]
- 2*Hassan A. Melouk, Plant Pathologist and Professor, USDA-ARS, Plant Science Research Laboratory, Department of Entomology and Plant Pathology, Oklahoma State University, Stillwater, OK 74078. [^]
- 3Phillip Mulder, Professor of Entomology, Department of Entomology and Plant Pathology, Oklahoma State University, Stillwater, OK 74078. [^]
Author Affiliations